Bohr Model and Quantum Model
... Which transition occurs when a hydrogen atom emits light with a wavelength of 434 nm? A) The electron jumps from n=2 to n=4 B) The electron jumps from n=2 to n=5 C) The electron falls from n=4 to n=2 D) The electron falls from n=5 to n=2 ...
... Which transition occurs when a hydrogen atom emits light with a wavelength of 434 nm? A) The electron jumps from n=2 to n=4 B) The electron jumps from n=2 to n=5 C) The electron falls from n=4 to n=2 D) The electron falls from n=5 to n=2 ...
Ch 11 WS Orbitals and Electron Arrangement
... 9. Principal energy levels are assigned values in order of ______________________ energy: n = 1, 2, 3, 4, and so forth. 10. In the quantum mechanical model the regions where electrons are likely to be found are called ______________________ and are denoted by______________________ . ...
... 9. Principal energy levels are assigned values in order of ______________________ energy: n = 1, 2, 3, 4, and so forth. 10. In the quantum mechanical model the regions where electrons are likely to be found are called ______________________ and are denoted by______________________ . ...
Total view of the AFM
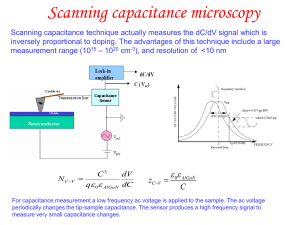
... voltage range of 20 – 30 keV, but for insulating samples 1 kV or less can be used. For insulating samples a thin metal coating can also be used. • The standard electron detector is an EverhartThornley design that is capable of amplifying electron currents by almost a million times. ...
... voltage range of 20 – 30 keV, but for insulating samples 1 kV or less can be used. For insulating samples a thin metal coating can also be used. • The standard electron detector is an EverhartThornley design that is capable of amplifying electron currents by almost a million times. ...
Tuesday Aug 19
... 1. According to the modern theory of the atom, where may an atom’s electrons be found? 2. How do electrons become “excited”? 3. What form of energy emission accompanies the return of excited electrons to the ground state? 4. Assume that an atom has a total of four possible energy levels and that an ...
... 1. According to the modern theory of the atom, where may an atom’s electrons be found? 2. How do electrons become “excited”? 3. What form of energy emission accompanies the return of excited electrons to the ground state? 4. Assume that an atom has a total of four possible energy levels and that an ...
The study of biology can help you better understand
... 13. What is the probability of finding an electron within the region indicated by the drawn electron cloud? ...
... 13. What is the probability of finding an electron within the region indicated by the drawn electron cloud? ...
Sugárkémiai áttekintés Schiller Róbert
... β irradiation of oxidized iron surfaces, after that electrode Impedance in aquoeus SO32- solution (hole capture). Equivalent circuit: ...
... β irradiation of oxidized iron surfaces, after that electrode Impedance in aquoeus SO32- solution (hole capture). Equivalent circuit: ...
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 4. Using the Pauli Exclusion Principle, explain why an s-orbital can hold a maximum of 2 electrons? There is only one s-orbital in each shell. Adding a third electron would mean that two of the three would have the same set of four quantum numbers. 5. Balance the following: C6H14 (l) + O2 (g) ...
... 4. Using the Pauli Exclusion Principle, explain why an s-orbital can hold a maximum of 2 electrons? There is only one s-orbital in each shell. Adding a third electron would mean that two of the three would have the same set of four quantum numbers. 5. Balance the following: C6H14 (l) + O2 (g) ...
Exam 2-1
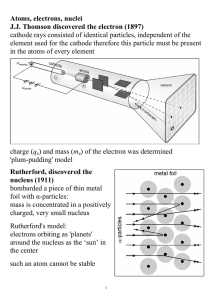
... Einstein’s explanation of the photoelectric effect. Rutherford’s experiment with a beam of α particles passing through gold foil. Boer’s model of the atom. Faraday’s experiment of the electroplating of metals. Binnig and Rohrer’s demonstration of the scanning tunneling microscope. ...
... Einstein’s explanation of the photoelectric effect. Rutherford’s experiment with a beam of α particles passing through gold foil. Boer’s model of the atom. Faraday’s experiment of the electroplating of metals. Binnig and Rohrer’s demonstration of the scanning tunneling microscope. ...
Chapter 5 Electrons In Atoms 5.1 Models of the Atom The
... An _______________________ orbital is often thought of as a region of space in which there is a high probability of finding an electron. The energy levels of _____________________________ in the quantum mechanical model are labeled by principal quantum numbers (n). These are assigned the values n=1, ...
... An _______________________ orbital is often thought of as a region of space in which there is a high probability of finding an electron. The energy levels of _____________________________ in the quantum mechanical model are labeled by principal quantum numbers (n). These are assigned the values n=1, ...
1to7
... #6) The energies of electrons are said to be quantized. Explain what that means. Each electron contains in its orbital a very specific amount or “quantum” of energy. If an electron gains or loses energy, it must change its orbital. ...
... #6) The energies of electrons are said to be quantized. Explain what that means. Each electron contains in its orbital a very specific amount or “quantum” of energy. If an electron gains or loses energy, it must change its orbital. ...
1-7-
... #6) The energies of electrons are said to be quantized. Explain what that means. Each electron contains in its orbital a very specific amount or “quantum” of energy. If an electron gains or loses energy, it must change its orbital. ...
... #6) The energies of electrons are said to be quantized. Explain what that means. Each electron contains in its orbital a very specific amount or “quantum” of energy. If an electron gains or loses energy, it must change its orbital. ...
Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory
... 2 valence electrons max: H, He 8 valence electrons max: 2nd row elements <8 valence electrons exceptions: Be=4, B=6 (formal charge) >8 valence electrons: 3rd row elements and below Consider formal charges in evaluating structures When to draw resonance structures, and how many (reso ...
... 2 valence electrons max: H, He 8 valence electrons max: 2nd row elements <8 valence electrons exceptions: Be=4, B=6 (formal charge) >8 valence electrons: 3rd row elements and below Consider formal charges in evaluating structures When to draw resonance structures, and how many (reso ...
2·QUIZLET VOCABULARY: Quantum Numbers Study online at
... 3. electron configuration: the arrangement of electrons around nucleus in an atom 4. Hunds rule: orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron, and all electrons in singly occupied orbitals must have the same spin 5. Magnetic (orbital) ...
... 3. electron configuration: the arrangement of electrons around nucleus in an atom 4. Hunds rule: orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron, and all electrons in singly occupied orbitals must have the same spin 5. Magnetic (orbital) ...
The Address of the Electrons
... electron has a set of four numbers, called quantum numbers Each electron has a different set of numbers ¡ No ...
... electron has a set of four numbers, called quantum numbers Each electron has a different set of numbers ¡ No ...
Electron-beam lithography

Electron-beam lithography (often abbreviated as e-beam lithography) is the practice of scanning a focused beam of electrons to draw custom shapes on a surface covered with an electron-sensitive film called a resist (""exposing""). The electron beam changes the solubility of the resist, enabling selective removal of either the exposed or non-exposed regions of the resist by immersing it in a solvent (""developing""). The purpose, as with photolithography, is to create very small structures in the resist that can subsequently be transferred to the substrate material, often by etching.The primary advantage of electron-beam lithography is that it can draw custom patterns (direct-write) with sub-10 nm resolution. This form of maskless lithography has high resolution and low throughput, limiting its usage to photomask fabrication, low-volume production of semiconductor devices, and research & development.