Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... recovered GO (pH = 3.95 at 0.29 mg mL−1) indicated the slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased t ...
... recovered GO (pH = 3.95 at 0.29 mg mL−1) indicated the slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased t ...
CHAPTER-7
... 21. In a reversible reaction K = Qc, what does it signify? Ans. The reaction is at equilibrium. 22. In a reversible reaction Qc > K, predict the direction in which the reaction proceeds? Ans. Net reaction goes from right to left i.e., towards backward direction. 23. Give the relationship Gibb’s free ...
... 21. In a reversible reaction K = Qc, what does it signify? Ans. The reaction is at equilibrium. 22. In a reversible reaction Qc > K, predict the direction in which the reaction proceeds? Ans. Net reaction goes from right to left i.e., towards backward direction. 23. Give the relationship Gibb’s free ...
Ch. 6 - Department of Chemistry and Biochemistry
... SN2 Reactions in Polar Protic Solvents ● In polar protic solvents, the Nu⊖ anion is solvated by the surrounding protic solvent which makes the e⊖ pair of the Nu⊖ less available and thus less reactive in SN2 reactions ...
... SN2 Reactions in Polar Protic Solvents ● In polar protic solvents, the Nu⊖ anion is solvated by the surrounding protic solvent which makes the e⊖ pair of the Nu⊖ less available and thus less reactive in SN2 reactions ...
Chapter 14 - Moore Public Schools
... products until a new position of equilibrium is found that has the same K. • Removing a product will increase the amounts of the other products and decrease the amounts of the reactants. – You can use this to drive a reaction to completion! • Equilibrium shifts away from the side with added chemical ...
... products until a new position of equilibrium is found that has the same K. • Removing a product will increase the amounts of the other products and decrease the amounts of the reactants. – You can use this to drive a reaction to completion! • Equilibrium shifts away from the side with added chemical ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... SOLUBILITY RULES: for Ionic Compounds (Salts) 1. All salts of alkali metals (IA) are soluble. 2. All NH4+ salts are soluble. 3. All salts containing the anions: NO3-, ClO3-, ClO4-, (C2H3O2-) are soluble. 4. All Cl-, Br-, and I- are soluble except for Hg22+, Ag+, and Pb2+ salts. 5. All SO42- are solu ...
... SOLUBILITY RULES: for Ionic Compounds (Salts) 1. All salts of alkali metals (IA) are soluble. 2. All NH4+ salts are soluble. 3. All salts containing the anions: NO3-, ClO3-, ClO4-, (C2H3O2-) are soluble. 4. All Cl-, Br-, and I- are soluble except for Hg22+, Ag+, and Pb2+ salts. 5. All SO42- are solu ...
Topic 3: Chemical Kinetics - Manitoba Education and Training
... Examples: analogies, concept frames, concept maps, manipulatives, particulate representations, roleplays, simulations, sort-and-predict frames, word cycles . . . ...
... Examples: analogies, concept frames, concept maps, manipulatives, particulate representations, roleplays, simulations, sort-and-predict frames, word cycles . . . ...
Direct conversion of cellulose into sorbitol using dual
... respectively. Liquid-phase mineral acids or heteropolyacids have been used together with metal catalysts containing Pt or Ru [8,9]. These acids, however, are difficult to recover and can cause corrosion of the reactor. In addition, a large amount of waste sludge is formed in the acid neutralization p ...
... respectively. Liquid-phase mineral acids or heteropolyacids have been used together with metal catalysts containing Pt or Ru [8,9]. These acids, however, are difficult to recover and can cause corrosion of the reactor. In addition, a large amount of waste sludge is formed in the acid neutralization p ...
CHM203 - National Open University of Nigeria
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
HIGHLY SELECTIVE RHODIUM–CATALYZED C–H BORYLATIONS IN
... List of Schemes Scheme 1-1. General process of directed ortho–Metalation (DoM).............................................. 3 Scheme 1-2. Sequential DoM/Suzuki–Miyaura cross–coupling for synthesis of 1,2–disubstituted biaryls by Snieckus and coworkers .............................................. ...
... List of Schemes Scheme 1-1. General process of directed ortho–Metalation (DoM).............................................. 3 Scheme 1-2. Sequential DoM/Suzuki–Miyaura cross–coupling for synthesis of 1,2–disubstituted biaryls by Snieckus and coworkers .............................................. ...
Kinetics
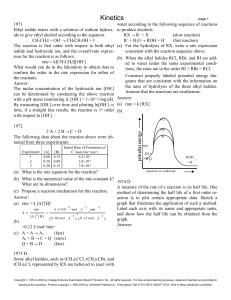
... ride and oxalate ion in hot aqueous solution is shown above. The reaction rate may be determined by meas- (b) A small increase in temperature causes a large inuring the initial rate of formation of chloride ion, at crease in the reaction rate. constant temperature, for various initial concentrations ...
... ride and oxalate ion in hot aqueous solution is shown above. The reaction rate may be determined by meas- (b) A small increase in temperature causes a large inuring the initial rate of formation of chloride ion, at crease in the reaction rate. constant temperature, for various initial concentrations ...
Forward
... in much the same way that Grignard reagents do, except that they function as hydride donors rather than as carbanion sources. Borohydride transfers a hydrogen with its pair of bonding electrons to the positively polarized carbon of a carbonyl group. The negatively polarized oxygen attacks boron. Ult ...
... in much the same way that Grignard reagents do, except that they function as hydride donors rather than as carbanion sources. Borohydride transfers a hydrogen with its pair of bonding electrons to the positively polarized carbon of a carbonyl group. The negatively polarized oxygen attacks boron. Ult ...
Chap 4 - Bakersfield College
... – The concept of oxidation numbers is a simple way of keeping track of electrons in a reaction. – The oxidation number (or oxidation state) of an atom in a substance is the actual charge of the atom if it exists as a monatomic ion. – Alternatively, it is hypothetical charge assigned to the atom in t ...
... – The concept of oxidation numbers is a simple way of keeping track of electrons in a reaction. – The oxidation number (or oxidation state) of an atom in a substance is the actual charge of the atom if it exists as a monatomic ion. – Alternatively, it is hypothetical charge assigned to the atom in t ...
Glycosyl amines
... • The method of preparation of glycosides from free aldoses or ketoses and aliphatic alcohols in the presence of anhydrous acids, usually hydrogen chloride. • In the course of the reaction a decrease in concentration of the starting aldose or ketose (in general, glycose) is accompanied by a rapid, b ...
... • The method of preparation of glycosides from free aldoses or ketoses and aliphatic alcohols in the presence of anhydrous acids, usually hydrogen chloride. • In the course of the reaction a decrease in concentration of the starting aldose or ketose (in general, glycose) is accompanied by a rapid, b ...
Improved Synthesis, Separation, Transition Metal Coordination and
... acetone-d6/D2O recorded at 15°C (light blue), 10°C (dark blue), 25°C (black), 50°C (orange), 80°C (purple), and 100°C (red). For higher temperatures the NMR tube was tube pressurized to 90 psi with O2 ............................................................................... 59 H spectra of m ...
... acetone-d6/D2O recorded at 15°C (light blue), 10°C (dark blue), 25°C (black), 50°C (orange), 80°C (purple), and 100°C (red). For higher temperatures the NMR tube was tube pressurized to 90 psi with O2 ............................................................................... 59 H spectra of m ...
Problem 28. TUNNELING IN CHEMISTRY
... In chemical reactions molecular structure changes over time so that the electronic state of a molecule is a function of time. In some cases structure of a molecule can be presented by a superposition of the initial and final states with time-dependent coefficients. Let’s assume that a molecule oscil ...
... In chemical reactions molecular structure changes over time so that the electronic state of a molecule is a function of time. In some cases structure of a molecule can be presented by a superposition of the initial and final states with time-dependent coefficients. Let’s assume that a molecule oscil ...
Ion Exchange Resins: Catalyst Recovery and Recycle
... general family of single-site catalysts whose features were detailed by Thomas et al.58 The catalytic performances of these heterogenized catalysts may vary enormously depending on the immobilization method and on the support, so that it is of outmost importance to get a systematic picture of favora ...
... general family of single-site catalysts whose features were detailed by Thomas et al.58 The catalytic performances of these heterogenized catalysts may vary enormously depending on the immobilization method and on the support, so that it is of outmost importance to get a systematic picture of favora ...
word - My eCoach
... Lab: Determining the Formula of a Hydrate Introduction When certain ionic solids crystallize from aqueous solutions, a definite number of molecules of water remain attached to the crystal. Ionic solids that contain a definite amount of water are called hydrates or hydrated salts and the water in th ...
... Lab: Determining the Formula of a Hydrate Introduction When certain ionic solids crystallize from aqueous solutions, a definite number of molecules of water remain attached to the crystal. Ionic solids that contain a definite amount of water are called hydrates or hydrated salts and the water in th ...
Asymmetric induction

Asymmetric induction (also enantioinduction) in stereochemistry describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis.Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist.Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst of chiral ligand. This method of asymmetric synthesis is economically most desirable.