Chapter 5 Practice Section 5-1 Discuss the placement (if any) of
... What is the wavelength of radiation with a frequency of 2.3 x 1014 Hz? What is the frequency of radiation with a wavelength of 1.8 x 10-9 m? Rank in order of increasing energy: Purple light, x-rays, Microwaves Rank the above in order of increasing frequency. Rank the above in order of increasing wav ...
... What is the wavelength of radiation with a frequency of 2.3 x 1014 Hz? What is the frequency of radiation with a wavelength of 1.8 x 10-9 m? Rank in order of increasing energy: Purple light, x-rays, Microwaves Rank the above in order of increasing frequency. Rank the above in order of increasing wav ...
Chapter 5 Electrons in Atoms
... The propeller blade has the same probability of being anywhere in the blurry region, but you cannot tell its location at any instant. The electron cloud of an atom can be compared to a spinning airplane propeller. ...
... The propeller blade has the same probability of being anywhere in the blurry region, but you cannot tell its location at any instant. The electron cloud of an atom can be compared to a spinning airplane propeller. ...
ppt
... If n = 1, l = 0 then we call it a 1s orbital If n = 3, l = 2 then we call it a 3d orbital ...
... If n = 1, l = 0 then we call it a 1s orbital If n = 3, l = 2 then we call it a 3d orbital ...
Quiz 9
... The Pauli Exclusion Principle states that more than one fermion – particles with 21 -integer spin – cannot exist in the exact same quantum mechanical state; i.e. at least one quantum number must differ. As a result, for any energy level, n, there are l = 0, 1, . . . , (n − 1) orbital angular momentu ...
... The Pauli Exclusion Principle states that more than one fermion – particles with 21 -integer spin – cannot exist in the exact same quantum mechanical state; i.e. at least one quantum number must differ. As a result, for any energy level, n, there are l = 0, 1, . . . , (n − 1) orbital angular momentu ...
Chemistry 102 Summary June 25th - Bohr model only works for one
... Orbitals define the allowed energy states where electrons can reside. There are four basic shapes: s, p, d and f Shapes represent where an electron will reside 90 % of the time in that allowed energy state. From Heisenberg – the exact location cannot be determined but instead the probability (Ψ2). E ...
... Orbitals define the allowed energy states where electrons can reside. There are four basic shapes: s, p, d and f Shapes represent where an electron will reside 90 % of the time in that allowed energy state. From Heisenberg – the exact location cannot be determined but instead the probability (Ψ2). E ...
Multi-electron Atoms
... Several distinct electron states (orbits) exist, all with the same n, forming a `shell' of states. In general, these states have different energies. The number of different orbital states in a shell of a given n is n2. However, also note that electrons obey the Pauli Exclusion Principle which states ...
... Several distinct electron states (orbits) exist, all with the same n, forming a `shell' of states. In general, these states have different energies. The number of different orbital states in a shell of a given n is n2. However, also note that electrons obey the Pauli Exclusion Principle which states ...
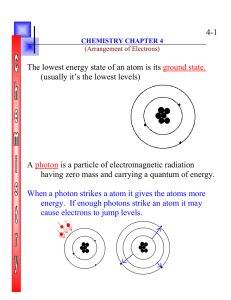
4-1 The lowest energy state of an atom is its ground state. (usually
... A student will be placed one floor higher if and only if there is an available room, which is two grades better. The following chart will take care of all rules for your. ...
... A student will be placed one floor higher if and only if there is an available room, which is two grades better. The following chart will take care of all rules for your. ...
Bohr model
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
Problem set #1 - U.C.C. Physics Department
... and let kx = 6π/L; ky = 4π/L. Draw out (i) for t = 0 and (ii) for t = π/ω, the lines along which ψ(x, y, t) = 1. Calculate the repeat distance of the wave along the x-direction, the y-direction, and its direction of motion. 2) Bohr’s atomic model Recall that Bohr derived Rydberg’s constant by assumi ...
... and let kx = 6π/L; ky = 4π/L. Draw out (i) for t = 0 and (ii) for t = π/ω, the lines along which ψ(x, y, t) = 1. Calculate the repeat distance of the wave along the x-direction, the y-direction, and its direction of motion. 2) Bohr’s atomic model Recall that Bohr derived Rydberg’s constant by assumi ...
Section 4-2 The Quantum Model of the Atom Problems with the Bohr
... iii. L = 2 is called a “D orbital” with a double Figure-8 shape iv. L = 3 is called an “F orbital” with a quadruple Figure-8 shape c. The possible shapes of “L” are defined by the size of “n” i. “L” can be any integer between 0 and (n-1). ii. If n = 2, then L can be 0 or 1 iii. If n = 4, then L can ...
... iii. L = 2 is called a “D orbital” with a double Figure-8 shape iv. L = 3 is called an “F orbital” with a quadruple Figure-8 shape c. The possible shapes of “L” are defined by the size of “n” i. “L” can be any integer between 0 and (n-1). ii. If n = 2, then L can be 0 or 1 iii. If n = 4, then L can ...
Electron-beam lithography

Electron-beam lithography (often abbreviated as e-beam lithography) is the practice of scanning a focused beam of electrons to draw custom shapes on a surface covered with an electron-sensitive film called a resist (""exposing""). The electron beam changes the solubility of the resist, enabling selective removal of either the exposed or non-exposed regions of the resist by immersing it in a solvent (""developing""). The purpose, as with photolithography, is to create very small structures in the resist that can subsequently be transferred to the substrate material, often by etching.The primary advantage of electron-beam lithography is that it can draw custom patterns (direct-write) with sub-10 nm resolution. This form of maskless lithography has high resolution and low throughput, limiting its usage to photomask fabrication, low-volume production of semiconductor devices, and research & development.