Rules and Clues for Determining Oxidation Number
... Before determining the charge/oxidation state on the metal, though, one must first distinguish between inner and outer sphere ligands in the molecular formula. An inner sphere ligand may be neutral or charged. It occupies a distinct position in the coordination sphere of the metal. It interacts dire ...
... Before determining the charge/oxidation state on the metal, though, one must first distinguish between inner and outer sphere ligands in the molecular formula. An inner sphere ligand may be neutral or charged. It occupies a distinct position in the coordination sphere of the metal. It interacts dire ...
Chapter 2 Study Outline
... Fatty acids with hydrogen at every position along the carbon chain are saturated; those with one or more double bonds are called ______________ fats. ____________ contain glycerol, two fatty acids, and a phosphate group, and are important in cell structures. _______________ are complex ring structur ...
... Fatty acids with hydrogen at every position along the carbon chain are saturated; those with one or more double bonds are called ______________ fats. ____________ contain glycerol, two fatty acids, and a phosphate group, and are important in cell structures. _______________ are complex ring structur ...
1 Name Chapter 3 Reading Guide Nucleic Acids, Proteins, and
... c. Explain the difference between your answer for the time of (A) and (B). Disulfide bridges are necessary for protein tertiary structure and must form before the enzyme active site can reappear, but there are other chemical interactions, such as hydrogen bonding and hydrophobic interactions, that o ...
... c. Explain the difference between your answer for the time of (A) and (B). Disulfide bridges are necessary for protein tertiary structure and must form before the enzyme active site can reappear, but there are other chemical interactions, such as hydrogen bonding and hydrophobic interactions, that o ...
bioinorganic 1
... In virtually all nitrogen-containing biomolecules the nitrogen is in its fully reduced form i.e. in the same oxidation state as ammonia (NH3) or ammonium cation (NH4+). The source of the nitrogen (and the principle form of the element on Earth) is dinitrogen, N2. So living systems need to convert di ...
... In virtually all nitrogen-containing biomolecules the nitrogen is in its fully reduced form i.e. in the same oxidation state as ammonia (NH3) or ammonium cation (NH4+). The source of the nitrogen (and the principle form of the element on Earth) is dinitrogen, N2. So living systems need to convert di ...
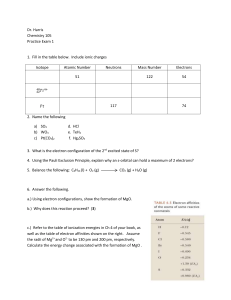
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 15. A photon with some energy Ep strikes a metal surface. An electron is ejected with a velocity of 5.00 Mm/s. The threshold frequency of the metal is 6.30 x 1013 s-1. What is the wavelength of the photon, in nm? Significant figures count. ...
... 15. A photon with some energy Ep strikes a metal surface. An electron is ejected with a velocity of 5.00 Mm/s. The threshold frequency of the metal is 6.30 x 1013 s-1. What is the wavelength of the photon, in nm? Significant figures count. ...
LN_atoms_etc
... Modern View of Atomic Structure Experiments by Thomson and Millikan confirmed the existence of electrons as the negatively charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the ce ...
... Modern View of Atomic Structure Experiments by Thomson and Millikan confirmed the existence of electrons as the negatively charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the ce ...
OXIDATION - REDUCTION
... weak acid that is oxidised to carbon dioxide CO2. Its anion is the oxalate ion C2O42, found in salts such as sodium oxalate. 9. Hydrogen sulfide, H2S – This gas can be used as a reductant as it can be oxidised to sulfur species in a higher oxidation state eg SO2. 10. Hydrogen peroxide, H2O2 – When ...
... weak acid that is oxidised to carbon dioxide CO2. Its anion is the oxalate ion C2O42, found in salts such as sodium oxalate. 9. Hydrogen sulfide, H2S – This gas can be used as a reductant as it can be oxidised to sulfur species in a higher oxidation state eg SO2. 10. Hydrogen peroxide, H2O2 – When ...
Ch. 3 Vocabs
... makes up proteins; a protein monomer peptide bond: the chemical bond that forms between the carboxyl group of one amino acid and the amino group of another amino acid polypeptide: a long chain of several amino acids enzyme: a type of protein or RNA molecule that speeds up metabolic reactions i ...
... makes up proteins; a protein monomer peptide bond: the chemical bond that forms between the carboxyl group of one amino acid and the amino group of another amino acid polypeptide: a long chain of several amino acids enzyme: a type of protein or RNA molecule that speeds up metabolic reactions i ...
Chemical reaction
... • Reactant: a compound or atom involved in a chemical reaction • Product: a compound formed by a chemical reaction ...
... • Reactant: a compound or atom involved in a chemical reaction • Product: a compound formed by a chemical reaction ...
bonding notes for votech
... Attraction between + nucleus and e- or between + and – ions Only valence e- involved in bonding Bonding occurs to have complete outermost energy levels – to become like noble gases ...
... Attraction between + nucleus and e- or between + and – ions Only valence e- involved in bonding Bonding occurs to have complete outermost energy levels – to become like noble gases ...
Zoology – The Chemical Basis of Animal Life
... single functional protein 4. Enzymes – mostly large, complex proteins that function as biological catalysts under specific conditions. (A special type of RNA can also function as an enzyme.) a.Enzymes do the following: 1) Break bonds and allow new bonds to form, facilitating chemical reactions 2) Sp ...
... single functional protein 4. Enzymes – mostly large, complex proteins that function as biological catalysts under specific conditions. (A special type of RNA can also function as an enzyme.) a.Enzymes do the following: 1) Break bonds and allow new bonds to form, facilitating chemical reactions 2) Sp ...
Introduction to Protein Science Architecture, Function
... Ex) enzymes in closely related species ...
... Ex) enzymes in closely related species ...
acids and bases
... The degree of acidity or alkalinity (basic) is important in organisms. The body must constantly maintain a near neutral pH (7) in the blood and body tissues. To do this, the body produces buffers that can neutralize acids. Acidic and basic conditions in the body occur due to different metabolic reac ...
... The degree of acidity or alkalinity (basic) is important in organisms. The body must constantly maintain a near neutral pH (7) in the blood and body tissues. To do this, the body produces buffers that can neutralize acids. Acidic and basic conditions in the body occur due to different metabolic reac ...
SURVEY OF BIOCHEMISTRY - Georgia Institute of Technology
... The arrangement of the regular structural elements and the positions of atoms in the protein are considered part of the ______. ...
... The arrangement of the regular structural elements and the positions of atoms in the protein are considered part of the ______. ...
UNIT 3 * Macromolecules and enzymes
... 10. Enzymes ________________ the activation energy of a chemical reaction. a. This makes the reaction occur more _________________ and ________________. 11. Enzymes are _______________________ protein molecules based on ______________. ...
... 10. Enzymes ________________ the activation energy of a chemical reaction. a. This makes the reaction occur more _________________ and ________________. 11. Enzymes are _______________________ protein molecules based on ______________. ...
Clinical biochemistry (0) introduction
... Composition of the cell A. Well-define nucleus (DNA),Mitochondria(oxidation reactions and energy), Vacuoles, Ribosomes(protein synthesis), Endoplasmic reticulum (many enzymes are located on) and Chemical constituents (simple and complex proteins, nucleoproteins, carbohydrates, lipid, intermediates ...
... Composition of the cell A. Well-define nucleus (DNA),Mitochondria(oxidation reactions and energy), Vacuoles, Ribosomes(protein synthesis), Endoplasmic reticulum (many enzymes are located on) and Chemical constituents (simple and complex proteins, nucleoproteins, carbohydrates, lipid, intermediates ...
Macromolecules - Teacher Pages
... Compounds containing C, H, O and often N, P, & S. Organic compounds make up all living things and are necessary for life. It can combine to form long chains which act as the backbone of large molecules. ...
... Compounds containing C, H, O and often N, P, & S. Organic compounds make up all living things and are necessary for life. It can combine to form long chains which act as the backbone of large molecules. ...
Bonding Nomenclature Notes
... ending second. 3) Add prefixes to both indicating the number of atoms of each element ...
... ending second. 3) Add prefixes to both indicating the number of atoms of each element ...
Biomolecules Review
... 14. To which of the four classes of amino acid does cysteine belong? 15. Draw the structure of a generic dipeptide using H3N+-CHR-CO2-. What type of bond links the amino acids? Draw an arrow to this bond. 16. What are the four levels of protein structure? What level is given for this tripeptide? ala ...
... 14. To which of the four classes of amino acid does cysteine belong? 15. Draw the structure of a generic dipeptide using H3N+-CHR-CO2-. What type of bond links the amino acids? Draw an arrow to this bond. 16. What are the four levels of protein structure? What level is given for this tripeptide? ala ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.