Studies toward the Stereoselective Synthesis of the
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
... In the dissertation the retrosynhetic analysis of this 3,7-dimethylundecane-1,5,6-triol building block identifies a simple precursor, ethyl 2-heptenoate, as the starting material for the proposed synthetic route toward this target. The Sharpless asymmetric epoxidation reaction plays a pivotal role i ...
Chem 240 - Napa Valley College
... 10) Please show the mechanism for the reaction between methyl cyclohexene, bromine, and water. Four products are formed during this reaction. Give the complete mechanism for just one product but show how each product is formed and label them R,S appropriately 11) Esters do not have to be made from a ...
... 10) Please show the mechanism for the reaction between methyl cyclohexene, bromine, and water. Four products are formed during this reaction. Give the complete mechanism for just one product but show how each product is formed and label them R,S appropriately 11) Esters do not have to be made from a ...
Triphase Catalysis. Applications to Organic Synthesis`
... was transferred to a separatory funnel containing 75 ml each of water and benzene. The aqueous layer was removed and the benzene solution was washed with water (100 ml), saturated aqueous NaHCO 1 (100 ml), and twice with 100-ml portions of water. After drying (MgSO,), solvent was removed on a rotary ...
... was transferred to a separatory funnel containing 75 ml each of water and benzene. The aqueous layer was removed and the benzene solution was washed with water (100 ml), saturated aqueous NaHCO 1 (100 ml), and twice with 100-ml portions of water. After drying (MgSO,), solvent was removed on a rotary ...
carbohydrates: monosaccharides. oligo
... formulas. Aldoses do not give some characteristic aldehyde reactions. Glucose, e.g., under normal conditions does not produce coloration with Schiff’s reagent. At the same time, some reactions (formation of glycosides) can not be explained by oxo forms. Task №2. What is the essence of tautomeric tra ...
... formulas. Aldoses do not give some characteristic aldehyde reactions. Glucose, e.g., under normal conditions does not produce coloration with Schiff’s reagent. At the same time, some reactions (formation of glycosides) can not be explained by oxo forms. Task №2. What is the essence of tautomeric tra ...
13. amines - WordPress.com
... Structure-basicity relationship of amines Basicity of amines is related to their structure. Basic character of an amine depends upon the ease of formation of the cation by accepting a proton from the acid. As the stability of the cation increases, the basicity also increases. a) Comparison of basici ...
... Structure-basicity relationship of amines Basicity of amines is related to their structure. Basic character of an amine depends upon the ease of formation of the cation by accepting a proton from the acid. As the stability of the cation increases, the basicity also increases. a) Comparison of basici ...
Learning materials
... 3. SN1 reaction 4. SN2 reaction 5. E1, E2 reactions 6. Applications of alkyl halides 7. Acidity and basicity of alcohols 8. Dehydration of alcohols 9. Synthesis of ethers 10. Solvents in organic chemistry ...
... 3. SN1 reaction 4. SN2 reaction 5. E1, E2 reactions 6. Applications of alkyl halides 7. Acidity and basicity of alcohols 8. Dehydration of alcohols 9. Synthesis of ethers 10. Solvents in organic chemistry ...
Nucleophilic Substitution on the Carbonyl Group
... example, the —OH group easily replaces the —Cl group. However, a —Cl group does not readily replace an —OH group. Thus, in a reaction between a strong basic nucleophile and a weaker basic leaving group in the acyl halide, the equilibrium favors the product. ...
... example, the —OH group easily replaces the —Cl group. However, a —Cl group does not readily replace an —OH group. Thus, in a reaction between a strong basic nucleophile and a weaker basic leaving group in the acyl halide, the equilibrium favors the product. ...
Ir-catalysed formation of C− F bonds. From allylic alcohols to α
... mixture of 5 : 1. Less water failed to dissolve SelectF, and more water increased by-product (3a) formation. The catalyst loading could be lowered to 1 mol% (Table 2, entry 1). At acidic pH, higher amounts of unwanted ketone 3a are obtained and the reaction is complete in a shorter time (15 min) (Ta ...
... mixture of 5 : 1. Less water failed to dissolve SelectF, and more water increased by-product (3a) formation. The catalyst loading could be lowered to 1 mol% (Table 2, entry 1). At acidic pH, higher amounts of unwanted ketone 3a are obtained and the reaction is complete in a shorter time (15 min) (Ta ...
10. Alkyl Halides - faculty at Chemeketa
... 10.3 Preparing Alkyl Halides from Alkanes: Radical Halogenation Alkane + Cl2 or Br2, heat or light replaces C-H with C- ...
... 10.3 Preparing Alkyl Halides from Alkanes: Radical Halogenation Alkane + Cl2 or Br2, heat or light replaces C-H with C- ...
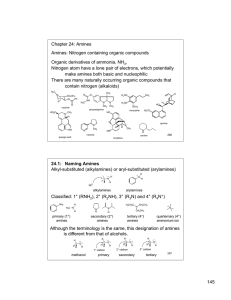
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Table 24.1 (p. 899): pKa values of ammonium ions Alkyl ammonium ions, R3NH+ X-, have pKa values in the range of 10-11 (ammonium ion, H4N+ X-, has a pKa ~ 9.25) The ammonium ions of aryl amines and heterocyclic aromatic amines are considerably less basic than alkyl amines (pKa ~ 5 or less). The nitr ...
... Table 24.1 (p. 899): pKa values of ammonium ions Alkyl ammonium ions, R3NH+ X-, have pKa values in the range of 10-11 (ammonium ion, H4N+ X-, has a pKa ~ 9.25) The ammonium ions of aryl amines and heterocyclic aromatic amines are considerably less basic than alkyl amines (pKa ~ 5 or less). The nitr ...
3.10 aromatic chemistry
... Just as hydroxy is used for the alcohols, OH when a higher priority group is present. Benzene becomes phenyl Common higher priority groups are - NH2 Amine, - OH Alcohols, - C=O aldehydes and ketones, and C=C alkenes: ...
... Just as hydroxy is used for the alcohols, OH when a higher priority group is present. Benzene becomes phenyl Common higher priority groups are - NH2 Amine, - OH Alcohols, - C=O aldehydes and ketones, and C=C alkenes: ...
Chapter 13 – Organic Chemistry
... An alkyl group is formed by removing an H atom from an alkane. They are the side chains (not on the longest continuous chain) of a hydrocarbon. All of the carbon atoms are not in the longest continuous chain of a branched hydrocarbon. Instead, these carbon atoms are in groups called side chains, whi ...
... An alkyl group is formed by removing an H atom from an alkane. They are the side chains (not on the longest continuous chain) of a hydrocarbon. All of the carbon atoms are not in the longest continuous chain of a branched hydrocarbon. Instead, these carbon atoms are in groups called side chains, whi ...
CH - UTH e
... • Awarded Nobel Prize in 1912 Victor Grignard Grignard, Victor , 1871–1935, French chemist. He shared the 1912 Nobel Prize in Chemistry for his work in organic synthesis based on his discovery (1900) of the Grignard Reagent. He taught at the Univ. of Nancy (1909 Grignard Reagent. He taught at the ...
... • Awarded Nobel Prize in 1912 Victor Grignard Grignard, Victor , 1871–1935, French chemist. He shared the 1912 Nobel Prize in Chemistry for his work in organic synthesis based on his discovery (1900) of the Grignard Reagent. He taught at the Univ. of Nancy (1909 Grignard Reagent. He taught at the ...
Rutgers...Ch17 Reactions of Aromatic Compounds
... In the case of toluene, ortho (and para) attack results in the positive charge being spread over two secondary carbons and one tertiary carbon atom (the one bearing the CH3 group). Since the sigma complexes for ortho (and para) attack have resonance forms with tertiary carbons, they are more stable ...
... In the case of toluene, ortho (and para) attack results in the positive charge being spread over two secondary carbons and one tertiary carbon atom (the one bearing the CH3 group). Since the sigma complexes for ortho (and para) attack have resonance forms with tertiary carbons, they are more stable ...
reactions of the conjugated dienes butadiene and isoprene alone
... was reduced; the only product of significance was limonene (Table 3). The use of certain solvents markedly affected the results as well. When isoprene was reacted with methanol in chloroform (b.p. 62~ or heptane (b.p. 97~ under reflux conditions the clay catalyst turned orange immediately after the ...
... was reduced; the only product of significance was limonene (Table 3). The use of certain solvents markedly affected the results as well. When isoprene was reacted with methanol in chloroform (b.p. 62~ or heptane (b.p. 97~ under reflux conditions the clay catalyst turned orange immediately after the ...
Copper(II) bromide as efficient catalyst for silyl
... The bis(methoxyphenyl)methyl (BMPM) ether derived from hexanol was produced in every case investigated, but at different rates. Monitoring the reaction revealed that TES, TPS, and TBS ethers exhibited a higher kinetic interconversion at the beginning of the transformation compared to TIPS and TBDPS ...
... The bis(methoxyphenyl)methyl (BMPM) ether derived from hexanol was produced in every case investigated, but at different rates. Monitoring the reaction revealed that TES, TPS, and TBS ethers exhibited a higher kinetic interconversion at the beginning of the transformation compared to TIPS and TBDPS ...
Chapter 7- Alcohols
... Example of a substituted reaction (OH replaced by X) Good synthetic route to alkyl halides Rate depends on degree of substitution 3o > 2o > 1o ...
... Example of a substituted reaction (OH replaced by X) Good synthetic route to alkyl halides Rate depends on degree of substitution 3o > 2o > 1o ...
Chem 314 Preorganic Evaluation
... allylic & benzylic RX are very reactive if a conjugated pi bond can form complete substitution at Cα (3o RX) shuts down SN2 and makes E2 the only choice, but there maay be many possible E2 products a completely substituted Cβ makes E2 impossible from that position, but if other Cβ's are present with ...
... allylic & benzylic RX are very reactive if a conjugated pi bond can form complete substitution at Cα (3o RX) shuts down SN2 and makes E2 the only choice, but there maay be many possible E2 products a completely substituted Cβ makes E2 impossible from that position, but if other Cβ's are present with ...
organic chemistry reaction scheme
... RH + Mg(OH)X *Note: RMgX is the Grignard reagent, alkylmagnesium halide. The alkyl group is covalently bonded to magnesium; and magnesium-halogen bond is ionic ie. [R:Mg]+[X]–. In the second step of the reaction, it is a displacement reaction in which water (the stronger acid) displacing the weake ...
... RH + Mg(OH)X *Note: RMgX is the Grignard reagent, alkylmagnesium halide. The alkyl group is covalently bonded to magnesium; and magnesium-halogen bond is ionic ie. [R:Mg]+[X]–. In the second step of the reaction, it is a displacement reaction in which water (the stronger acid) displacing the weake ...
Ch. 14 Alcohols, Ethers, & Thiols
... Physical Properties of Alcohols • Because of increase london forces between larger molecules, the B.P. of all types of compounds, including alcohols, increase as molecular weight increases • Alcohols are much more soluble in H2O due to their H-bonding capacity. • As MW increases, the water solubili ...
... Physical Properties of Alcohols • Because of increase london forces between larger molecules, the B.P. of all types of compounds, including alcohols, increase as molecular weight increases • Alcohols are much more soluble in H2O due to their H-bonding capacity. • As MW increases, the water solubili ...
Preparation of Cyclic Urethanes from Amino Alcohols and Carbon
... of amino alcohols with urea can produce cyclic urethanes with good yields [8-10]. However, these reactions will require the recovery of ammonia, if they are employed for the production of cyclic urethane in large scales. This can be an economical disadvantage of them. Under these circumstances, the ...
... of amino alcohols with urea can produce cyclic urethanes with good yields [8-10]. However, these reactions will require the recovery of ammonia, if they are employed for the production of cyclic urethane in large scales. This can be an economical disadvantage of them. Under these circumstances, the ...
Organic Chemistry II Introduction
... ketones using NADH as the source of the equivalent of HThe transfer resembles that in the Cannizzaro reaction but the carbonyl of the acceptor is polarized by an acid from the enzyme, lowering the energy barrier ...
... ketones using NADH as the source of the equivalent of HThe transfer resembles that in the Cannizzaro reaction but the carbonyl of the acceptor is polarized by an acid from the enzyme, lowering the energy barrier ...
Presentation8_108
... alcohols intermolecular hydrogen bonding exists due to which a large amount of energy is required to break these bonds. •Among isomeric alcohols, the boiling point decreases with increase in branching in the alkyl group Boiling points of 1o alcohol < 2o alcohol < 3o alcohol •As number of hydroxyl gr ...
... alcohols intermolecular hydrogen bonding exists due to which a large amount of energy is required to break these bonds. •Among isomeric alcohols, the boiling point decreases with increase in branching in the alkyl group Boiling points of 1o alcohol < 2o alcohol < 3o alcohol •As number of hydroxyl gr ...
Reactions of Alcohols
... The ZnCl2 coordinates to the hydroxyl oxygen, and this generates a far superior leaving group. Primary alcohols react in a similar fashion except the free cation is not generated, and the substitution is of S N2 ...
... The ZnCl2 coordinates to the hydroxyl oxygen, and this generates a far superior leaving group. Primary alcohols react in a similar fashion except the free cation is not generated, and the substitution is of S N2 ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.