NOTES: 2.1 - Intro to Chemistry
... Life is made up of MATTER! ● MATTER can be defined as anything that has mass and takes up space…this includes individual atoms!! ...
... Life is made up of MATTER! ● MATTER can be defined as anything that has mass and takes up space…this includes individual atoms!! ...
1 Types of Chemical Reactions
... 2 KI + Pb(NO3)2 See chapter 4, lesson 16 , Saunders Interactive Chemistry ...
... 2 KI + Pb(NO3)2 See chapter 4, lesson 16 , Saunders Interactive Chemistry ...
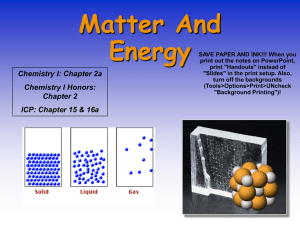
Matter and Energy
... • Extensive properties are dependent upon the amount of substance present. Ex- mass, length • Intensive property is independent of the amount of substance present. Ex- density, temperature ...
... • Extensive properties are dependent upon the amount of substance present. Ex- mass, length • Intensive property is independent of the amount of substance present. Ex- density, temperature ...
Exam Review – Part 1
... If enough oxygen is present, they will burn completely and release all of their energy and produce only two products: carbon dioxide and water (complete combustion) word equation ...
... If enough oxygen is present, they will burn completely and release all of their energy and produce only two products: carbon dioxide and water (complete combustion) word equation ...
Volatile Organic Compounds
... Volatile Organic Compounds (VOC's) are organic (carbon-containing) chemicals that have a high vapours pressure and easily form vapours at normal temperature and pressure. VOC’s include trichloroethylene, benzene, toluene, styrene, acetone, ethyl benzene, mixed xylenes methyl ethyl ketone, alcohols, ...
... Volatile Organic Compounds (VOC's) are organic (carbon-containing) chemicals that have a high vapours pressure and easily form vapours at normal temperature and pressure. VOC’s include trichloroethylene, benzene, toluene, styrene, acetone, ethyl benzene, mixed xylenes methyl ethyl ketone, alcohols, ...
The Chemical Basis of Life
... Isotopes are atoms of an element that all have the same number of protons, but have different numbers of neutrons. Radioisotopes – radioactive decay The time it takes for half of a radioactive substance to decay is called its half-life. ...
... Isotopes are atoms of an element that all have the same number of protons, but have different numbers of neutrons. Radioisotopes – radioactive decay The time it takes for half of a radioactive substance to decay is called its half-life. ...
unit 2 - chemistry
... corn oil, safflower oil, sunflower oil, cottonseed oil, sesame oil, soybean oil help reduce cholesterol 3.Proteins - C, H, O, N body structure, physiological activities ( catalysts) amino acids – 20 different and are the building blocks - amino group NH2 - carboxyl group COOH - side chain – ...
... corn oil, safflower oil, sunflower oil, cottonseed oil, sesame oil, soybean oil help reduce cholesterol 3.Proteins - C, H, O, N body structure, physiological activities ( catalysts) amino acids – 20 different and are the building blocks - amino group NH2 - carboxyl group COOH - side chain – ...
Chapter 1: Chemistry and You
... This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests and quizzes, homework, etc. Ask ques ...
... This review sheet is a list of topics and sample practice problems only. The practice problems are good representation of what to expect on the midterm, but it is not enough to just study from the review. You need to look over your notes, old review sheets, tests and quizzes, homework, etc. Ask ques ...
2.1 The Nature of Matter - Sonoma Valley High School
... Some elements have isotopes, with different #s of neutrons and different mass. All isotopes of an element have the same chemical properties b/c their electrons are the same. ...
... Some elements have isotopes, with different #s of neutrons and different mass. All isotopes of an element have the same chemical properties b/c their electrons are the same. ...
First Semester Final - Review Questions
... 47. Atoms combine to form molecules by sharing electrons to form covalent or metallic bonds or by exchanging electrons to form ionic bonds. 48. The chemical bonds between atoms in molecules such as H2, CH4, NH3, H2CCH2, N2, Cl2, and many large biological molecules are ionic. 49. Large molecules (pol ...
... 47. Atoms combine to form molecules by sharing electrons to form covalent or metallic bonds or by exchanging electrons to form ionic bonds. 48. The chemical bonds between atoms in molecules such as H2, CH4, NH3, H2CCH2, N2, Cl2, and many large biological molecules are ionic. 49. Large molecules (pol ...
PowerPoint
... All one kind of atom. Compounds are substances that can be broken down by chemical methods • When they are broken down, the pieces have completely different properties than the compound. • Made of molecules- two or more atoms ...
... All one kind of atom. Compounds are substances that can be broken down by chemical methods • When they are broken down, the pieces have completely different properties than the compound. • Made of molecules- two or more atoms ...
heats of reaction
... 2.) Write balanced heat of formation reactions for each of the following. Do not forget to include the ∆Hf values from the table for the phases listed. Use your appendix (∆H values) to write balanced formation equations determine whether these reactions are exo or endothermic. a. ...
... 2.) Write balanced heat of formation reactions for each of the following. Do not forget to include the ∆Hf values from the table for the phases listed. Use your appendix (∆H values) to write balanced formation equations determine whether these reactions are exo or endothermic. a. ...
Valence Electrons and Chemical Bonding
... eight electrons in their outer energy level or, in the case of elements 1-5, two in their outer shell level. ...
... eight electrons in their outer energy level or, in the case of elements 1-5, two in their outer shell level. ...
Atomic Structure, Molecular Structure & Bonding
... – H is never central; C is often central 3. Draw in electrons to fulfill octet and duet rules – C “likes” 8 electrons; H “likes” 2 electrons 4. Count ve-’s and compare to #2 5. If too many e-’s, make a double bond 6. Calculate formal charge (FC) to double check structure – No or low FCs (e.g. +1) mo ...
... – H is never central; C is often central 3. Draw in electrons to fulfill octet and duet rules – C “likes” 8 electrons; H “likes” 2 electrons 4. Count ve-’s and compare to #2 5. If too many e-’s, make a double bond 6. Calculate formal charge (FC) to double check structure – No or low FCs (e.g. +1) mo ...
HIBBING COMMUNITY COLLEGE
... 42. name alkenes and alkynes and draw their structures. 43. identify and name isomers. 44. name and describe cyclic compounds.. 45. recognize compounds containing the major organic functional groups. 46. explain the concept of thermal equilibrium and the chemistry of fire. 47. assign oxidation numbe ...
... 42. name alkenes and alkynes and draw their structures. 43. identify and name isomers. 44. name and describe cyclic compounds.. 45. recognize compounds containing the major organic functional groups. 46. explain the concept of thermal equilibrium and the chemistry of fire. 47. assign oxidation numbe ...
Chemistry is a material science
... definition of matter leads to a common property of matter, namely, _______. Density is equal to the mass divided by the volume. The “__________” of matter is its composition. This addresses the question what ______ of atoms and molecules are present in this matter. Matter composed of _______ kind of ...
... definition of matter leads to a common property of matter, namely, _______. Density is equal to the mass divided by the volume. The “__________” of matter is its composition. This addresses the question what ______ of atoms and molecules are present in this matter. Matter composed of _______ kind of ...
Science 9
... What are the two types of reactions that involve a gain or a release of energy? Explain each type. ...
... What are the two types of reactions that involve a gain or a release of energy? Explain each type. ...
a) air c) milk f) beer
... compounds for a fixed mass of the other element are in ratios of small whole numbers. ...
... compounds for a fixed mass of the other element are in ratios of small whole numbers. ...
2. Chemistry of Living Things Outline
... catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present fo ...
... catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but must be present fo ...
Chemistry of Living Things Outline
... reaction they catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but ...
... reaction they catalyze. In organisms, _____________ allow the chemical reactions of ______________ to take place more efficiently than they otherwise would at body temperature. For example, amino acids are produced from protein digestion. The enzymes needed for this reaction are not changed but ...
Grade 11 Unit 8 - Amazon Web Services
... uniqueness of the carbon atom is the basis of all life. The choice of this atom with its designed characteristics is indeed a mark of an omnipotent Creator. No work of chance could have happened upon the combination necessary to produce life. David proclaimed in Psalm 19:1, “The heavens declare the ...
... uniqueness of the carbon atom is the basis of all life. The choice of this atom with its designed characteristics is indeed a mark of an omnipotent Creator. No work of chance could have happened upon the combination necessary to produce life. David proclaimed in Psalm 19:1, “The heavens declare the ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.