Atoms, molecules and ions
... • For a binary covalent compound, we name the first element in the formula first – ex.; HCl : hydrogen chloride NO : nitric oxide SiC : silicon carbide ...
... • For a binary covalent compound, we name the first element in the formula first – ex.; HCl : hydrogen chloride NO : nitric oxide SiC : silicon carbide ...
1.5.16(Chem) - mrcarlsonschemistryclass
... • Draw the funny way to remember cations and anions: ...
... • Draw the funny way to remember cations and anions: ...
Packet
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
Unit A Remediation Review
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
Organometallic Compounds and Catalysis: Synthesis
... Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industrial chemistry. A catalyst is defined as a substance that accelerates the rate of achieving chemical equilibrium, and which can be recovered unchanged at the end of a reaction ...
... Research interest in this area is largely fueled by potential applications of organometallic compounds as catalysts in industrial chemistry. A catalyst is defined as a substance that accelerates the rate of achieving chemical equilibrium, and which can be recovered unchanged at the end of a reaction ...
Welcome to AP Chemistry!
... 5. Oxygen has an oxidation number of –2 unless it is combined with F, when it is +2, or it is in a peroxide, when it is –1. 6. The oxidation state of hydrogen in most of its compounds is+1 unless it combined with a metal, in which case it is –1. 7. In compounds, the elements of groups 1 and 2 as wel ...
... 5. Oxygen has an oxidation number of –2 unless it is combined with F, when it is +2, or it is in a peroxide, when it is –1. 6. The oxidation state of hydrogen in most of its compounds is+1 unless it combined with a metal, in which case it is –1. 7. In compounds, the elements of groups 1 and 2 as wel ...
2. Chemistry of Living Things Outline
... Equations are used to describe ____________________________. The substances that start the reaction are called the _____________. o The reactants are placed on the _______ side of the equation. The substances formed by the reaction are called the ____________. o The products are placed on the ______ ...
... Equations are used to describe ____________________________. The substances that start the reaction are called the _____________. o The reactants are placed on the _______ side of the equation. The substances formed by the reaction are called the ____________. o The products are placed on the ______ ...
Chemistry of Living Things Outline
... Equations are used to describe ____________________________. The substances that start the reaction are called the _____________. o The reactants are placed on the _______ side of the equation. The substances formed by the reaction are called the ____________. o The products are placed on the ...
... Equations are used to describe ____________________________. The substances that start the reaction are called the _____________. o The reactants are placed on the _______ side of the equation. The substances formed by the reaction are called the ____________. o The products are placed on the ...
Reactive Materials - NC State University
... Reactive liquids are chemicals that react vigorously with moisture or oxygen or other substances. Reactive solids are chemicals that react vigorously with moisture and other substances. The most common reactive solids include sodium, potassium and lithium metals, acid anhydrides and acid chlorides. ...
... Reactive liquids are chemicals that react vigorously with moisture or oxygen or other substances. Reactive solids are chemicals that react vigorously with moisture and other substances. The most common reactive solids include sodium, potassium and lithium metals, acid anhydrides and acid chlorides. ...
30.09.2013 1 Chapter 2 Atoms and Molecules Warning!! Chapter
... • Compounds have different properties than their constituent atoms. • Ionic compounds contain cations and anions, usually arranged in a lattice. • Molecular formulas indicate the elements and number of atoms of each element actually contained in a discrete unit of a compound. • Empirical formulas in ...
... • Compounds have different properties than their constituent atoms. • Ionic compounds contain cations and anions, usually arranged in a lattice. • Molecular formulas indicate the elements and number of atoms of each element actually contained in a discrete unit of a compound. • Empirical formulas in ...
Review Packet
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
Packet
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
... a. include changes of state of a substance b. include mass and color c. include changes that alter the identity of a substance d. can be observed without altering the identity of a substance 34. Identify each as an element, compound, or mixture. For mixtures, identify it as homogeneous or heterogene ...
Biologically Important Inorganic Elements Occurrence and Availability
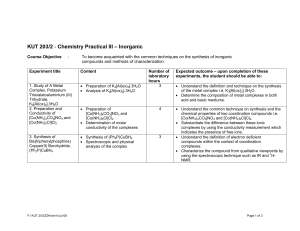
... Course Outline and Objectives Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. In CHEM 646, we will apply fundamental principles of inorganic chemistry, particularly transition metal coordination chemistry and ligand field theory, to understand t ...
... Course Outline and Objectives Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. In CHEM 646, we will apply fundamental principles of inorganic chemistry, particularly transition metal coordination chemistry and ligand field theory, to understand t ...
Bonding in Atoms
... • A chemical formula shows the kinds of atoms in the molecule as well as the amount of atoms in the molecule • A formula unit is expressed in the smallest whole number ratio for the molecule ...
... • A chemical formula shows the kinds of atoms in the molecule as well as the amount of atoms in the molecule • A formula unit is expressed in the smallest whole number ratio for the molecule ...
Naming Ionic Compounds
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
Inorganic chemistry

Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds), which are the subjects of organic chemistry. The distinction between the two disciplines is far from absolute, and there is much overlap, most importantly in the sub-discipline of organometallic chemistry. It has applications in every aspect of the chemical industry–including catalysis, materials science, pigments, surfactants, coatings, medicine, fuel, and agriculture.