... goes from a non-equilibirium position to an equilibrium position. It is related to the standard energy as follows (4 pts) Choice A: The addition of a phosphate to a sugar (e.g. conversion of glucose to glucose-6-P) is unfavorable, with a standard energy change, ∆Go of +15 kJ/mol, yet reactions of th ...
vitamine
... – Coenzymes are lost upon dialysis because they bind the enzyme loosely. – When they bind enzymes tightly, they are considered prosthetic group. – The term cofactor includes such compounds but also includes other molecules such as metal ions that may be necessary for enzyme activity. ...
... – Coenzymes are lost upon dialysis because they bind the enzyme loosely. – When they bind enzymes tightly, they are considered prosthetic group. – The term cofactor includes such compounds but also includes other molecules such as metal ions that may be necessary for enzyme activity. ...
Inhibition of Carbonic Anhydrase Results in Blood Lactate
... induced diabetic rats. Because diabetes results in a situation in which glucose homeostasis is impaired and that it may account for the excess conversion of glucose to lactate, due to decreased rate of oxidative metabolism of glucose, including reduced rates of muscle glycogen synthesis, as suggeste ...
... induced diabetic rats. Because diabetes results in a situation in which glucose homeostasis is impaired and that it may account for the excess conversion of glucose to lactate, due to decreased rate of oxidative metabolism of glucose, including reduced rates of muscle glycogen synthesis, as suggeste ...
BIOSYNTHESIS OF AMINO ACIDS, NUCLEOTIDES, AND
... of cells, and in extracellular fluids in higher organisms, one or both of these amino acids are present at higher concentrations—sometimes an order of magnitude or more higher—than other amino acids. An Escherichia coli cell requires so much glutamate that this amino acid is one of the primary solut ...
... of cells, and in extracellular fluids in higher organisms, one or both of these amino acids are present at higher concentrations—sometimes an order of magnitude or more higher—than other amino acids. An Escherichia coli cell requires so much glutamate that this amino acid is one of the primary solut ...
Autotrophic CO2 fixation via the reductive tricarboxylic acid cycle in
... genome sequence (A. aeolicus) or carbon isotopic measurements (Tc. ruber) both organisms were previously suspected to use this pathway, but direct evidence had been lacking (Deckert et al., 1998; Jahnke et al., 2001). Isocitrate dehydrogenase, as well as malate dehydrogenase, was found to be NAD(H)- ...
... genome sequence (A. aeolicus) or carbon isotopic measurements (Tc. ruber) both organisms were previously suspected to use this pathway, but direct evidence had been lacking (Deckert et al., 1998; Jahnke et al., 2001). Isocitrate dehydrogenase, as well as malate dehydrogenase, was found to be NAD(H)- ...
Internal expression of Yarrowia NDH2
... mitochondrial inner membrane. We thus inferred that an internal alternative enzyme could be sufficient to carry out electron transfer from NADH to the respiratory chain ubiquinone pool in the absence of complex I. Plasmid pUB22 (Fig. 2), which contains the NDH2i gene encoding an internal version of ...
... mitochondrial inner membrane. We thus inferred that an internal alternative enzyme could be sufficient to carry out electron transfer from NADH to the respiratory chain ubiquinone pool in the absence of complex I. Plasmid pUB22 (Fig. 2), which contains the NDH2i gene encoding an internal version of ...
Plant Mitochondrial Electron Transfer and Molecular
... (Taiz and Zeiger, 1991).Severa1 types of reduced carbon compounds, including fatty acids, organic acids, and amino acids, can serve as the primary reducing substrates for plant respiration. However, the most common substrate used by plant tissues for respiration is carbohydrate(CHpO).The complete ox ...
... (Taiz and Zeiger, 1991).Severa1 types of reduced carbon compounds, including fatty acids, organic acids, and amino acids, can serve as the primary reducing substrates for plant respiration. However, the most common substrate used by plant tissues for respiration is carbohydrate(CHpO).The complete ox ...
Fasting induces ketoacidosis and hypothermia in PDHK2/PDHK4
... complex is inactivated by phosphorylation by PDHKs (pyruvate dehydrogenase kinases) to conserve three carbon compounds for the production of glucose [2]. The four PDHK isoenzymes responsible for phosphorylating the PDH complex are expressed in a tissue-specific manner [3– 5]. Among the four, PDHK2 a ...
... complex is inactivated by phosphorylation by PDHKs (pyruvate dehydrogenase kinases) to conserve three carbon compounds for the production of glucose [2]. The four PDHK isoenzymes responsible for phosphorylating the PDH complex are expressed in a tissue-specific manner [3– 5]. Among the four, PDHK2 a ...
Plant Mitochondrial Electron Transfer and Molecular
... (Taiz and Zeiger, 1991).Severa1 types of reduced carbon compounds, including fatty acids, organic acids, and amino acids, can serve as the primary reducing substrates for plant respiration. However, the most common substrate used by plant tissues for respiration is carbohydrate(CHpO).The complete ox ...
... (Taiz and Zeiger, 1991).Severa1 types of reduced carbon compounds, including fatty acids, organic acids, and amino acids, can serve as the primary reducing substrates for plant respiration. However, the most common substrate used by plant tissues for respiration is carbohydrate(CHpO).The complete ox ...
Chapter 1
... inhibition • With feedback inhibition, a product late in a series of enzyme-catalyzed reactions serves as an inhibitor for a previous allosteric enzyme earlier in the series • In this example, product F serves to inhibit the activity of enzyme E1 – Product F acts as a negative allosteric effector on ...
... inhibition • With feedback inhibition, a product late in a series of enzyme-catalyzed reactions serves as an inhibitor for a previous allosteric enzyme earlier in the series • In this example, product F serves to inhibit the activity of enzyme E1 – Product F acts as a negative allosteric effector on ...
as a PDF
... The natural portal of entry for the parasite is any region of the skin leading to an inflammation called chagoma. However, since the insect usually bites during sleeping, the face is preferred. In that case, the insect, during the blood meal deposits feces and urine and the victim transports this ma ...
... The natural portal of entry for the parasite is any region of the skin leading to an inflammation called chagoma. However, since the insect usually bites during sleeping, the face is preferred. In that case, the insect, during the blood meal deposits feces and urine and the victim transports this ma ...
Vitamins and Coenzymes
... • This glycoaldehyde is said to be activated because it is also a carbanion and readily undergoes nucleophilic addition to the carbonyl group of an aldose (erythrose 4phosphate here) • Following another deprotonation the nascent a-ketose is released from TPP ...
... • This glycoaldehyde is said to be activated because it is also a carbanion and readily undergoes nucleophilic addition to the carbonyl group of an aldose (erythrose 4phosphate here) • Following another deprotonation the nascent a-ketose is released from TPP ...
Organic Chemistry Notes Student
... A. Carbohydrates - ______________________________________ Monosaccharides are the simplest ______________________. • The carbohydrate monomers are ___________________ • A monosaccharide has a formula that is a multiple of CH2O ______________________ ...
... A. Carbohydrates - ______________________________________ Monosaccharides are the simplest ______________________. • The carbohydrate monomers are ___________________ • A monosaccharide has a formula that is a multiple of CH2O ______________________ ...
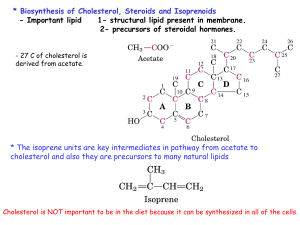
14steriod
... * Function : to keep the lipid soluble for transporting them between organs and also provide efficient mechanism for delivering their lipid contents to the tissues. * Composition of plasma lipoproteins - TG and cholesterol esters are mainly carried by lipoprotein (In the core : TG + cholesterol este ...
... * Function : to keep the lipid soluble for transporting them between organs and also provide efficient mechanism for delivering their lipid contents to the tissues. * Composition of plasma lipoproteins - TG and cholesterol esters are mainly carried by lipoprotein (In the core : TG + cholesterol este ...
S17 Cholesterol And Steroids Biosynthesis
... * Function : to keep the lipid soluble for transporting them between organs and also provide efficient mechanism for delivering their lipid contents to the tissues. * Composition of plasma lipoproteins - TG and cholesterol esters are mainly carried by lipoprotein (In the core : TG + cholesterol este ...
... * Function : to keep the lipid soluble for transporting them between organs and also provide efficient mechanism for delivering their lipid contents to the tissues. * Composition of plasma lipoproteins - TG and cholesterol esters are mainly carried by lipoprotein (In the core : TG + cholesterol este ...
Introduction to Nutrition and Metabolism, Third Edition
... ‘why?’ – by exploring the need for food and the uses to which that food is put in the body. Some discussion of chemistry and biochemistry is obviously essential in order to investigate the fate of food in the body, and why there is a continuous need for food throughout life. Therefore, in the follow ...
... ‘why?’ – by exploring the need for food and the uses to which that food is put in the body. Some discussion of chemistry and biochemistry is obviously essential in order to investigate the fate of food in the body, and why there is a continuous need for food throughout life. Therefore, in the follow ...
Presentazione standard di PowerPoint
... evaluated for the treatment of hepatic and metabolic disorders, including NAFLD5, 6. Two FXR-encoding genes have been identified, FXRα and FXR , although only F↓Rα senses bile acids in humans. FXRα is expressed mainly in the liver, intestine, kidney and adrenal glands, and at lower levels in adipose ...
... evaluated for the treatment of hepatic and metabolic disorders, including NAFLD5, 6. Two FXR-encoding genes have been identified, FXRα and FXR , although only F↓Rα senses bile acids in humans. FXRα is expressed mainly in the liver, intestine, kidney and adrenal glands, and at lower levels in adipose ...
Triglyceride Measurements: a Review of Methods and Interferences
... are stored or if ATP has been processed with other reagents such as in a lyophilized product. ADP can also be generated by the action of alkaline phosphatase at pH 7.0-9.0. Its effect can be negated with a reagent blank; but consumption of NADH during pre-incubation reduces the dynamic range of the ...
... are stored or if ATP has been processed with other reagents such as in a lyophilized product. ADP can also be generated by the action of alkaline phosphatase at pH 7.0-9.0. Its effect can be negated with a reagent blank; but consumption of NADH during pre-incubation reduces the dynamic range of the ...
Genetic Disorders Leading to Hypoglycaemia
... level. Glucose and other substrates, such as amino acids, by raising the intracytosolic ATP/ADP ratio in the β-cell of pancreas cause insulin secretion. In the β-cell, rise in ATP inhibits the plasma membrane sulphonylurea receptor1 (SUR1), leading to a sequence of events; closure of the KATP channe ...
... level. Glucose and other substrates, such as amino acids, by raising the intracytosolic ATP/ADP ratio in the β-cell of pancreas cause insulin secretion. In the β-cell, rise in ATP inhibits the plasma membrane sulphonylurea receptor1 (SUR1), leading to a sequence of events; closure of the KATP channe ...
Enzymes upload
... blocks enzyme bacteria use to build cell walls ◦ disulfiram (Antabuse) treats chronic alcoholism blocks enzyme that breaks down alcohol severe hangover & vomiting 5-10 minutes after drinking ...
... blocks enzyme bacteria use to build cell walls ◦ disulfiram (Antabuse) treats chronic alcoholism blocks enzyme that breaks down alcohol severe hangover & vomiting 5-10 minutes after drinking ...
Answer
... When this reaction is carried out in solution in a test tube via direct mixing of NADH with dissolved oxygen, the reaction releases a significant amount of heat. However, when the reaction occurs in mitochondria during respiration, it produces very little heat. Explain why the heat evolved is much l ...
... When this reaction is carried out in solution in a test tube via direct mixing of NADH with dissolved oxygen, the reaction releases a significant amount of heat. However, when the reaction occurs in mitochondria during respiration, it produces very little heat. Explain why the heat evolved is much l ...
Electron transport chain…
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Glycolysis
Glycolysis (from glycose, an older term for glucose + -lysis degradation) is the metabolic pathway that converts glucose C6H12O6, into pyruvate, CH3COCOO− + H+. The free energy released in this process is used to form the high-energy compounds ATP (adenosine triphosphate) and NADH (reduced nicotinamide adenine dinucleotide).Glycolysis is a determined sequence of ten enzyme-catalyzed reactions. The intermediates provide entry points to glycolysis. For example, most monosaccharides, such as fructose and galactose, can be converted to one of these intermediates. The intermediates may also be directly useful. For example, the intermediate dihydroxyacetone phosphate (DHAP) is a source of the glycerol that combines with fatty acids to form fat.Glycolysis is an oxygen independent metabolic pathway, meaning that it does not use molecular oxygen (i.e. atmospheric oxygen) for any of its reactions. However the products of glycolysis (pyruvate and NADH + H+) are sometimes disposed of using atmospheric oxygen. When molecular oxygen is used in the disposal of the products of glycolysis the process is usually referred to as aerobic, whereas if the disposal uses no oxygen the process is said to be anaerobic. Thus, glycolysis occurs, with variations, in nearly all organisms, both aerobic and anaerobic. The wide occurrence of glycolysis indicates that it is one of the most ancient metabolic pathways. Indeed, the reactions that constitute glycolysis and its parallel pathway, the pentose phosphate pathway, occur metal-catalyzed under the oxygen-free conditions of the Archean oceans, also in the absence of enzymes. Glycolysis could thus have originated from chemical constraints of the prebiotic world.Glycolysis occurs in most organisms in the cytosol of the cell. The most common type of glycolysis is the Embden–Meyerhof–Parnas (EMP pathway), which was discovered by Gustav Embden, Otto Meyerhof, and Jakub Karol Parnas. Glycolysis also refers to other pathways, such as the Entner–Doudoroff pathway and various heterofermentative and homofermentative pathways. However, the discussion here will be limited to the Embden–Meyerhof–Parnas pathway.The entire glycolysis pathway can be separated into two phases: The Preparatory Phase – in which ATP is consumed and is hence also known as the investment phase The Pay Off Phase – in which ATP is produced.↑ ↑ 2.0 2.1 ↑ ↑ ↑ ↑ ↑ ↑