chemistry form iii - Covington Latin School
... Course Description: Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry; stoichiometry; atomic theory; the ch ...
... Course Description: Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry; stoichiometry; atomic theory; the ch ...
STUDY GUIDE for DIGESTION and NUTRITION
... Check off the box once you have studied and understand the concept ...
... Check off the box once you have studied and understand the concept ...
Chemical Bonding
... • Proteins are complex, organic compounds made up of amino acids. • Lipids are organic compounds that contains fats, oils, waxes, phospholipids, and steroids. They are made of fatty acid chains. • Nucleic acids are large, complex molecules that contain the hereditary information for all living thing ...
... • Proteins are complex, organic compounds made up of amino acids. • Lipids are organic compounds that contains fats, oils, waxes, phospholipids, and steroids. They are made of fatty acid chains. • Nucleic acids are large, complex molecules that contain the hereditary information for all living thing ...
Organic Chemistry and Medicine
... Organic Chemistry and Medicine Or·gan·ic \ȯr-ˈga-nik\ adj : 1. of, relating to, or arising in a bodily organ; 2. of, relating to, or containing carbon compounds; 3. relating to, being, or dealt with by a branch of chemistry concerned with the carbon compounds of living beings and most other carbon ...
... Organic Chemistry and Medicine Or·gan·ic \ȯr-ˈga-nik\ adj : 1. of, relating to, or arising in a bodily organ; 2. of, relating to, or containing carbon compounds; 3. relating to, being, or dealt with by a branch of chemistry concerned with the carbon compounds of living beings and most other carbon ...
Name:______ Chemistry 114 First Hour Exam
... aromatic ring. Circle and identify each of these groups in your structure. Include all hydrogens and all lone pairs of electrons in your structure because it has to make complete chemical sense. ...
... aromatic ring. Circle and identify each of these groups in your structure. Include all hydrogens and all lone pairs of electrons in your structure because it has to make complete chemical sense. ...
Ch. 2 The Chemistry of Life
... - Base – a _____________ that produces _____________ ions in solution - _________, or alkaline, solutions have __________ concentrations of _____ ions than pure ________ & have ____ values _____________ - The ________ the concentration of _____ ions, the _________ the ____ value (up to ___) - Buffer ...
... - Base – a _____________ that produces _____________ ions in solution - _________, or alkaline, solutions have __________ concentrations of _____ ions than pure ________ & have ____ values _____________ - The ________ the concentration of _____ ions, the _________ the ____ value (up to ___) - Buffer ...
Organic Chemistry
... • 1780: Organic compounds are very complex and only obtained from living sources (vitalism 生机说) • Vitalism: Belief that a "magic" vital force, present in plants and animals, is necessary for the synthesis of organic compounds • 1789: Antoine Laurent Lavoisier observed that organic compounds are comp ...
... • 1780: Organic compounds are very complex and only obtained from living sources (vitalism 生机说) • Vitalism: Belief that a "magic" vital force, present in plants and animals, is necessary for the synthesis of organic compounds • 1789: Antoine Laurent Lavoisier observed that organic compounds are comp ...
Chapter 8: Chemical Reactions and Physical Changes
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
... • Mass number: total protons and neutrons in an atom’s nucleus • Atomic mass: the average mass of a sample of atoms of that element found in nature • Periodic table: chart that arranges elements by atomic number into rows and columns according to similarities in their properties ...
Chapter 6: Chemistry in Biology
... When molecules come close together, the attractive forces between slightly positive and negative regions pull on the molecules and hold them together. ...
... When molecules come close together, the attractive forces between slightly positive and negative regions pull on the molecules and hold them together. ...
Organic Chemistry
... Nonsystematic names do not indicate the structure of the compound. Nonsystematic names are common for complex molecules, which includes most natural products. Thus, the informally named lysergic acid diethylamide is systematically named (6aR,9R)-N,N-diethyl7-methyl-4,6,6a,7,8,9-hexahydroindolo-[4,3- ...
... Nonsystematic names do not indicate the structure of the compound. Nonsystematic names are common for complex molecules, which includes most natural products. Thus, the informally named lysergic acid diethylamide is systematically named (6aR,9R)-N,N-diethyl7-methyl-4,6,6a,7,8,9-hexahydroindolo-[4,3- ...
+ O2 (g)
... Combustion of Methane, Balanced To show the reaction obeys the Law of Conservation of Mass, the equation must be balanced. CH4(g) + O2(g) ➜ CO2(g) + H2O(g) ! ...
... Combustion of Methane, Balanced To show the reaction obeys the Law of Conservation of Mass, the equation must be balanced. CH4(g) + O2(g) ➜ CO2(g) + H2O(g) ! ...
Microbial Metabolism
... factors • Micronutrients: Metals and metalloids – Generally not necessary to add to medium – Deficiencies can arise when medium constituents are very pure ...
... factors • Micronutrients: Metals and metalloids – Generally not necessary to add to medium – Deficiencies can arise when medium constituents are very pure ...
Biology project Lz
... compound known to occur only in living organisms, from cyanogen.[citation needed] A more decisive experiment was Wöhler's 1828 synthesis of urea from the inorganic salts potassium cyanate and ammonium sulfate. Urea had long been considered an "organic" compound, as it was known to occur only in the ...
... compound known to occur only in living organisms, from cyanogen.[citation needed] A more decisive experiment was Wöhler's 1828 synthesis of urea from the inorganic salts potassium cyanate and ammonium sulfate. Urea had long been considered an "organic" compound, as it was known to occur only in the ...
Oppgave 5.
... Predict the 13C-NMR chemical shift values for the following compounds. For compound a): Use tables and charts posted or displayed in the texbook AND Chemdraw. For compounds b-e): use Chemdraw only. a) b) c) d) e) ...
... Predict the 13C-NMR chemical shift values for the following compounds. For compound a): Use tables and charts posted or displayed in the texbook AND Chemdraw. For compounds b-e): use Chemdraw only. a) b) c) d) e) ...
Group IV Elements
... Sn,Pb have been known since long time, because they can be just melted out of their minerals Ge was discovered after its existance has been predicted. It is purified from coal and zinc ore concentrates ...
... Sn,Pb have been known since long time, because they can be just melted out of their minerals Ge was discovered after its existance has been predicted. It is purified from coal and zinc ore concentrates ...
Efficiently Studying Organic Chemistry
... Most text books are designed to accompany one or more specific courses and occasionally may even replace a lecture – if thoroughly worked through. They provide a step-bystep introduction to the given topic and are usually on a high educational level. A much smaller number of text books rather show t ...
... Most text books are designed to accompany one or more specific courses and occasionally may even replace a lecture – if thoroughly worked through. They provide a step-bystep introduction to the given topic and are usually on a high educational level. A much smaller number of text books rather show t ...
Summary from Organic Chemistry Packet:
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
Microsoft Word
... ecological balance. Way to minimize the consumption of energy and raw materials used in the synthesis must be devised so the optimal value of resources could be realized. There by environmentally benign products are obtained at afford able costs such that a concept though has received enormous atten ...
... ecological balance. Way to minimize the consumption of energy and raw materials used in the synthesis must be devised so the optimal value of resources could be realized. There by environmentally benign products are obtained at afford able costs such that a concept though has received enormous atten ...
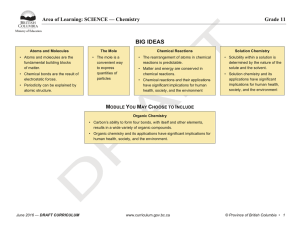
BIG IDEAS - BC Curriculum - Province of British Columbia
... • Carbon’s ability to form four bonds, with itself and other elements, results in a wide variety of organic compounds. • Organic chemistry and its applications have significant implications for human health, society, and the environment. ...
... • Carbon’s ability to form four bonds, with itself and other elements, results in a wide variety of organic compounds. • Organic chemistry and its applications have significant implications for human health, society, and the environment. ...
Organic compounds are covalent compounds composed of carbon
... molecules made by living organisms are nucleic acids. Nucleic acids are bio-chemicals made up of nucleotides. Nucleotides are molecules made of carbon, hydrogen, oxygen, nitrogen, and phosphorus atoms. ...
... molecules made by living organisms are nucleic acids. Nucleic acids are bio-chemicals made up of nucleotides. Nucleotides are molecules made of carbon, hydrogen, oxygen, nitrogen, and phosphorus atoms. ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.