Review over Glucose Metabolism
... Review over Glucose Metabolism Match each of the following occurrences to the correct part of the glucose metabolism process. If more than one answer is correct, write all letters in the blank. A. Glycolysis B. Fermentation C. Cellular Respiration (aerobic) D. Krebs Cycle E. Electron Transport Chain ...
... Review over Glucose Metabolism Match each of the following occurrences to the correct part of the glucose metabolism process. If more than one answer is correct, write all letters in the blank. A. Glycolysis B. Fermentation C. Cellular Respiration (aerobic) D. Krebs Cycle E. Electron Transport Chain ...
Study Guide to Chapter 5 Ð DNA
... 3. RNA is an intermediary in making a protein. The first step is called ______________________ a) transcription b) translation, in which a DNA serves as a template to make a strand of ___________________ a) mRNA b) tRNA. This occurs in __________ a) the nucleus b) the ribosome). Once the strand is c ...
... 3. RNA is an intermediary in making a protein. The first step is called ______________________ a) transcription b) translation, in which a DNA serves as a template to make a strand of ___________________ a) mRNA b) tRNA. This occurs in __________ a) the nucleus b) the ribosome). Once the strand is c ...
Transcription & Translation PowerPoint
... 3. What would the amino acid sequence be if the first cytosine of the mRNA molecule was replaced with a uracil? (This would be due to a substitution mutation occurring to the DNA molecule which transcribed this mRNA.) ...
... 3. What would the amino acid sequence be if the first cytosine of the mRNA molecule was replaced with a uracil? (This would be due to a substitution mutation occurring to the DNA molecule which transcribed this mRNA.) ...
DNA and Proteins
... 8. tRNA then goes and finds the corresponding Amino Acid and brings them to the ribosome to be synthesized into protein. 9. The process of converting codons into anticodons and then amino acids is called Translation. More specifically, the mRNA creates codes for the proteins from DNA. These codes oc ...
... 8. tRNA then goes and finds the corresponding Amino Acid and brings them to the ribosome to be synthesized into protein. 9. The process of converting codons into anticodons and then amino acids is called Translation. More specifically, the mRNA creates codes for the proteins from DNA. These codes oc ...
All amino acids participate in these reactions at some
... All amino acids participate in these reactions at some point in their catabolism *** This is false; serine and threonine are not transaminated ¾ they are oxidatively deaminated (release NH3) by a dehydratase enzyme to form pyruvate and propionyl coA respectively. The first step in the catabolism of ...
... All amino acids participate in these reactions at some point in their catabolism *** This is false; serine and threonine are not transaminated ¾ they are oxidatively deaminated (release NH3) by a dehydratase enzyme to form pyruvate and propionyl coA respectively. The first step in the catabolism of ...
Chemistry SL HL Assessment Statements 2009 Revised
... The aim of this option is to give students an understanding of the chemistry of important molecules found in the human body, and the need for a balanced and healthy diet. Although the role that these molecules play in the body should be appreciated, the emphasis is placed on their chemistry, and stu ...
... The aim of this option is to give students an understanding of the chemistry of important molecules found in the human body, and the need for a balanced and healthy diet. Although the role that these molecules play in the body should be appreciated, the emphasis is placed on their chemistry, and stu ...
Biomolecule Review Worksheet
... five separate parts. The first is a central carbon atom. Second is a carboxyl group (-COOH). Third is an amino group (-NH2). Fourth is a hydrogen. The fifth group is a variable ‘R’ group. The only difference in the 20 kinds of amino acids is the “R” group. Some “R” groups are very small, others are ...
... five separate parts. The first is a central carbon atom. Second is a carboxyl group (-COOH). Third is an amino group (-NH2). Fourth is a hydrogen. The fifth group is a variable ‘R’ group. The only difference in the 20 kinds of amino acids is the “R” group. Some “R” groups are very small, others are ...
Name__________________________ Date______ Period
... 2. Proteins are made of chains of ___________ __________ held together by ___________ bonds. 3. How many amino acids are there? 5. Each combination of three nucleotides on mRNA is called a _____________ and codes for a specific __________ __________. 7. Do some amino acids have more than one codon? ...
... 2. Proteins are made of chains of ___________ __________ held together by ___________ bonds. 3. How many amino acids are there? 5. Each combination of three nucleotides on mRNA is called a _____________ and codes for a specific __________ __________. 7. Do some amino acids have more than one codon? ...
Biochemistry…
... catalyst is a substance that decreases the activation energy for a reaction and increases the rate of reaction Enzymes ...
... catalyst is a substance that decreases the activation energy for a reaction and increases the rate of reaction Enzymes ...
Metabolism of Glucose C6H12O6+6O2 1 unit of Glucose 38 ATP
... In exercise, first you burn off muscle glycogen, then liver glycogen, then lipid. If you only exercise In Diabetes, the cells cannot use glucose for energy. Only lipids. It depends on lipids, but the the Fatty Acids produce much more acetyl-CoA that kreb cycle cannot handle. This results in an accu ...
... In exercise, first you burn off muscle glycogen, then liver glycogen, then lipid. If you only exercise In Diabetes, the cells cannot use glucose for energy. Only lipids. It depends on lipids, but the the Fatty Acids produce much more acetyl-CoA that kreb cycle cannot handle. This results in an accu ...
gln.val.tyr.ala lys.arg.glu.trp met.his.leu.asp cys.pro.gly.asn F-A-D
... (amino terminal or carboxyl terminal?) is then removed in _______________ (acid or alkaline?) conditions, and the resulting amino acid derivative, known as a ___________________, is analyzed by chromatography. ...
... (amino terminal or carboxyl terminal?) is then removed in _______________ (acid or alkaline?) conditions, and the resulting amino acid derivative, known as a ___________________, is analyzed by chromatography. ...
CHAPTER-III CARBOHYDRATE METABOLISM
... organisms to generate energy through the oxidization of acetate derived from carbohydrates, fats and proteins into carbon dioxide. In addition, the cycle provides precursors including certain amino acids as well as the reducing agent NADH that is used in numerous biochemical reactions. Its central ...
... organisms to generate energy through the oxidization of acetate derived from carbohydrates, fats and proteins into carbon dioxide. In addition, the cycle provides precursors including certain amino acids as well as the reducing agent NADH that is used in numerous biochemical reactions. Its central ...
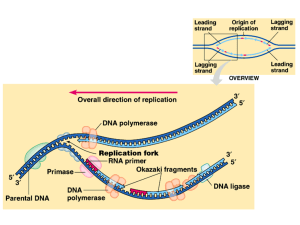
10 DNA Vocabulary - Petal School District
... 3. nucleotide—the monomer for nucleic acids; made of a phosphate, sugar, and nitrogen base 4. hydrogen bonds—hold nitrogen base pairs together 5. genetic code—the sequence of the nitrogen bases (nucleotides) on DNA 6. DNA replication—process that copies the DNA 7. template strands—the original stran ...
... 3. nucleotide—the monomer for nucleic acids; made of a phosphate, sugar, and nitrogen base 4. hydrogen bonds—hold nitrogen base pairs together 5. genetic code—the sequence of the nitrogen bases (nucleotides) on DNA 6. DNA replication—process that copies the DNA 7. template strands—the original stran ...
structure of proteins
... amino acid. Ninhydrin test result is positive for alpha amino acids, proteins and also peptides. Ninhydrin reagent is a strong oxidizing agent which reacts with amino acid to give purple colour due to the formation of a complex called Ruhemann’s purple. ...
... amino acid. Ninhydrin test result is positive for alpha amino acids, proteins and also peptides. Ninhydrin reagent is a strong oxidizing agent which reacts with amino acid to give purple colour due to the formation of a complex called Ruhemann’s purple. ...
Section 7-1
... 1. The fermentation pathways can operate in the absence of oxygen. 2. The energy-containing products are NADH, ATP, and pyruvic acid. 3. These pathways regenerate NAD_, which the cells can use to keep glycolysis going to make more ATP in the absence of oxygen. 4. Without niacin or the ability to mak ...
... 1. The fermentation pathways can operate in the absence of oxygen. 2. The energy-containing products are NADH, ATP, and pyruvic acid. 3. These pathways regenerate NAD_, which the cells can use to keep glycolysis going to make more ATP in the absence of oxygen. 4. Without niacin or the ability to mak ...
Organic Compounds
... • Hydrophobic (held together by non-polar covalent bonds) • Contains MORE energy-rich C-H bonds than carbs • Types include: neutral fats, phospholipids, steroids, carotenoids, waxes – Neutral fats: most abundant type of lipid 3 Fatty Acids + glycerol • Simplest lipid = fatty acid ...
... • Hydrophobic (held together by non-polar covalent bonds) • Contains MORE energy-rich C-H bonds than carbs • Types include: neutral fats, phospholipids, steroids, carotenoids, waxes – Neutral fats: most abundant type of lipid 3 Fatty Acids + glycerol • Simplest lipid = fatty acid ...
Introduction to Plant Products and Human Affairs
... – Thus, converting carbon dioxide and water into sugar needs energy from sunlight added, and metabolizing sugar back into carbon dioxide and ...
... – Thus, converting carbon dioxide and water into sugar needs energy from sunlight added, and metabolizing sugar back into carbon dioxide and ...
(key)
... ld("ATPase e. Dehydrogenase f. 5· In what process is ubiquitin used? Breifly describe it action. LJN4 t ~UWl, '! ~oftt;tt~s- 74...-~~ pr"-f_.:.~ {;7 chJY~nWI ...
... ld("ATPase e. Dehydrogenase f. 5· In what process is ubiquitin used? Breifly describe it action. LJN4 t ~UWl, '! ~oftt;tt~s- 74...-~~ pr"-f_.:.~ {;7 chJY~nWI ...
ECA Biochemistry Gizmos
... The 2 kinds of nucleic acids are DNA and RNA which store and transmit genetic information (the “blueprint” to make you). ...
... The 2 kinds of nucleic acids are DNA and RNA which store and transmit genetic information (the “blueprint” to make you). ...
Chapter 2 Chemistry Test Review
... 1. What are the three subatomic particles and their charges? a. Protons (+) b. Neutrons (0) c. Electrons (-) 2. What are the four organic compounds found in living things and what do they do? a. Carbohydrates – Main source of energy b. Lipids – Store energy c. Proteins – Chemical messengers d. Nucle ...
... 1. What are the three subatomic particles and their charges? a. Protons (+) b. Neutrons (0) c. Electrons (-) 2. What are the four organic compounds found in living things and what do they do? a. Carbohydrates – Main source of energy b. Lipids – Store energy c. Proteins – Chemical messengers d. Nucle ...
K,Mg,Ca,Na… 0,4%
... Microorganisms require about 10 elements in large quantities (C, N, H, O, S, P, K, Ca, Mg, Fe); others as microelements or trace elements (Mn, Zn, Co, Mo, Cu…..) – part of enzymes and cofactors Nutrients are used in biosynthesis and as a source of energy (chemotrophs) CARBON – the skeleton of ...
... Microorganisms require about 10 elements in large quantities (C, N, H, O, S, P, K, Ca, Mg, Fe); others as microelements or trace elements (Mn, Zn, Co, Mo, Cu…..) – part of enzymes and cofactors Nutrients are used in biosynthesis and as a source of energy (chemotrophs) CARBON – the skeleton of ...
sci 7 study guide
... 2 types of cells: Prokaryotes (bacteria), Eukaryotes (plants & animals) Mammals: breathe air, give birth to live young Genus-group of species; when a scientist discovers a new species, it is placed with the species with which it shares the most characteristics Structure and Function of Living Organi ...
... 2 types of cells: Prokaryotes (bacteria), Eukaryotes (plants & animals) Mammals: breathe air, give birth to live young Genus-group of species; when a scientist discovers a new species, it is placed with the species with which it shares the most characteristics Structure and Function of Living Organi ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.