Cellular Respiration
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
Handout12B
... end to end. Each of these smaller molecules can be any one of twenty so called amino acids. This long chain appears in a cell folded up on itself in a complicated fashion. In particular, its interactions with the other molecules in the cell are determined very much by the particular pattern of foldi ...
... end to end. Each of these smaller molecules can be any one of twenty so called amino acids. This long chain appears in a cell folded up on itself in a complicated fashion. In particular, its interactions with the other molecules in the cell are determined very much by the particular pattern of foldi ...
4 Amino Acids - School of Chemistry and Biochemistry
... Pyrrolysine (Pyl or O) is a genetically coded amino acid used by some methanogenic archaea and one known bacterium. Pyrrolysine is used in enzymes that are part of methaneproducing metabolism. Pyrrolysine is similar to lysine, but with an added pyrroline ring linked to the end of the lysine side cha ...
... Pyrrolysine (Pyl or O) is a genetically coded amino acid used by some methanogenic archaea and one known bacterium. Pyrrolysine is used in enzymes that are part of methaneproducing metabolism. Pyrrolysine is similar to lysine, but with an added pyrroline ring linked to the end of the lysine side cha ...
Summary for Chapter 7 – Metabolism: Transformations
... The fatty acids of a triglyceride, on the other hand, cannot make glucose, but they can provide abundant acetyl CoA. Acetyl CoA may then enter the TCA cycle to release energy or combine with other molecules of acetyl CoA to make body fat. The body can use some amino acids to produce glucose, whereas ...
... The fatty acids of a triglyceride, on the other hand, cannot make glucose, but they can provide abundant acetyl CoA. Acetyl CoA may then enter the TCA cycle to release energy or combine with other molecules of acetyl CoA to make body fat. The body can use some amino acids to produce glucose, whereas ...
Carbon-based molecules are life`s building blocks.
... Carbohydrates include sugars and starches found in foods such as bread and pasta. Many lipids are fats or oils. Proteins are necessary for many functions in the body, including the formation of muscle tissue. Nucleic acids are the molecules that carry the genetic code for all living things. As you r ...
... Carbohydrates include sugars and starches found in foods such as bread and pasta. Many lipids are fats or oils. Proteins are necessary for many functions in the body, including the formation of muscle tissue. Nucleic acids are the molecules that carry the genetic code for all living things. As you r ...
Powerpoint
... contains the information to code for one complete protein PROTEINS are made up of a chain of amino acids Proteins determine many of the traits in an organism ...
... contains the information to code for one complete protein PROTEINS are made up of a chain of amino acids Proteins determine many of the traits in an organism ...
protein synthesis
... contains ribose as its sugar and substitutes the nitrogenous base uracil for thymine. • An RNA molecules almost always consists of a single ...
... contains ribose as its sugar and substitutes the nitrogenous base uracil for thymine. • An RNA molecules almost always consists of a single ...
ChemicalBondingPowerpoint
... H2O) probably dominated Earth’s early atmosphere, but H2, NH3, and CH4 were also present in sufficient amounts to form H2CO and HCN. ...
... H2O) probably dominated Earth’s early atmosphere, but H2, NH3, and CH4 were also present in sufficient amounts to form H2CO and HCN. ...
Chapter 6 study guide key
... 2. Large molecules containing carbon atoms are called micromolecules. ...
... 2. Large molecules containing carbon atoms are called micromolecules. ...
File
... Name: Period: (1) Explain how monomers are related to polymers. (2) Explain the process that occurs when polymers are broken down into monomers, like for example when carbohydrates are broken down into simple sugars. ...
... Name: Period: (1) Explain how monomers are related to polymers. (2) Explain the process that occurs when polymers are broken down into monomers, like for example when carbohydrates are broken down into simple sugars. ...
GEE Science Assessment PowerPoint WITH answers
... • Thorugh, RNA, the DNA code leaves the nucleus and guides the construction of the protein in the ribosomes in cytoplasm. OR • RNA is used to pick up amino acids and bring them to where the protein is being made (that is, the ribosome). OR • RNA makes sure the building blocks of proteins (that is, a ...
... • Thorugh, RNA, the DNA code leaves the nucleus and guides the construction of the protein in the ribosomes in cytoplasm. OR • RNA is used to pick up amino acids and bring them to where the protein is being made (that is, the ribosome). OR • RNA makes sure the building blocks of proteins (that is, a ...
Amino Acids, Peptides and Proteins
... 2. Disulfide bonds are formed between two ______________________ amino acids. 3. In the condensation of two amino acids one molecule of ___________ is lost. 4. Amino acids linked together in a chain are called a _____________. 5. Protein in the food we eat is digested by enzymes called ______. 6. A ...
... 2. Disulfide bonds are formed between two ______________________ amino acids. 3. In the condensation of two amino acids one molecule of ___________ is lost. 4. Amino acids linked together in a chain are called a _____________. 5. Protein in the food we eat is digested by enzymes called ______. 6. A ...
Ch 2 ppt - Dover High School
... • sources of necessary ions (Na+, Cl-, K+, Ca2+, etc.) • play important roles in metabolism ...
... • sources of necessary ions (Na+, Cl-, K+, Ca2+, etc.) • play important roles in metabolism ...
Chapter 2b
... • The chain of Organic carbon atoms in an organic molecule is the carbon skeleton. • Functional groups are responsible for most of the chemical properties of a particular organic ...
... • The chain of Organic carbon atoms in an organic molecule is the carbon skeleton. • Functional groups are responsible for most of the chemical properties of a particular organic ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 16. What are the laws of thermodynamics? 17. Write about acid-base balance. 18. Explain the role of metals as cofactors of Enzymes. 19. Draw the structure of purines and pyrimidines. 20. List the phospholipids of membrane. SECTION B V. Answer any five questions, each in not more than 350 words (5X8= ...
... 16. What are the laws of thermodynamics? 17. Write about acid-base balance. 18. Explain the role of metals as cofactors of Enzymes. 19. Draw the structure of purines and pyrimidines. 20. List the phospholipids of membrane. SECTION B V. Answer any five questions, each in not more than 350 words (5X8= ...
Al - Iraqia university/ college of medicine
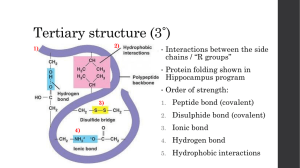
... Amino acids differ according to their R group. R groups range from a single hydrogen atom to a complicated ring compound. Some R groups are polar & some are not. Also, amino acid cysteine ends with an —SH group, which often serves to connect one chain of amino acids to another by a disulfide bond, — ...
... Amino acids differ according to their R group. R groups range from a single hydrogen atom to a complicated ring compound. Some R groups are polar & some are not. Also, amino acid cysteine ends with an —SH group, which often serves to connect one chain of amino acids to another by a disulfide bond, — ...
Chapter 3- DNA, Proteins and Proteomes
... 8. How many strands does DNA have? 9. How many strands does RNA have? 10. In DNA, state which bases pair up to form complementary base pairing. ...
... 8. How many strands does DNA have? 9. How many strands does RNA have? 10. In DNA, state which bases pair up to form complementary base pairing. ...
Document
... 1. Deduce the structure of one of the dipeptides formed between isoleucine and serine (2) ...
... 1. Deduce the structure of one of the dipeptides formed between isoleucine and serine (2) ...
Chapter 12 - Pathways to Biomolecules
... • Normally a small amount of this protein is present in the blood. • Abnormal cancerous or non-cancerous cell growths block the ducts in the prostate, forcing more of this protein into the bloodstream. • The concentration of this protein in the blood is used as a marker for possible prostate cancer. ...
... • Normally a small amount of this protein is present in the blood. • Abnormal cancerous or non-cancerous cell growths block the ducts in the prostate, forcing more of this protein into the bloodstream. • The concentration of this protein in the blood is used as a marker for possible prostate cancer. ...
Unit 2 Review Sheet - Discover more about NYLearns.org
... ___________________________________________________________________________________________ What is the equation for cellular respiration? What ORGANELLE does cellular respiration take place in? ___________________________________________________________________________________________ ...
... ___________________________________________________________________________________________ What is the equation for cellular respiration? What ORGANELLE does cellular respiration take place in? ___________________________________________________________________________________________ ...
DOC
... Molecules of ionic compounds contain ions. Ions are formed by complete transfer of electrons from one atom to another. You will recall that in the formation of sodium chloride (NaCl), one electron was transferred from sodium to chlorine. The result was formation of a sodium ion (Na+) and a chloride ...
... Molecules of ionic compounds contain ions. Ions are formed by complete transfer of electrons from one atom to another. You will recall that in the formation of sodium chloride (NaCl), one electron was transferred from sodium to chlorine. The result was formation of a sodium ion (Na+) and a chloride ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.