Proteins - Boardworks
... When more amino acids are added to a dipeptide, a polypeptide chain is formed. A protein consists of one or more polypeptide chains folded into a highly specific 3D shape. There are up to four levels of structure in a protein: primary, secondary, tertiary and quaternary. Each of these play an import ...
... When more amino acids are added to a dipeptide, a polypeptide chain is formed. A protein consists of one or more polypeptide chains folded into a highly specific 3D shape. There are up to four levels of structure in a protein: primary, secondary, tertiary and quaternary. Each of these play an import ...
Nuclease Digestion
... • Fatty acids: building blocks • Composed of a hydrophilic “head” and hydrophobic ...
... • Fatty acids: building blocks • Composed of a hydrophilic “head” and hydrophobic ...
Proteins
... 6. Label the type of bond used to make proteins. 7. Draw arrows to identify these bonds in your model 8. Label the N-terminus and C-terminus 9. Put SQUARES around the R groups 10. Use your amino acid chart to identify & label the type of R group (non-polar, polar, charge basic, charged acidic, etc) ...
... 6. Label the type of bond used to make proteins. 7. Draw arrows to identify these bonds in your model 8. Label the N-terminus and C-terminus 9. Put SQUARES around the R groups 10. Use your amino acid chart to identify & label the type of R group (non-polar, polar, charge basic, charged acidic, etc) ...
Key Points Folding
... Prions and Protein Folding • Protein structure (primary, secondary, tertiary) • Proteins have many possible conformations (ways to fold up into a 3D structure) • Proteins can spontaneously fold into the correct (biologically functional) 3D structure demonstrated by Christian Anfinsen in the 1950’s • ...
... Prions and Protein Folding • Protein structure (primary, secondary, tertiary) • Proteins have many possible conformations (ways to fold up into a 3D structure) • Proteins can spontaneously fold into the correct (biologically functional) 3D structure demonstrated by Christian Anfinsen in the 1950’s • ...
Proteins - Kaikoura High School
... • Primary structure = order of amino acids • Secondary structure = how tight the coil or chain is • Tertiary structure = how the chains loop back on themselves again and again. • Quaternary structure = how separate ...
... • Primary structure = order of amino acids • Secondary structure = how tight the coil or chain is • Tertiary structure = how the chains loop back on themselves again and again. • Quaternary structure = how separate ...
Peptide Bonds
... Emil Fischer formulated the lock-and-key mechanism for enzymes All reactions which occur in living cells are mediated by enzymes and are catalysed by 106-108 Some enzymes may require the presence of a Cofactor. This may be a metal atom, which is essential for its redox activity. Others may require t ...
... Emil Fischer formulated the lock-and-key mechanism for enzymes All reactions which occur in living cells are mediated by enzymes and are catalysed by 106-108 Some enzymes may require the presence of a Cofactor. This may be a metal atom, which is essential for its redox activity. Others may require t ...
The_Structure_of_Protein_Activity
... chains (example: fibrous proteins such as α-keratin in hair have helical chains of amino acids held together by hydrogen bonds); Tertiary structure describes the overall 3D folding and shape of a protein. This is held together with hydrogen bonds and other wesk interactions between the R groups. Pro ...
... chains (example: fibrous proteins such as α-keratin in hair have helical chains of amino acids held together by hydrogen bonds); Tertiary structure describes the overall 3D folding and shape of a protein. This is held together with hydrogen bonds and other wesk interactions between the R groups. Pro ...
Proteins - CasimiroSBI4U
... Peptide bond = covalent bond formed by condensation reaction that links carboxyl group of one amino acid to amino group of another. ...
... Peptide bond = covalent bond formed by condensation reaction that links carboxyl group of one amino acid to amino group of another. ...
Supplementary data 1,2,3,4,6,7,8,9 include N, Total (ProtScore)
... Supplementary data 1,2,3,4,6,7,8,9 include N, Total (ProtScore), %Cov(95), Accession, Name, Conf, Sequence,Modifications for the identified proteins. The definitions of the table fields are described as follows: N is the rank of the specified protein relative to all other proteins in the list of det ...
... Supplementary data 1,2,3,4,6,7,8,9 include N, Total (ProtScore), %Cov(95), Accession, Name, Conf, Sequence,Modifications for the identified proteins. The definitions of the table fields are described as follows: N is the rank of the specified protein relative to all other proteins in the list of det ...
Unit1-KA4-Revision
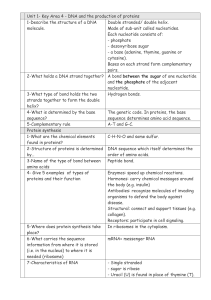
... 2-What holds a DNA strand together? A bond between the sugar of one nucleotide and the phosphate of the adjacent nucleotide. 3-What type of bond holds the two Hydrogen bonds. strands together to form the double helix? 4-What is determined by the base The genetic code. In proteins, the base sequence? ...
... 2-What holds a DNA strand together? A bond between the sugar of one nucleotide and the phosphate of the adjacent nucleotide. 3-What type of bond holds the two Hydrogen bonds. strands together to form the double helix? 4-What is determined by the base The genetic code. In proteins, the base sequence? ...
Proteins and Enzymes - Downtown Magnets High School
... • Compounds that contain N, C, H, and O. • Made of polymers of amino acids. ...
... • Compounds that contain N, C, H, and O. • Made of polymers of amino acids. ...
Knuffke Prezi- Macromolecules
... Organic Compounds Substances which contain Carbon. All living things contain Carbon, it is known as the backbone of life. ...
... Organic Compounds Substances which contain Carbon. All living things contain Carbon, it is known as the backbone of life. ...
Proteins - Wesleyan College Faculty
... Proteins • Primary Structure – Sequence of Amino Acids – Met-Gly-Ser-His-Ala- Leu-Trp-Ile-Thr ...
... Proteins • Primary Structure – Sequence of Amino Acids – Met-Gly-Ser-His-Ala- Leu-Trp-Ile-Thr ...
Amide Bond Formation
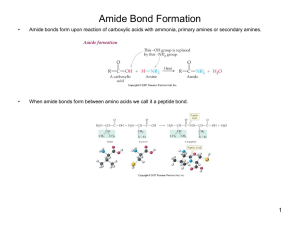
... Amide bonds form upon reaction of carboxylic acids with ammonia, primary amines or secondary amines. ...
... Amide bonds form upon reaction of carboxylic acids with ammonia, primary amines or secondary amines. ...
Ch 4 Reading Guide
... 2. Give three reasons it is important to know the primary sequence of a protein. 3. Give a chemical explanation of why a peptide bond is planar. 4. Why are almost all peptide bonds in proteins trans rather than cis. 5. Regular, folded segments of amino acids near one another in linear sequence is ca ...
... 2. Give three reasons it is important to know the primary sequence of a protein. 3. Give a chemical explanation of why a peptide bond is planar. 4. Why are almost all peptide bonds in proteins trans rather than cis. 5. Regular, folded segments of amino acids near one another in linear sequence is ca ...
Cyclol

The cyclol hypothesis is the first structural model of a folded, globular protein. It was developed by Dorothy Wrinch in the late 1930s, and was based on three assumptions. Firstly, the hypothesis assumes that two peptide groups can be crosslinked by a cyclol reaction (Figure 1); these crosslinks are covalent analogs of non-covalent hydrogen bonds between peptide groups. These reactions have been observed in the ergopeptides and other compounds. Secondly, it assumes that, under some conditions, amino acids will naturally make the maximum possible number of cyclol crosslinks, resulting in cyclol molecules (Figure 2) and cyclol fabrics (Figure 3). These cyclol molecules and fabrics have never been observed. Finally, the hypothesis assumes that globular proteins have a tertiary structure corresponding to Platonic solids and semiregular polyhedra formed of cyclol fabrics with no free edges. Such ""closed cyclol"" molecules have not been observed either.Although later data demonstrated that this original model for the structure of globular proteins needed to be amended, several elements of the cyclol model were verified, such as the cyclol reaction itself and the hypothesis that hydrophobic interactions are chiefly responsible for protein folding. The cyclol hypothesis stimulated many scientists to research questions in protein structure and chemistry, and was a precursor of the more accurate models hypothesized for the DNA double helix and protein secondary structure. The proposal and testing of the cyclol model also provides an excellent illustration of empirical falsifiability acting as part of the scientific method.