Chemistry 464 Biochemistry First Hour Exam
... nucleus and does not form into compact chromosomes. Because many Eukariotic cells are found in multicelluar organisms they have many complicated structures and organizations to help them interact with their neighboring cells. Prokariots and usually unicellular, so they have simpler cell surface inte ...
... nucleus and does not form into compact chromosomes. Because many Eukariotic cells are found in multicelluar organisms they have many complicated structures and organizations to help them interact with their neighboring cells. Prokariots and usually unicellular, so they have simpler cell surface inte ...
Lehninger Principles of Biochemistry
... carboxypeptidase A – removes carboxy-terminal amino acids aminopeptidase – removes amino terminal amino acids ...
... carboxypeptidase A – removes carboxy-terminal amino acids aminopeptidase – removes amino terminal amino acids ...
Cell Molecules * materials for biotechnology
... Primary components of cell walls Plants - cellulose Bacteria cell walls Insects – exoskeleton (chitin) ...
... Primary components of cell walls Plants - cellulose Bacteria cell walls Insects – exoskeleton (chitin) ...
Words to Pronounce
... electrons in atoms. To understand the body, understand how chemicals in the body either attract or repel each other. For example, this same force causes oxygen to cling to red blood cells. Also, muscle contraction is dependent upon the electrical attraction and repulsion forces of calcium (Ca2+), so ...
... electrons in atoms. To understand the body, understand how chemicals in the body either attract or repel each other. For example, this same force causes oxygen to cling to red blood cells. Also, muscle contraction is dependent upon the electrical attraction and repulsion forces of calcium (Ca2+), so ...
Department of Biochemistry and Molecular Biology
... Brief outlines for the University of B.C. Biochemistry 301 and 303 courses are given below. Please outline all undergraduate biochemistry courses that you have successfully completed that are equivalent to or more advanced than these courses. Bioc 301 Biochemistry Laboratory – Techniques by which th ...
... Brief outlines for the University of B.C. Biochemistry 301 and 303 courses are given below. Please outline all undergraduate biochemistry courses that you have successfully completed that are equivalent to or more advanced than these courses. Bioc 301 Biochemistry Laboratory – Techniques by which th ...
AP Biology Summer Vocabulary Assignment
... unravels and loses its native conformation, thereby becoming biologically inactive. For DNA, the separation of the two strands of the double helix. Denaturation occurs under extreme conditions of pH, salt concentration, and temperature. ...
... unravels and loses its native conformation, thereby becoming biologically inactive. For DNA, the separation of the two strands of the double helix. Denaturation occurs under extreme conditions of pH, salt concentration, and temperature. ...
Essential Biochemistry. 3rd Edition Brochure
... Brochure More information from http://www.researchandmarkets.com/reports/2239494/ ...
... Brochure More information from http://www.researchandmarkets.com/reports/2239494/ ...
Chemistry of Life
... – Absorb and release water with little temp. change – Environmental changes (internal and external) ...
... – Absorb and release water with little temp. change – Environmental changes (internal and external) ...
AP Biology, Chapter 5, 9th ed. The Structure and Function of Large
... Ionic bonds between basic and acidic side chains 19. Using collagen and hemoglobin as examples, describe quaternary protein structure. Quaternary = more than one amino acid chain bound together Collagen: three helical amino acid chains wound into a triple helix Hemoglobin: 2 alpha + 2 beta chains + ...
... Ionic bonds between basic and acidic side chains 19. Using collagen and hemoglobin as examples, describe quaternary protein structure. Quaternary = more than one amino acid chain bound together Collagen: three helical amino acid chains wound into a triple helix Hemoglobin: 2 alpha + 2 beta chains + ...
Making Proteins
... the active I am going tomore try rate ofyou a chemical reaction ready for some rememberbindsI am a product, too. The substrate convert you.substrate sucrose! – Build up me? ortobreak down to me. I am a fructose now. • Fit together with its substrate like a “lock” and a I am now“key” a product. In ad ...
... the active I am going tomore try rate ofyou a chemical reaction ready for some rememberbindsI am a product, too. The substrate convert you.substrate sucrose! – Build up me? ortobreak down to me. I am a fructose now. • Fit together with its substrate like a “lock” and a I am now“key” a product. In ad ...
Buffers
... E) None of these peptide will be alpha helical because they all contain lefthanded amino acids. ...
... E) None of these peptide will be alpha helical because they all contain lefthanded amino acids. ...
aminoacids 2
... Characteristics of Homocystinuria high plasma and urinary levels of homocysteine and methionine and low levels of cysteine. ectopia lentis (displacement of the lens of the eye), skeletal abnormalities, a tendency to form thrombi (blood clots), osteoporosis, and ...
... Characteristics of Homocystinuria high plasma and urinary levels of homocysteine and methionine and low levels of cysteine. ectopia lentis (displacement of the lens of the eye), skeletal abnormalities, a tendency to form thrombi (blood clots), osteoporosis, and ...
2.1 Molecules to metabolim
... involving carbon. http://commons.wikimedia.org/wiki/File:Carbon-atom.jpg http://www.ks.uiuc.edu/images/ofmonth/2002-11/titin.jpg http://upload.wikimedia.org/wikipedia/commons/3/3e/Methane-2D-dot-cross.png ...
... involving carbon. http://commons.wikimedia.org/wiki/File:Carbon-atom.jpg http://www.ks.uiuc.edu/images/ofmonth/2002-11/titin.jpg http://upload.wikimedia.org/wikipedia/commons/3/3e/Methane-2D-dot-cross.png ...
BB350 Lecture 36 Highlights
... 1. Anticancer drugs and antibiotics sometimes target folates. Bacterial cells start synthesis of folates with para-amino benzoic acid and drugs that mimic this are the sulfa drugs. Humans get folate in their diet, so we are not susceptible to action of these drugs. Methotrexate is an anti-cancer dru ...
... 1. Anticancer drugs and antibiotics sometimes target folates. Bacterial cells start synthesis of folates with para-amino benzoic acid and drugs that mimic this are the sulfa drugs. Humans get folate in their diet, so we are not susceptible to action of these drugs. Methotrexate is an anti-cancer dru ...
Cellular Energy Unit Vocabulary California Standard
... In order to accomplish these lofty goals and be successful on the CSTs (proficient or advanced), you will be responsible for knowing and understanding the following vocabulary terms. ...
... In order to accomplish these lofty goals and be successful on the CSTs (proficient or advanced), you will be responsible for knowing and understanding the following vocabulary terms. ...
carbohydrate metabolism
... Catabolism of glucose • The paths that cells use to oxidize glucose completely to carbon dioxide involve many individual chemical reactions. These reactions occur in three different stages. These are: - initial break down of glucose to pyruvate in glycolysis, - further degradation of pyruvate to ac ...
... Catabolism of glucose • The paths that cells use to oxidize glucose completely to carbon dioxide involve many individual chemical reactions. These reactions occur in three different stages. These are: - initial break down of glucose to pyruvate in glycolysis, - further degradation of pyruvate to ac ...
Learning Guide: Molecules of Life Bill Activity #19 1st Read About
... o List the monomer of carbohydrates and its basic formula. List the two functional groups that all sugars have. o Sketch the abbreviated ring structure of glucose. Number the carbons on the structure. Glucose is a monosaccharide. Describe the important function of these molecules. o Explain/sketch h ...
... o List the monomer of carbohydrates and its basic formula. List the two functional groups that all sugars have. o Sketch the abbreviated ring structure of glucose. Number the carbons on the structure. Glucose is a monosaccharide. Describe the important function of these molecules. o Explain/sketch h ...
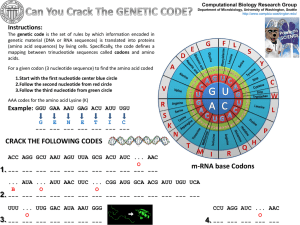
Slide 1
... The genetic code is the set of rules by which information encoded in genetic material (DNA or RNA sequences) is translated into proteins (amino acid sequences) by living cells. Specifically, the code defines a mapping between trinucleotide sequences called codons and amino acids. For a given codon ( ...
... The genetic code is the set of rules by which information encoded in genetic material (DNA or RNA sequences) is translated into proteins (amino acid sequences) by living cells. Specifically, the code defines a mapping between trinucleotide sequences called codons and amino acids. For a given codon ( ...
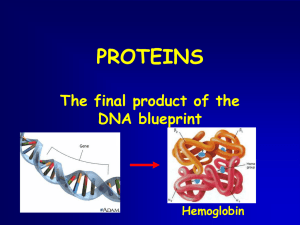
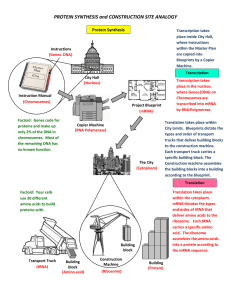
PROTEIN SYNTHESIS and CONSTRUCTION SITE ANALOGY
... within the Master Plan are copied into Blueprints by a Copier Machine. ...
... within the Master Plan are copied into Blueprints by a Copier Machine. ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.