chapter 5 the structure and function of macromolecules
... be built from a small set of monomers • Each cell has thousands of different macromolecules. – These molecules vary among cells of the same individual, even more among unrelated individuals of a species, and are even greater between species. • This diversity comes from various combinations of the 40 ...
... be built from a small set of monomers • Each cell has thousands of different macromolecules. – These molecules vary among cells of the same individual, even more among unrelated individuals of a species, and are even greater between species. • This diversity comes from various combinations of the 40 ...
DNA to Protein Name____________ Period______ DNA Location
... 1. DNA is contained in the nucleus of eukaryotes (plants/animals) 2. DNA mRNA The DNA message gets copied into mRNA. This is called transcription. 3. The mRNA leaves nucleus and sticks to ribosomes. (The ribosomes can be floating in cytoplasm (free) or stuck to rough endoplasmic reticulum.) 4. Ribo ...
... 1. DNA is contained in the nucleus of eukaryotes (plants/animals) 2. DNA mRNA The DNA message gets copied into mRNA. This is called transcription. 3. The mRNA leaves nucleus and sticks to ribosomes. (The ribosomes can be floating in cytoplasm (free) or stuck to rough endoplasmic reticulum.) 4. Ribo ...
Organic Chemistry
... help of enzymes to make and break bonds • Enzymes are specialized proteins that speed up the chemical reactions in cells • Enzymes are extremely important – without them, many reactions cannot take place. If you lack lactase, you cannot hydrolyze the bond in lactose (lactose intolerant) ...
... help of enzymes to make and break bonds • Enzymes are specialized proteins that speed up the chemical reactions in cells • Enzymes are extremely important – without them, many reactions cannot take place. If you lack lactase, you cannot hydrolyze the bond in lactose (lactose intolerant) ...
LE - 2 - Organic Molecules
... Elements in Life • Water! Our bodies are 65% water, but water is inorganic • Organic molecules can be broken down into 4 main categories. These substances are needed for life to grow and function properly. ...
... Elements in Life • Water! Our bodies are 65% water, but water is inorganic • Organic molecules can be broken down into 4 main categories. These substances are needed for life to grow and function properly. ...
CHAPTER 2 VOCABULARY (Highlighted)
... Hydrocarbon chain often bonded to glycerol in a lipid Nonpolar molecule composed of carbon, hydrogen, and oxygen; includes fats and oils. Molecular subunit of a polymer Polymer of nucleotides; the genetic material of organisms. Large, carbon-based molecule formed by monomers. Polymer composed of ami ...
... Hydrocarbon chain often bonded to glycerol in a lipid Nonpolar molecule composed of carbon, hydrogen, and oxygen; includes fats and oils. Molecular subunit of a polymer Polymer of nucleotides; the genetic material of organisms. Large, carbon-based molecule formed by monomers. Polymer composed of ami ...
No Slide Title
... The C, C, N and O atoms are called backbone atoms. R denotes any one of the 20 possible side chains. ...
... The C, C, N and O atoms are called backbone atoms. R denotes any one of the 20 possible side chains. ...
DNA replication
... • In non-biological environ: two types are equally found • In living organisms: only left-handed amino acids are found! All Earth life forms were originated from an ancestral life using only left-handed amino acids! ...
... • In non-biological environ: two types are equally found • In living organisms: only left-handed amino acids are found! All Earth life forms were originated from an ancestral life using only left-handed amino acids! ...
omproteinsandnucleicacids
... Note: The “R” shows represents where the carboxyl group attaches to an amino acid (also where it would attach to carbohydrates, lipids, and nucleic acids) ...
... Note: The “R” shows represents where the carboxyl group attaches to an amino acid (also where it would attach to carbohydrates, lipids, and nucleic acids) ...
Blue Flashcards (CR) - mvhs
... intermediates that are oxidizing agents. They hold ___________ that are eventually discarded in the oxidative phosphorylation, to allow _____ to be made. ...
... intermediates that are oxidizing agents. They hold ___________ that are eventually discarded in the oxidative phosphorylation, to allow _____ to be made. ...
moluceular lab 1
... build cell’s nutrients and to perform biological functions. Structural organic order is as follows: Organism–System–Organs–Tissues–Cells–Organelles– Molecules-Atoms The body of Living organisms consists of : two types of Molecules: Inorganic molecules : (water, salts, acids and bases) they are basic ...
... build cell’s nutrients and to perform biological functions. Structural organic order is as follows: Organism–System–Organs–Tissues–Cells–Organelles– Molecules-Atoms The body of Living organisms consists of : two types of Molecules: Inorganic molecules : (water, salts, acids and bases) they are basic ...
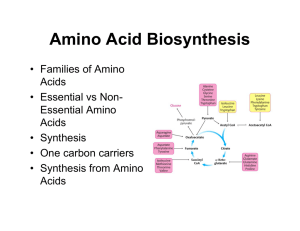
Amino Acid Biosynthesis
... Nitrogen • Atmospheric N2 is the ultimate source of biological nitrogen • Nitrogen fixation: a few bacteria possess nitrogenase which can reduce N2 to ammonia • Nitrogen is recycled in nature through the nitrogen cycle ...
... Nitrogen • Atmospheric N2 is the ultimate source of biological nitrogen • Nitrogen fixation: a few bacteria possess nitrogenase which can reduce N2 to ammonia • Nitrogen is recycled in nature through the nitrogen cycle ...
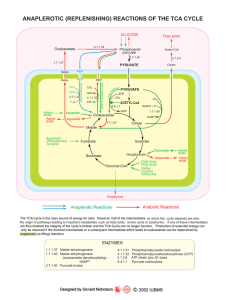
Amino Acid Catabolism
... Amino Acid Catabolism • Amino acids from degraded proteins or from diet can be used for the biosynthesis of new proteins • During starvation proteins are degraded to amino acids to support glucose formation • First step is often removal of the α-amino group • Carbon chains are altered for entry int ...
... Amino Acid Catabolism • Amino acids from degraded proteins or from diet can be used for the biosynthesis of new proteins • During starvation proteins are degraded to amino acids to support glucose formation • First step is often removal of the α-amino group • Carbon chains are altered for entry int ...
Cell Biology
... enzyme’s shape is altered, meaning the active site no longer matches the substrate shape. The enzyme is denatured. The temperature that the enzyme’s activity is the highest is known as the optimum temperature. ...
... enzyme’s shape is altered, meaning the active site no longer matches the substrate shape. The enzyme is denatured. The temperature that the enzyme’s activity is the highest is known as the optimum temperature. ...
Honors Biology Midterm Reviewаа BASIC CHEMISTRY
... Starch storage polysaccharide composed of glucose monomers and found in plants Glycogen storage polysaccharide composed of glucose, hydrolyzed by animals when glucose is needed Cellulose polymer of glucose that forms plant cell walls ...
... Starch storage polysaccharide composed of glucose monomers and found in plants Glycogen storage polysaccharide composed of glucose, hydrolyzed by animals when glucose is needed Cellulose polymer of glucose that forms plant cell walls ...
Biology 155 - Quiz 6 1. In theory, how many molecules of ATP can
... Biology 155 - Quiz 6 1. In theory, how many molecules of ATP can be produced from one molecule of acetylCoA if its carbons are completely metabolized in respiration? a. 7.5 b. 8 c. 9 d. 9.5 e. 15 f. 10 (none of the choices a to e were correct.) 2. In eukaryotic cells, the Krebs Cycle occurs in a. th ...
... Biology 155 - Quiz 6 1. In theory, how many molecules of ATP can be produced from one molecule of acetylCoA if its carbons are completely metabolized in respiration? a. 7.5 b. 8 c. 9 d. 9.5 e. 15 f. 10 (none of the choices a to e were correct.) 2. In eukaryotic cells, the Krebs Cycle occurs in a. th ...
Chapter 10 .1 The Function of Digestion MACROMOLECULES AND
... LIPIDS – a macromolecule/ an organic compound that does not dissolve in water, such as fat and oil - Basic structure of lipids is a molecule of glycerol (an alcohol) consisting of three carbon atoms, each attached to a fatty acid chain (an acid with a long tail of carbon and hydrogen atoms) - Lipids ...
... LIPIDS – a macromolecule/ an organic compound that does not dissolve in water, such as fat and oil - Basic structure of lipids is a molecule of glycerol (an alcohol) consisting of three carbon atoms, each attached to a fatty acid chain (an acid with a long tail of carbon and hydrogen atoms) - Lipids ...
Section 2-1: Nature of Matter
... 42. Carbon has _____ electrons in its outer energy level. As such, carbon is _____ electrons short of filling the outer energy level and being stable. Therefore, carbon readily forms four _____________ bonds with other elements. 43. monomers: 44. polymer: Note: • Polymers of living things are constr ...
... 42. Carbon has _____ electrons in its outer energy level. As such, carbon is _____ electrons short of filling the outer energy level and being stable. Therefore, carbon readily forms four _____________ bonds with other elements. 43. monomers: 44. polymer: Note: • Polymers of living things are constr ...
Chapter 2: The Chemical Level of Organization
... • C, H, O, N, and P • Large organic molecules, found in the nucleus, which store and process information at the molecular level • DNA – deoxyribonucleic acid • RNA – ribonucleic acid ...
... • C, H, O, N, and P • Large organic molecules, found in the nucleus, which store and process information at the molecular level • DNA – deoxyribonucleic acid • RNA – ribonucleic acid ...
A hidden genetic code: Researchers identify key
... proteins efficiently under stressful conditions, it also acts as a biological failsafe, allowing the nearcomplete shutdown in the production of other proteins as a way to preserve limited resources. Given the universal nature of the genetic code—the system works the same way for all organisms, from ...
... proteins efficiently under stressful conditions, it also acts as a biological failsafe, allowing the nearcomplete shutdown in the production of other proteins as a way to preserve limited resources. Given the universal nature of the genetic code—the system works the same way for all organisms, from ...
Table 1 The Essential Amino Acids and Their Plant Sources
... molecules, and any excess proteins you eat are broken down into their amino acids and transported to cells via the bloodstream. Protein breakdown produces the waste urea, which is filtered from the blood by the kidneys. Urea is what gives your urine its yellow color. Why protein is needed Dietary pr ...
... molecules, and any excess proteins you eat are broken down into their amino acids and transported to cells via the bloodstream. Protein breakdown produces the waste urea, which is filtered from the blood by the kidneys. Urea is what gives your urine its yellow color. Why protein is needed Dietary pr ...
Chemistry 464 Biochemistry First Hour Exam
... nucleus and does not form into compact chromosomes. Because many Eukariotic cells are found in multicelluar organisms they have many complicated structures and organizations to help them interact with their neighboring cells. Prokariots and usually unicellular, so they have simpler cell surface inte ...
... nucleus and does not form into compact chromosomes. Because many Eukariotic cells are found in multicelluar organisms they have many complicated structures and organizations to help them interact with their neighboring cells. Prokariots and usually unicellular, so they have simpler cell surface inte ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.