Chap. 3 : Biochemistry - Fort Thomas Independent Schools
... and a long alcohol chain ( the length is what determines how good a waterproofer it is.) D. Steroid – made up of 4-carbon rings - considered a lipid because they do not dissolve in water. ...
... and a long alcohol chain ( the length is what determines how good a waterproofer it is.) D. Steroid – made up of 4-carbon rings - considered a lipid because they do not dissolve in water. ...
Solution Key - Chemistry With BT
... Show the homolysis of the bromine molecule using curly arrows ...
... Show the homolysis of the bromine molecule using curly arrows ...
Types of Reactions in Organic Chemistry Chemistry
... The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitution reaction takes place when methane rea ...
... The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitution reaction takes place when methane rea ...
Blank Final Exam from 2004 - Department of Chemistry | Oregon
... amine group in compound A was neutralized with HCl to produce structure B, an ammonium salt. ...
... amine group in compound A was neutralized with HCl to produce structure B, an ammonium salt. ...
Organic Molecule Building Block
... DNA has the information for the synthesis of proteins from amino acids. Some of these proteins are enzymes that control the rate of chemical reactions in a cell. ...
... DNA has the information for the synthesis of proteins from amino acids. Some of these proteins are enzymes that control the rate of chemical reactions in a cell. ...
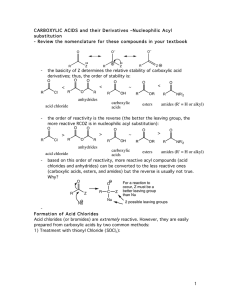
carboxylic acids esters amides (R
... So, remember...anhydrides react just about like acid chlorides. ...
... So, remember...anhydrides react just about like acid chlorides. ...
18 Important and sometimes forgotten) organic transformations
... •Phosphines can also be used •DMAP and DBU are better in some cases ...
... •Phosphines can also be used •DMAP and DBU are better in some cases ...
Unit 3 Goals - kimscience.com
... o complete and balance combustion reactions of organic molecules containing carbon, hydrogen and oxygen, and explain the reaction in terms of bonds breaking and forming, enthalpy, and entropy change. o distinguish between complete and incomplete combustion in terms of reaction conditions, resulting ...
... o complete and balance combustion reactions of organic molecules containing carbon, hydrogen and oxygen, and explain the reaction in terms of bonds breaking and forming, enthalpy, and entropy change. o distinguish between complete and incomplete combustion in terms of reaction conditions, resulting ...
ANSWERS Concept Checks: Ch. 5 The Molecules of Life Concept
... 4. Which parts of an amino acid's structure are the same in all amino acids? Which part is unique? Central carbon is bonded to a hydrogen atom, an amino group, and a carboxyl group. The fourth bond is with a side or functional group that is unique to that amino acid. Concept Check 5.5 1. Explain the ...
... 4. Which parts of an amino acid's structure are the same in all amino acids? Which part is unique? Central carbon is bonded to a hydrogen atom, an amino group, and a carboxyl group. The fourth bond is with a side or functional group that is unique to that amino acid. Concept Check 5.5 1. Explain the ...
doc CHEM 222 Lab exam with Answers
... temperature and then allowing them to come back out of solution. 2.__T___ The purpose of refluxing is to carry out a reaction at the boiling point of the solvent. 3.__F___ All chemical reactions must take place in solution. 4.__T___ When a carbene is formed in the presence of an alkene, a cyclopropa ...
... temperature and then allowing them to come back out of solution. 2.__T___ The purpose of refluxing is to carry out a reaction at the boiling point of the solvent. 3.__F___ All chemical reactions must take place in solution. 4.__T___ When a carbene is formed in the presence of an alkene, a cyclopropa ...
Organic Chemistry –Syllabus- one Semester Sackler faculty of
... Organic Compounds + Alkanes double bond equivalent, alkyl group, Nomenclature (IUPAC rules), intermolecular forces( van der Waals force, Dipole–dipole interaction, Hydrogen bonds), Solubility, Conformations of alkanes(staggered-eclipsd) , Cycloalkanes, geometric isomers, The chair conformation of cy ...
... Organic Compounds + Alkanes double bond equivalent, alkyl group, Nomenclature (IUPAC rules), intermolecular forces( van der Waals force, Dipole–dipole interaction, Hydrogen bonds), Solubility, Conformations of alkanes(staggered-eclipsd) , Cycloalkanes, geometric isomers, The chair conformation of cy ...
Name: Chem 22 Final exam Spring `00 What product is formed when
... e) addtion of a hydride ion and a proton more or less at the same time 18. Which of the following describes “reductive amination?” a) an aldehyde or a ketone + a tertiary amine + H2/zeolite b) an aldehyde or a ketone + ammonia or a primary or a secondary amine + ...
... e) addtion of a hydride ion and a proton more or less at the same time 18. Which of the following describes “reductive amination?” a) an aldehyde or a ketone + a tertiary amine + H2/zeolite b) an aldehyde or a ketone + ammonia or a primary or a secondary amine + ...
10.4b Organic Practice Test Version 2
... a) Carbon atoms in the organic product are bonded to fewer atoms than the carbon atoms in the organic reactant. b) A hydrogen atom or functional group is replaced with a different atom or functional group. c) Atoms are added to a double or triple carbon–carbon bond. d) Two molecules are combined and ...
... a) Carbon atoms in the organic product are bonded to fewer atoms than the carbon atoms in the organic reactant. b) A hydrogen atom or functional group is replaced with a different atom or functional group. c) Atoms are added to a double or triple carbon–carbon bond. d) Two molecules are combined and ...
Unit 4_Carbonyl and carboxylic acid questions
... 4. Explain in terms of bonding why carbonyls have higher b.ps than corresponding alkanes but lower b.ps than corresponding alcohols. ...
... 4. Explain in terms of bonding why carbonyls have higher b.ps than corresponding alkanes but lower b.ps than corresponding alcohols. ...
Organic Chemistry Chapter 1
... • Hydrolysis of Nitriles, Esters, or Amides with The Addition of Acid or Base Roles of Acid or Base !!!!!! (page 298, 10.15) • Reaction of Grignard Reagents with Carbon Dioxide ...
... • Hydrolysis of Nitriles, Esters, or Amides with The Addition of Acid or Base Roles of Acid or Base !!!!!! (page 298, 10.15) • Reaction of Grignard Reagents with Carbon Dioxide ...
Document
... Give the structural formula of the monomer (containing two functional groups) that could be used to make this polyester and state the names of the two functional groups. ...
... Give the structural formula of the monomer (containing two functional groups) that could be used to make this polyester and state the names of the two functional groups. ...
Practical, Asymmetric Redox-Neutral Chemical Synthesis via Borrowing Hydrogen
... available alcohols through a multi-step process involving oxidation of alcohol to ketone, condensation with amine to imine followed by reduction. As both the oxidation and reduction steps in this process utilize stoichiometric reagents, much waste is generated. The concept of “borrowing hydrogen” ha ...
... available alcohols through a multi-step process involving oxidation of alcohol to ketone, condensation with amine to imine followed by reduction. As both the oxidation and reduction steps in this process utilize stoichiometric reagents, much waste is generated. The concept of “borrowing hydrogen” ha ...
اســـم المـــدرس: د
... 2) Write the chemical and the reaction mechanism for the reaction of benzaldehyde with excess methanol and acid catalyst. ...
... 2) Write the chemical and the reaction mechanism for the reaction of benzaldehyde with excess methanol and acid catalyst. ...
dr. Zdenko Časar - Fakulteta za kemijo in kemijsko tehnologijo
... building blocks, which were till now prepared with limited number of synthetic methods. Moreover, halo substituted analogues like chiral (α-chloroalkyl) boronic esters are even more interesting chiral building blocks, which can be utilized in various coupling reactions and can undergo functional gro ...
... building blocks, which were till now prepared with limited number of synthetic methods. Moreover, halo substituted analogues like chiral (α-chloroalkyl) boronic esters are even more interesting chiral building blocks, which can be utilized in various coupling reactions and can undergo functional gro ...
Key Practice Exam 3
... alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimm ...
... alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimm ...
Preparation and Reaction of Carboxylic Acids - IDC
... Two other useful procedures for preparing carboxylic acids involve hydrolysis of nitriles and carboxylation of organometallic intermediates. As shown in the following diagram, both methods begin with an organic halogen compound and the carboxyl group eventually replaces the halogen. Both methods req ...
... Two other useful procedures for preparing carboxylic acids involve hydrolysis of nitriles and carboxylation of organometallic intermediates. As shown in the following diagram, both methods begin with an organic halogen compound and the carboxyl group eventually replaces the halogen. Both methods req ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.