Lecture 39 - Chemistry at Winthrop University
... Progressively Oxidizing (adding Oxygen) the alcohol allows us to go from an alcohol to a carboxylic acid ...
... Progressively Oxidizing (adding Oxygen) the alcohol allows us to go from an alcohol to a carboxylic acid ...
- professional publication
... Reactions of Aromatic Amines and Alcohols Migration to Electron Deficient Nitrogen, Hofmanns, Beckmanns, Curtius, Smith, Diazotisation and Diazonium Salts and its Applications, Sandmeyers Diazocoupling Reactions. Basicity of Amines, Effect of Substituents on Basicity. Acidity of Phenols, Effect of S ...
... Reactions of Aromatic Amines and Alcohols Migration to Electron Deficient Nitrogen, Hofmanns, Beckmanns, Curtius, Smith, Diazotisation and Diazonium Salts and its Applications, Sandmeyers Diazocoupling Reactions. Basicity of Amines, Effect of Substituents on Basicity. Acidity of Phenols, Effect of S ...
DESIGN OF CHIRAL IMINO- AND AMINOPYRIDINE LIGANDS
... Doctoral Thesis by Victor Hernández Olmos The synthesis of new molecules that can work as chiral ligands in metal catalyzed enantioselective reactions is currently an area of great interest.1,2 These ligands must be capable of coordinating with the metal ion, generating around it a highly asymmetric ...
... Doctoral Thesis by Victor Hernández Olmos The synthesis of new molecules that can work as chiral ligands in metal catalyzed enantioselective reactions is currently an area of great interest.1,2 These ligands must be capable of coordinating with the metal ion, generating around it a highly asymmetric ...
Synthesis of an Ester: Fischer Esterification The ester is synthesised
... fewer carbons have moderate water solubility. As a result, they are partially extracted into the aqueous layer during work-up, reducing the amount recovered. ...
... fewer carbons have moderate water solubility. As a result, they are partially extracted into the aqueous layer during work-up, reducing the amount recovered. ...
Synthesis of a Family of Chiral Asymmetric Schiff - Blogs at H-SC
... Carbon-Carbon bond-forming reactions are essential synthetic methods for organic chemists. Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds have been shown in some cases to act as catalysts to give condensation products in high yi ...
... Carbon-Carbon bond-forming reactions are essential synthetic methods for organic chemists. Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds have been shown in some cases to act as catalysts to give condensation products in high yi ...
Procedure Notes
... once equilibrium is reached there could be a large amount of the starting materials remaining. This could result in a poor yield of the desired ester. Adding additional products could shift the reaction to the right, or adding additional water could lead to a shift to the left. O ...
... once equilibrium is reached there could be a large amount of the starting materials remaining. This could result in a poor yield of the desired ester. Adding additional products could shift the reaction to the right, or adding additional water could lead to a shift to the left. O ...
Alcohol Worksheet Key
... 4-chlorophenol is more acidic than 3-chlorophenol due to the inductive effect of the chlorine at position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
... 4-chlorophenol is more acidic than 3-chlorophenol due to the inductive effect of the chlorine at position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
CH 102 Practice exam This represents the new material that
... The amine salts tend to be less soluble than the amine. ...
... The amine salts tend to be less soluble than the amine. ...
Chapter 20: Carboxylic Acids and Nitriles
... -- NABH4 is less reactive than LAH therefore, only LAH works on acids and derivatives ...
... -- NABH4 is less reactive than LAH therefore, only LAH works on acids and derivatives ...
CHEMISTRY 3
... Because esters are made by forcing the equilibrium reaction between a carboxylic acid and an alcohol to the right, it should also be possible to split an ester back into its constituents by forcing the same equilibrium to shift itself in the reverse direction, ie to the left. (a) ...
... Because esters are made by forcing the equilibrium reaction between a carboxylic acid and an alcohol to the right, it should also be possible to split an ester back into its constituents by forcing the same equilibrium to shift itself in the reverse direction, ie to the left. (a) ...
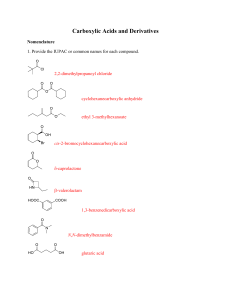
Derivatives of Carboxylic Acids
... • Salicylic acid is a bifunctional molecule (acid and phenol) which is itself a disinfectant. • Aspirin comes from the willow bark which was used in the Middle Ages by Jesuit missionaries. ...
... • Salicylic acid is a bifunctional molecule (acid and phenol) which is itself a disinfectant. • Aspirin comes from the willow bark which was used in the Middle Ages by Jesuit missionaries. ...
Chemistry_
... alcohols. The alcohols have a similar structure to the alkanes but with a –OH group replacing one of the hydrogens. ...
... alcohols. The alcohols have a similar structure to the alkanes but with a –OH group replacing one of the hydrogens. ...
Slide 1
... • Nucleophiles that form unstable addition products form conjugated addition products, because the conjugate addition is not reversible ...
... • Nucleophiles that form unstable addition products form conjugated addition products, because the conjugate addition is not reversible ...
Honors Chemistry Organic Chemistry
... b. carcinogen in burned meat and cigarettes c. organic bases d. hydrogenation e. hydroxyl f. carboxyl g. carbonyl h. containing benzene or benzene-like structures i. from wood distillation / metabolized into formaldehyde j. benzene as a substituent k. reaction in the formation of esters l. phenol ...
... b. carcinogen in burned meat and cigarettes c. organic bases d. hydrogenation e. hydroxyl f. carboxyl g. carbonyl h. containing benzene or benzene-like structures i. from wood distillation / metabolized into formaldehyde j. benzene as a substituent k. reaction in the formation of esters l. phenol ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.