lec-2- 211(ES +Add)
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
... HX which can donate a proton, H2O should be able to add to alkenes in the same way as HBr, for example, resulting in the hydration of an alkene. However, for the addition of H2O to alkenes to occur acid catalysts are required. ...
Revision
... A C7H13Br compound reacts with KOH in ethanol to form 3methylcyclohexene as the major product. What is a likely structure for the starting alkyl bromide? ...
... A C7H13Br compound reacts with KOH in ethanol to form 3methylcyclohexene as the major product. What is a likely structure for the starting alkyl bromide? ...
Carboxylic Acid Derivatives
... Esters can also react with amines or ammonia to form amides. This reaction doesn't involve acid catalysis, so the first step is nucleophilic attack at the carbonyl carbon. Proton transfer follows and loss of the alcohol portion of the ester. ...
... Esters can also react with amines or ammonia to form amides. This reaction doesn't involve acid catalysis, so the first step is nucleophilic attack at the carbonyl carbon. Proton transfer follows and loss of the alcohol portion of the ester. ...
Aldehydes - Sanfordchemistrystudentwork
... Aldehydes A wide variety of Aldehydes have been isolated from plants and animals. Many of them, particularly those with high molar masses, have fragrant or penetrating odors. They are usually known by their common names, which can indicate their natural sources or perhaps a characteristic property ...
... Aldehydes A wide variety of Aldehydes have been isolated from plants and animals. Many of them, particularly those with high molar masses, have fragrant or penetrating odors. They are usually known by their common names, which can indicate their natural sources or perhaps a characteristic property ...
Practical, Asymmetric Redox-Neutral Chemical Synthesis via Borrowing Hydrogen
... available alcohols through a multi-step process involving oxidation of alcohol to ketone, condensation with amine to imine followed by reduction. As both the oxidation and reduction steps in this process utilize stoichiometric reagents, much waste is generated. The concept of “borrowing hydrogen” ha ...
... available alcohols through a multi-step process involving oxidation of alcohol to ketone, condensation with amine to imine followed by reduction. As both the oxidation and reduction steps in this process utilize stoichiometric reagents, much waste is generated. The concept of “borrowing hydrogen” ha ...
CHEM 201 Name Quiz 10 (Ch 17) ID Q1. Which of the following
... the ester shown below with LiAlH4? O O ...
... the ester shown below with LiAlH4? O O ...
Document
... The formation of carbon-carbon bonds is one of the most widely studied areas in organic synthesis. One class of carbon-carbon bond forming reactions involves the nucleophilic addition of vinyl or allyl organometallics to aldhydes, yielding allylic or homoallylic alcohols. The stereochemical unpredic ...
... The formation of carbon-carbon bonds is one of the most widely studied areas in organic synthesis. One class of carbon-carbon bond forming reactions involves the nucleophilic addition of vinyl or allyl organometallics to aldhydes, yielding allylic or homoallylic alcohols. The stereochemical unpredic ...
SN1 vs. SN2 Reactions - Master Organic Chemistry
... Alternatively, look for alcohols (OH) if acid is present Once you've identified the leaving group, instpect the carbon it is attached to. How many carbons is that carbon connected to? That will tell you if the carbon is primary, secondary, or tertiary. If there are no attached carbons, that's the sp ...
... Alternatively, look for alcohols (OH) if acid is present Once you've identified the leaving group, instpect the carbon it is attached to. How many carbons is that carbon connected to? That will tell you if the carbon is primary, secondary, or tertiary. If there are no attached carbons, that's the sp ...
Chem 400 Review Chem 350 JJ.S17
... Alcohols are names with the suffix –ol The acidity of an alcohol/phenol can be assessed using its conjugate base: Consider Atomic size/Electronegativity, Resonance, Induction, Orbital Type (ARIO) Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabili ...
... Alcohols are names with the suffix –ol The acidity of an alcohol/phenol can be assessed using its conjugate base: Consider Atomic size/Electronegativity, Resonance, Induction, Orbital Type (ARIO) Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabili ...
Microsoft Word - Final Exam Study Guide
... stability, elimination reactions, Zaitsev’s rule, E1 mechanism, E2 mechanism, antiperiplanar, comparing substitution and elimination mechanisms, synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesi ...
... stability, elimination reactions, Zaitsev’s rule, E1 mechanism, E2 mechanism, antiperiplanar, comparing substitution and elimination mechanisms, synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesi ...
CHE 322
... a. 2-heptanone from simple alcohols of 4 C or fewer. No strong base like LDA is available. (10 pt) ...
... a. 2-heptanone from simple alcohols of 4 C or fewer. No strong base like LDA is available. (10 pt) ...
Unit 4_Carbonyl and carboxylic acid questions
... provided with pentanoyl chloride, water and ammonia as possible starting reagents. Give reagents and conditions for each step required and name every compound used/produced in the reaction sequence. 12. Answer Q6 attached (June 08 unit paper). Try to answer as much as you can if attempting this ques ...
... provided with pentanoyl chloride, water and ammonia as possible starting reagents. Give reagents and conditions for each step required and name every compound used/produced in the reaction sequence. 12. Answer Q6 attached (June 08 unit paper). Try to answer as much as you can if attempting this ques ...
Chapter 18 - Aldehydes and Ketones
... However, it is possible to reduce the reactivity of the hydride reagent by replacing some hydrogen atoms by alkoxy groups. In such cases, the reagent is not active enough to reduce the intermediate, and the aldehyde is obtained. ...
... However, it is possible to reduce the reactivity of the hydride reagent by replacing some hydrogen atoms by alkoxy groups. In such cases, the reagent is not active enough to reduce the intermediate, and the aldehyde is obtained. ...
Exam 2 Review A
... three things: 1. React with nucleophiles, 2. -eliminate (lose H+) to form an alkene, 3. undergo skeletal rearrangements via 1,2-hydride shifts or 1,2-methanide shifts. [we will defer discussion of #3 until Chapter 7]. Remember, carbocation stability plays a role in analyzing transition states, whic ...
... three things: 1. React with nucleophiles, 2. -eliminate (lose H+) to form an alkene, 3. undergo skeletal rearrangements via 1,2-hydride shifts or 1,2-methanide shifts. [we will defer discussion of #3 until Chapter 7]. Remember, carbocation stability plays a role in analyzing transition states, whic ...
슬라이드 1
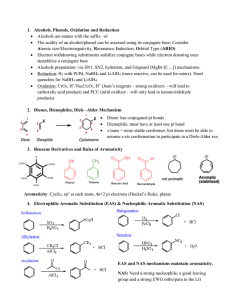
... Existing substituent groups such as CH3, OCH3, and +NMe3 exert a directive effect, often resulting in a major amount of the meta substitution product. ...
... Existing substituent groups such as CH3, OCH3, and +NMe3 exert a directive effect, often resulting in a major amount of the meta substitution product. ...
Asymmetric induction

Asymmetric induction (also enantioinduction) in stereochemistry describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis.Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist.Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst of chiral ligand. This method of asymmetric synthesis is economically most desirable.