Chemistry at Karlsruhe 1860
... • Like the Berzelians they dealt only with empirical data and equivalent weights • Organic Compounds generally are made up of carbon, hydrogen and oxygen • Organic compounds when combusted in air form water, and carbon dioxide • Liebig determined the simplest formulas (empirical formulas) of any com ...
... • Like the Berzelians they dealt only with empirical data and equivalent weights • Organic Compounds generally are made up of carbon, hydrogen and oxygen • Organic compounds when combusted in air form water, and carbon dioxide • Liebig determined the simplest formulas (empirical formulas) of any com ...
A millennial overview of transition metal chemistry
... It is, perhaps, a moot point whether this field should be included in the list of evolutionary developments. Certain types of organo compounds seem not entirely discontinuous from the Werner complexes. Zeise’s anion is an example, provided we recognize that electrons can be donated (and back-accepted ...
... It is, perhaps, a moot point whether this field should be included in the list of evolutionary developments. Certain types of organo compounds seem not entirely discontinuous from the Werner complexes. Zeise’s anion is an example, provided we recognize that electrons can be donated (and back-accepted ...
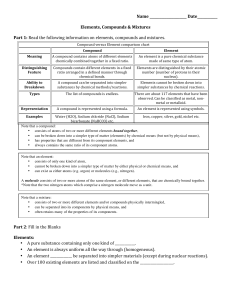
Document
... Percent, or parts or solute per 100 parts of solvent Molarity, or moles per liter (M) A mole of an element or compound is equal to its atomic or molecular weight (sum of atomic weights) in grams One mole of any substance contains exactly the same number of solute particles (6.02 x 1023) 37. Colloids ...
... Percent, or parts or solute per 100 parts of solvent Molarity, or moles per liter (M) A mole of an element or compound is equal to its atomic or molecular weight (sum of atomic weights) in grams One mole of any substance contains exactly the same number of solute particles (6.02 x 1023) 37. Colloids ...
General Chemistry - Review for final exam: (Make sure you bring
... d. Ionization energy e. 2nd ionization energy 41. What are cations and anions? Which type of elements form each? 42. Write the ions formed by the following: State how many electrons were gained or lost. a. F b. Ca c. H d. S e. Al 43. What is the octet rule? 44. What are some basic properties of ioni ...
... d. Ionization energy e. 2nd ionization energy 41. What are cations and anions? Which type of elements form each? 42. Write the ions formed by the following: State how many electrons were gained or lost. a. F b. Ca c. H d. S e. Al 43. What is the octet rule? 44. What are some basic properties of ioni ...
Chemistry for Bio 11
... electrons in “planetary” orbitals • Each orbit holds a determined number of electrons (first holds two, 2nd and 3rd hold eight ...
... electrons in “planetary” orbitals • Each orbit holds a determined number of electrons (first holds two, 2nd and 3rd hold eight ...
Chapter 10 The Periodic Law
... 10-16. Naming Compounds •A compound ending in -ide usually is composed of only two elements. Hydroxides which contain the OH- ion are an exception. Sodium Chloride=NaCl •A compound ending in -ate contains oxygen and two or more other elements. Calcium Sulfate=CaSO4 ...
... 10-16. Naming Compounds •A compound ending in -ide usually is composed of only two elements. Hydroxides which contain the OH- ion are an exception. Sodium Chloride=NaCl •A compound ending in -ate contains oxygen and two or more other elements. Calcium Sulfate=CaSO4 ...
Units 3 and 4 Revision
... These elements have the same number of electrons in their outer energy level – this gives them similar properties. Q10. Chlorine atoms exists as two different isotopes 35Cl and 37Cl. ...
... These elements have the same number of electrons in their outer energy level – this gives them similar properties. Q10. Chlorine atoms exists as two different isotopes 35Cl and 37Cl. ...
Atoms, compounds and elements - Mrs. Tes de Luna`s Science Class
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
Branches of Chemistry
... Inorganic chemists study the chemistry of all the elements and their compounds, except for those compounds that contain mainly carbon and hydrogen. Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other re ...
... Inorganic chemists study the chemistry of all the elements and their compounds, except for those compounds that contain mainly carbon and hydrogen. Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other re ...
Inorganometallic Chemistry
... direct ionic or covalent localized or delocalized bond between one or more carbon atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded t ...
... direct ionic or covalent localized or delocalized bond between one or more carbon atoms of an organic group or molecule and the atom from the main group, transition, lanthanide or actinide metals. According to the IUPAC rules, organometallic compounds are those in which the carbon atoms are bonded t ...
ap chemistry – 2013-2014
... 7. Understand energy changes, thermodynamics, physical and chemical behavior of gases. 8. Be able to use and understand the Periodic Table, predict general patterns from the table, and the electronic structure of atoms. 9. Build an understanding of ionic, covalent, network covalent, and intermolecul ...
... 7. Understand energy changes, thermodynamics, physical and chemical behavior of gases. 8. Be able to use and understand the Periodic Table, predict general patterns from the table, and the electronic structure of atoms. 9. Build an understanding of ionic, covalent, network covalent, and intermolecul ...
Chemistry Semester One Exam Review Name:
... d) Double replacement e) Single replacement 17. Complete the word equation, write and balance the equation using symbols and indicate the type of the reaction on the left. a. Propane (C3H8) burns in air b. Magnesium chloride + silver nitrate c. Zn reacts with hydrochloric acid d. Nitrogen gas ...
... d) Double replacement e) Single replacement 17. Complete the word equation, write and balance the equation using symbols and indicate the type of the reaction on the left. a. Propane (C3H8) burns in air b. Magnesium chloride + silver nitrate c. Zn reacts with hydrochloric acid d. Nitrogen gas ...
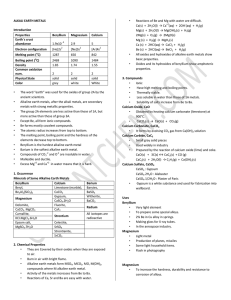
ALKALI EARTH METALS Introduction Properties Beryllium
... Ca (s) + 2HCI(aq) → CaCl2 + H2(g) Be (s) + 2HCI(aq) → BeCI2 + H2(g) All oxides and hydroxides of alkaline earth metals show basic properties. Oxides and its hydroxides of beryllium show amphoteric properties. ...
... Ca (s) + 2HCI(aq) → CaCl2 + H2(g) Be (s) + 2HCI(aq) → BeCI2 + H2(g) All oxides and hydroxides of alkaline earth metals show basic properties. Oxides and its hydroxides of beryllium show amphoteric properties. ...
chapter 12_LO - Faculty Websites
... What is the difference between organic compounds and inorganic compounds? Why are there so many more organic compounds than inorganic compounds? You should be able to recognize and describe the different properties of organic compounds. Which elements are typically found in organic compounds? What i ...
... What is the difference between organic compounds and inorganic compounds? Why are there so many more organic compounds than inorganic compounds? You should be able to recognize and describe the different properties of organic compounds. Which elements are typically found in organic compounds? What i ...
CHM 2045C - State College of Florida
... Investigate the thermodynamics of chemical processes using current terminology and mathematical relationships which include heat capacity and state functions. ...
... Investigate the thermodynamics of chemical processes using current terminology and mathematical relationships which include heat capacity and state functions. ...
Fundamentals of Chemistry
... • The Central Science – The study of matter – Explains every aspect of daily life – Is essential to understand nearly all scientific, medical and pharmaceutical disciplines, and ...
... • The Central Science – The study of matter – Explains every aspect of daily life – Is essential to understand nearly all scientific, medical and pharmaceutical disciplines, and ...
AP Chemistry Summer Assignment
... d. __ Fe2(SO4)3 + __ HCl __ FeCl3+ __ H2O + __ SO3 e. __ C12H24 + __ O2 __ CO2 + __ H2O f. __ Al + __O2 __ Al2O3 g. __ C9H16 + __ O2 __ CO2 + __ H2O h. __ Cr(SO3)2 + __ H2 __ SO2 + __ Cr + __ H2O i. __ C4H8O4 + __ O2 __ CO2 + __ H2O. 41. Define limiting reagent, theoretical yield, and ac ...
... d. __ Fe2(SO4)3 + __ HCl __ FeCl3+ __ H2O + __ SO3 e. __ C12H24 + __ O2 __ CO2 + __ H2O f. __ Al + __O2 __ Al2O3 g. __ C9H16 + __ O2 __ CO2 + __ H2O h. __ Cr(SO3)2 + __ H2 __ SO2 + __ Cr + __ H2O i. __ C4H8O4 + __ O2 __ CO2 + __ H2O. 41. Define limiting reagent, theoretical yield, and ac ...
Topic 3&4 Atoms and the per.table
... These elements have the same number of electrons in their outer energy level – this gives them similar properties. Q10. Chlorine atoms exists as two different isotopes 35Cl and 37Cl. ...
... These elements have the same number of electrons in their outer energy level – this gives them similar properties. Q10. Chlorine atoms exists as two different isotopes 35Cl and 37Cl. ...
Compound Name
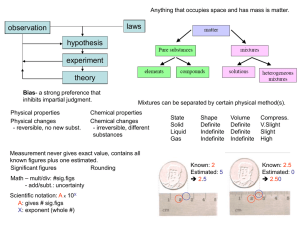
... What’s the difference between a physical and chemical change? Physical changes: examples – change of state, dissolving; Chemical changes (new substance formed): heat or light given off, ...
... What’s the difference between a physical and chemical change? Physical changes: examples – change of state, dissolving; Chemical changes (new substance formed): heat or light given off, ...
Inorganic chemistry

Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds), which are the subjects of organic chemistry. The distinction between the two disciplines is far from absolute, and there is much overlap, most importantly in the sub-discipline of organometallic chemistry. It has applications in every aspect of the chemical industry–including catalysis, materials science, pigments, surfactants, coatings, medicine, fuel, and agriculture.