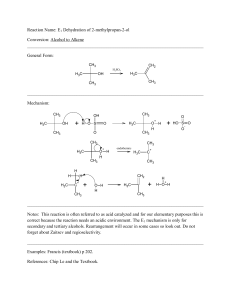
Hydroxyl-Directed Stereoselective Diboration of Alkenes
... providing the product in 87% conversion and 12.5:1 diastereomer ratio favoring the syn isomer of product. Reasoning that the substrate alkoxide likely participates in reversible acid−base equilibrium with methanol, a catalytic amount of NaOtBu in the presence of methanol was examined and found to pr ...
... providing the product in 87% conversion and 12.5:1 diastereomer ratio favoring the syn isomer of product. Reasoning that the substrate alkoxide likely participates in reversible acid−base equilibrium with methanol, a catalytic amount of NaOtBu in the presence of methanol was examined and found to pr ...
RELATIONSHIP BETWEEN Sn1 and E1 REACTIONS
... by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat of hydrogenation, the lower the stability, because the higher the heat of hydrogenation, the higher the potential ...
... by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat of hydrogenation, the lower the stability, because the higher the heat of hydrogenation, the higher the potential ...
CBSEGuess.com
... reacted With HBr to give (c) which is an isomer of (a). when (a) is reacted with Na metal it give (d), C818 which is different from the compound formed when n-butyl bromide is reacted with Na metal . Give the structural formula of (a) and write the equations. ...
... reacted With HBr to give (c) which is an isomer of (a). when (a) is reacted with Na metal it give (d), C818 which is different from the compound formed when n-butyl bromide is reacted with Na metal . Give the structural formula of (a) and write the equations. ...
Chapter 22 HEIN
... remove water. Therefore the double bond can be located in different positions. • The major product in such cases is the alkene in which the C=C bond has the greatest number of alkyl substituents on it (or the least number of hydrogens). H H3C ...
... remove water. Therefore the double bond can be located in different positions. • The major product in such cases is the alkene in which the C=C bond has the greatest number of alkyl substituents on it (or the least number of hydrogens). H H3C ...
File - Rasapalli Research Group
... Can be accomplished by inorganic reagents, such as KMnO4, CrO3, and Na2Cr2O7 or by more selective, expensive reagents ...
... Can be accomplished by inorganic reagents, such as KMnO4, CrO3, and Na2Cr2O7 or by more selective, expensive reagents ...
GRIGNARD REAGENTS
... 7 (b) It is also possible to draw a dipolar resonance form of a carbonyl group. ...
... 7 (b) It is also possible to draw a dipolar resonance form of a carbonyl group. ...
992_4th_ Exam_1000615
... 7) Which of the following statements concerning alpha decay is INCORRECT? A) It involves nuclides with atomic number larger than 83 and mass number larger than 200. B) It has great penetrating power, but little ionizing power. C) It often leaves the nucleus in an excited state. D) The alpha particl ...
... 7) Which of the following statements concerning alpha decay is INCORRECT? A) It involves nuclides with atomic number larger than 83 and mass number larger than 200. B) It has great penetrating power, but little ionizing power. C) It often leaves the nucleus in an excited state. D) The alpha particl ...
Orbitals
... • If neutral, the nucleophile usually carries a hydrogen atom that can be subsequently eliminated ...
... • If neutral, the nucleophile usually carries a hydrogen atom that can be subsequently eliminated ...
CHEMICAL REACTIVITY AND MECHANISMS, AND SUBSTITUTION REACTIONS 1.
... steric effect; larger groups interfere with the approaching nucleophile). SN1 reactions are faster for 3° substrates (because the more stable the carbocation, the faster the reaction; this means 3° > 2° >> 1° > CH3). Vinylic (R2C=CR-) and aromatic substrates are unreactive in either reaction type. A ...
... steric effect; larger groups interfere with the approaching nucleophile). SN1 reactions are faster for 3° substrates (because the more stable the carbocation, the faster the reaction; this means 3° > 2° >> 1° > CH3). Vinylic (R2C=CR-) and aromatic substrates are unreactive in either reaction type. A ...
Nuggets of Knowledge for Chapter 12 – Alcohols
... • The following reactions that we have previously learned may be used to synthesize alcohols. Make sure to review these reactions, as you will be responsible for them in this chapter. o SN2 reactions between primary alkyl halides and sodium hydroxide (Ch 8) o Acid-catalyzed hydration of alkenes, acc ...
... • The following reactions that we have previously learned may be used to synthesize alcohols. Make sure to review these reactions, as you will be responsible for them in this chapter. o SN2 reactions between primary alkyl halides and sodium hydroxide (Ch 8) o Acid-catalyzed hydration of alkenes, acc ...
Amines and amides
... The presence of the OH-(aq) ion means that aqueous solutions of amines are ALKALINE. Like NH3 amines react with acids, H3O+ ion is more effective proton donor than H2O, therefore reaction goes to completion therefore amines ...
... The presence of the OH-(aq) ion means that aqueous solutions of amines are ALKALINE. Like NH3 amines react with acids, H3O+ ion is more effective proton donor than H2O, therefore reaction goes to completion therefore amines ...
Organic Chemistry Durham School Board March
... Dispersion forces increase as the number of carbons increases. Boiling point increases as the number of carbon chains increases in the molecule. Have low density; most are less dense than water. Immiscible (not soluble) with water. ...
... Dispersion forces increase as the number of carbons increases. Boiling point increases as the number of carbon chains increases in the molecule. Have low density; most are less dense than water. Immiscible (not soluble) with water. ...
Alcohols and Phenols
... Alcohols contain an OH group connected to a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to a carbon in a benzene ...
... Alcohols contain an OH group connected to a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to a carbon in a benzene ...
Exam 3 - Napa Valley College
... smaller nuc- and larger substrate in #1 the reaction would go SN1 which would mean that you would get a lot of by-products but you would end up getting more product also (SN1 major, E1 minor). 4) There are a number of ways of substituting a halogen for an alcohol group, but some ways are better than ...
... smaller nuc- and larger substrate in #1 the reaction would go SN1 which would mean that you would get a lot of by-products but you would end up getting more product also (SN1 major, E1 minor). 4) There are a number of ways of substituting a halogen for an alcohol group, but some ways are better than ...
Aldehydes, Ketones and Carboxylic acids
... Aldehydes and ketones undergo nucleophilic substitution reaction. In nucleophilic substitution, a species that has extra electron or lone pair of electron attacks the carbonyl carbon of the ketone or the aldehyde leading to an alkoxide intermediate as shown in the middle figure below. This alkoxide ...
... Aldehydes and ketones undergo nucleophilic substitution reaction. In nucleophilic substitution, a species that has extra electron or lone pair of electron attacks the carbonyl carbon of the ketone or the aldehyde leading to an alkoxide intermediate as shown in the middle figure below. This alkoxide ...
C - Deans Community High School
... Ethanol is formed industrially from ethene. The reaction conditions are 300oC , high pressure of 60 atmospheres and phosphoric acid catalyst. Correct because ……………….. ...
... Ethanol is formed industrially from ethene. The reaction conditions are 300oC , high pressure of 60 atmospheres and phosphoric acid catalyst. Correct because ……………….. ...
Alcohols and Phenols
... not), forming soluble salts that are soluble in dilute aqueous A phenolic component can be separated from an organic solution by extraction into basic aqueous solution and is isolated after acid is added to the solution ...
... not), forming soluble salts that are soluble in dilute aqueous A phenolic component can be separated from an organic solution by extraction into basic aqueous solution and is isolated after acid is added to the solution ...
Overview of the Reactions of Carbonyl Compounds
... to a carbonyl compound. Each way leads to a different nucleophilic addition reaction but the mechanisms for both reactions involves the same 1st step. • In this step, the nucleophile bonds to the carbonyl carbon and thereby causes a carbon-oxygen bond to break. The carbonyl carbon rehybridizes from ...
... to a carbonyl compound. Each way leads to a different nucleophilic addition reaction but the mechanisms for both reactions involves the same 1st step. • In this step, the nucleophile bonds to the carbonyl carbon and thereby causes a carbon-oxygen bond to break. The carbonyl carbon rehybridizes from ...
CHAPTER 17: Carbonyl group (1)
... An alternative method is the PCC oxidation (CrO3, HCl, pyridine) that oxidizes primary and secondary alcohols only to the corresponding carbonyl groups (no overoxidation to acids). The PCC oxidation is performed in organic solvents, such as CH2Cl2. In the absense of H2O the primary aldehyde cannot b ...
... An alternative method is the PCC oxidation (CrO3, HCl, pyridine) that oxidizes primary and secondary alcohols only to the corresponding carbonyl groups (no overoxidation to acids). The PCC oxidation is performed in organic solvents, such as CH2Cl2. In the absense of H2O the primary aldehyde cannot b ...
REASONING QUESTIONS IN ORGANIC CHEMISTRY
... Ans: Chloroform react with air in presence of sun light it forms poisonous phosogene. 2 CHCl3 + O2 2 COCl2+ 2 HCl 11. Out of C6H5CH2Cl & C6 H5CHCl C6H5 Which is more easily hydrolyzed by KOH? Ans; The second comound is more resonance stabilized hence easily forms carbo cation and react faster by S ...
... Ans: Chloroform react with air in presence of sun light it forms poisonous phosogene. 2 CHCl3 + O2 2 COCl2+ 2 HCl 11. Out of C6H5CH2Cl & C6 H5CHCl C6H5 Which is more easily hydrolyzed by KOH? Ans; The second comound is more resonance stabilized hence easily forms carbo cation and react faster by S ...
MHS Student Guide to Organic Chemistry
... Organic nomenclature part (1) Chemicals compounds that contain the element Carbon are known as organic compounds. “Organic” comes from the fact that until the mid 1800’s it was thought that these chemicals could only be derived from living plant or animal components. In 1828 Friedrich Woher convert ...
... Organic nomenclature part (1) Chemicals compounds that contain the element Carbon are known as organic compounds. “Organic” comes from the fact that until the mid 1800’s it was thought that these chemicals could only be derived from living plant or animal components. In 1828 Friedrich Woher convert ...
Reactions of 2, 6-cycloheptadienone and 2, 7
... The LiClO4 served only as a source of HC104 and could be replaced by any inorganic perchlorate which would dissolve sufficiently in H z S O ~to liberate the required perchloric acid. Alternatively 70% HClO4 could be added directly to 967' HzSOd. If a large amount of perchloric acid was used, it was ...
... The LiClO4 served only as a source of HC104 and could be replaced by any inorganic perchlorate which would dissolve sufficiently in H z S O ~to liberate the required perchloric acid. Alternatively 70% HClO4 could be added directly to 967' HzSOd. If a large amount of perchloric acid was used, it was ...
CHEM120 - ORGANIC CHEMISTRY WORKSHEET 1
... Be able to determine which of two groups is the better base and be able to predict whether a substitution reaction will occur or not. If a reaction can occur, you must be able to predict the product. ...
... Be able to determine which of two groups is the better base and be able to predict whether a substitution reaction will occur or not. If a reaction can occur, you must be able to predict the product. ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.