figueroa, ingrid
... Phspholipids: substitutes the third fatty acid of fats with a negatively charged phosphate group, which may be joined by a smaller molecule. Steroids: lipids characterized by a carbon skeleton consisting of four interconnected rings. Saturated fatty acids have the maximum number of hydrogen atom ...
... Phspholipids: substitutes the third fatty acid of fats with a negatively charged phosphate group, which may be joined by a smaller molecule. Steroids: lipids characterized by a carbon skeleton consisting of four interconnected rings. Saturated fatty acids have the maximum number of hydrogen atom ...
Cell Energetics - Practice Test - Biology
... a. The runner's cells need oxygen to make lactic acid. b. The runner's cells continue the fermentation process. c. The runner's cells are making up for an oxygen deficit d. The runner's cells cannot perform glycolysis. ...
... a. The runner's cells need oxygen to make lactic acid. b. The runner's cells continue the fermentation process. c. The runner's cells are making up for an oxygen deficit d. The runner's cells cannot perform glycolysis. ...
Mid-Term Exam 1a - Buffalo State College Faculty and Staff Web
... A. A water molecule is split to restore the hydroxyl and hydrogen on the monomers B. The energy stored in the molecule is released during splitting and temporarily stored in water. C. A water molecule needs to be divided into an hydroxyl and a hydrogen to release energy to split the polymers. D. A h ...
... A. A water molecule is split to restore the hydroxyl and hydrogen on the monomers B. The energy stored in the molecule is released during splitting and temporarily stored in water. C. A water molecule needs to be divided into an hydroxyl and a hydrogen to release energy to split the polymers. D. A h ...
Proteins - Northern Highlands
... Secondary Structure in Proteins Within a single protein, different regions of the polypeptide chain assume different conformations determined by the primary sequence of amino acids. α-Helix: formation is stabilized by H-bonds between amino nitrogens and carbonyl carbons of the peptide bonds every 4 ...
... Secondary Structure in Proteins Within a single protein, different regions of the polypeptide chain assume different conformations determined by the primary sequence of amino acids. α-Helix: formation is stabilized by H-bonds between amino nitrogens and carbonyl carbons of the peptide bonds every 4 ...
bioCHEMISTRY 480 Molecular Biochemistry-‐
... There will be handed out as problem sets. They are intended to be practice for the next test. They will not be all graded but keys will be posted on the website. In addition, there will be ...
... There will be handed out as problem sets. They are intended to be practice for the next test. They will not be all graded but keys will be posted on the website. In addition, there will be ...
Organic Compounds
... chain, R, varies from one amino acid to another. KEY: H = hydrogen, N = nitrogen, C = carbon, O = oxygen, R = variable side chain. ...
... chain, R, varies from one amino acid to another. KEY: H = hydrogen, N = nitrogen, C = carbon, O = oxygen, R = variable side chain. ...
Sample
... 8. A food that contains an imbalanced protein means a. It is not nutritionally good to eat b.It will not yield and adequate protein-to-calorie ratio c. It does not contain enough of one or more essential amino acids d.It is non-digestible e. Its amino acid chains are branched asymmetrically ...
... 8. A food that contains an imbalanced protein means a. It is not nutritionally good to eat b.It will not yield and adequate protein-to-calorie ratio c. It does not contain enough of one or more essential amino acids d.It is non-digestible e. Its amino acid chains are branched asymmetrically ...
BHS 150.1 – Biochemistry II Date: 2/8/2013, 2sndhalf Notetaker: Kim
... 12. Ascorbate in the aqueous is needed by the cornea for which two of the following functions: antioxidant properties collagen synthesis 13. A missense mutation occurs when the: amino acid sequence changes 14. During fasting, what are some possible sources of carbons for gluconeogenesis: amino acids ...
... 12. Ascorbate in the aqueous is needed by the cornea for which two of the following functions: antioxidant properties collagen synthesis 13. A missense mutation occurs when the: amino acid sequence changes 14. During fasting, what are some possible sources of carbons for gluconeogenesis: amino acids ...
presentation source
... 7. compare the lactic acid pathway and aerobic respiration in terms of initial substrates, final products, cellular locations, and the total number of ATP molecules produced per glucose respired. 8. explain how glucose and glycogen can be interconverted, and how the liver can secrete free glucose de ...
... 7. compare the lactic acid pathway and aerobic respiration in terms of initial substrates, final products, cellular locations, and the total number of ATP molecules produced per glucose respired. 8. explain how glucose and glycogen can be interconverted, and how the liver can secrete free glucose de ...
Biochemistry-lab-identifying
... through donating or sharing energy from electrons. When two or more elements join together we call this a molecule. All living things are made a handful of the same molecules. The molecules essential for life to function are called biomolecules and are considered “organic”. Carbon, Hydrogen, Oxygen, ...
... through donating or sharing energy from electrons. When two or more elements join together we call this a molecule. All living things are made a handful of the same molecules. The molecules essential for life to function are called biomolecules and are considered “organic”. Carbon, Hydrogen, Oxygen, ...
Regents Biology Homework Packet Unit 4: Biochemistry
... 8. Why are a hydrogen and hydroxyl(OH) removed during a dehydration synthesis reaction? ...
... 8. Why are a hydrogen and hydroxyl(OH) removed during a dehydration synthesis reaction? ...
Chapter 3 Review - Nutley Public Schools
... Base your answers to questions 34 to 36 on the information beloH' and on your knowledge of biology. Use one or more complete sentences to answer each question. People try to lose weight by dieting. Researchers now know that dieting may reduce the amount of fat in the body, but it also changes how th ...
... Base your answers to questions 34 to 36 on the information beloH' and on your knowledge of biology. Use one or more complete sentences to answer each question. People try to lose weight by dieting. Researchers now know that dieting may reduce the amount of fat in the body, but it also changes how th ...
biochemistry
... body by serving as a catalyst. In otherwords, an enzyme speeds up a reaction without being used up in the reaction. A chemical reaction is a process in which one set of chemicals, known as reactants are changed into another set of chemicals known as the product. An enzyme works by lowering the activ ...
... body by serving as a catalyst. In otherwords, an enzyme speeds up a reaction without being used up in the reaction. A chemical reaction is a process in which one set of chemicals, known as reactants are changed into another set of chemicals known as the product. An enzyme works by lowering the activ ...
PowerPoint Presentation - No Slide Title
... Ribosomes are large protein / RNA complexes that are the site of translation. The structure of ribosomes reflects ribosomal function. Each ribosome consists of large and small subunits, with binding sites for mRNA and three tRNA molecules. During translation, the growing polypeptide is atached to t ...
... Ribosomes are large protein / RNA complexes that are the site of translation. The structure of ribosomes reflects ribosomal function. Each ribosome consists of large and small subunits, with binding sites for mRNA and three tRNA molecules. During translation, the growing polypeptide is atached to t ...
5.Amino acids
... amino acids), most applications are for L-form sometime DL or D maybe required. 3. MICROBIOLOGICAL SYNTHESIS a. Direct fermentation: MO use carbon sources and produce aa. Carbon like glu, fructose, alkanes, ethanol, glycerol, molasses, starch, methanol etc. b. Conversion of metabolic intermediates t ...
... amino acids), most applications are for L-form sometime DL or D maybe required. 3. MICROBIOLOGICAL SYNTHESIS a. Direct fermentation: MO use carbon sources and produce aa. Carbon like glu, fructose, alkanes, ethanol, glycerol, molasses, starch, methanol etc. b. Conversion of metabolic intermediates t ...
Life, 6th Edition
... Macromolecules are formed by covalent bonds between monomers and include polysaccharides, proteins, and nucleic acids. Lipids are crucial biomolecules, but are not considered ‘macromolecules’ ...
... Macromolecules are formed by covalent bonds between monomers and include polysaccharides, proteins, and nucleic acids. Lipids are crucial biomolecules, but are not considered ‘macromolecules’ ...
Analysis on Organic Compounds Submitted by WWW
... Another important polysaccharide is glycogen, which is related to starch. Many bacteria have glycogen in thier cytoplasm. Still another is cellulose. Cellulose is also composed of glucose units, but the units cannot be released from one another except by a few species of microorganisms, especially ...
... Another important polysaccharide is glycogen, which is related to starch. Many bacteria have glycogen in thier cytoplasm. Still another is cellulose. Cellulose is also composed of glucose units, but the units cannot be released from one another except by a few species of microorganisms, especially ...
Unit 3
... sources, for example meat, poultry, full-fat dairy products and tropical oils, such as palm and coconut oils. Unsaturated fatty acids contain one or more double bonds between carbon atoms within the fatty acid chain. Unsaturated fats originate from plant-based foods for example olive oil, olives, av ...
... sources, for example meat, poultry, full-fat dairy products and tropical oils, such as palm and coconut oils. Unsaturated fatty acids contain one or more double bonds between carbon atoms within the fatty acid chain. Unsaturated fats originate from plant-based foods for example olive oil, olives, av ...
Biochemistry - Plano Science Tutor
... • Sugars supply immediate energy, whereas starches must first be broken down to sugars ...
... • Sugars supply immediate energy, whereas starches must first be broken down to sugars ...
Macromolecules of Life
... Sucrose (table sugar) The most abundant disaccharide throughout the plant kingdom Lactose is the milk sugar Chemical properties depend on the nature of the linked monosaccharides, the carbon atoms involved in bonding, and the form of the linkage. ...
... Sucrose (table sugar) The most abundant disaccharide throughout the plant kingdom Lactose is the milk sugar Chemical properties depend on the nature of the linked monosaccharides, the carbon atoms involved in bonding, and the form of the linkage. ...
Worksheet – Proteins Proteins are polymers of amino acids, joined
... interact via LDF. These are also called hydrophobic interactions, since these groups can not interact with water. For this reason, they are usually buried in the interior of proteins, away from water. They can be disrupted by the addition of detergent. The polar side chains interact via H bonding si ...
... interact via LDF. These are also called hydrophobic interactions, since these groups can not interact with water. For this reason, they are usually buried in the interior of proteins, away from water. They can be disrupted by the addition of detergent. The polar side chains interact via H bonding si ...
CHAPTER 4 - HCC Learning Web
... Each cell has thousands of different macromolecules. The collection varies one type of cells to another even with in the same organism The inherent difference between human siblings reflect small variations in polymers, particularly: ...
... Each cell has thousands of different macromolecules. The collection varies one type of cells to another even with in the same organism The inherent difference between human siblings reflect small variations in polymers, particularly: ...
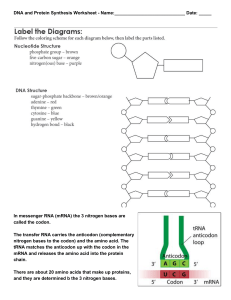
Protein Synthesis
... Step 2: Translation Location: in the cytoplasm, on the ribosome Purpose: to convert the instructions of RNA (order of bases) into amino acids, this will make up the protein. Events of translation: 1.) The first three bases of mRNA (codon) join the ribosome. AUG – is the start codon 2.) tRNA brings t ...
... Step 2: Translation Location: in the cytoplasm, on the ribosome Purpose: to convert the instructions of RNA (order of bases) into amino acids, this will make up the protein. Events of translation: 1.) The first three bases of mRNA (codon) join the ribosome. AUG – is the start codon 2.) tRNA brings t ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.