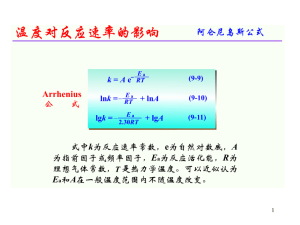
Unit 1 - Measurement Atomic Theory
... Liquid – measure using instrument (cylinder, beaker, buret, flask, etc.) ...
... Liquid – measure using instrument (cylinder, beaker, buret, flask, etc.) ...
Organic Chemistry
... • Carbon forms a variety of strong covalent bonds (共价键) to itself and other atoms. • This allows organic compounds to be structurally diverse. ...
... • Carbon forms a variety of strong covalent bonds (共价键) to itself and other atoms. • This allows organic compounds to be structurally diverse. ...
Student Worksheet The Chemistry of Water Quality Tests
... AP Chemistry: Enduring understanding 3.A: Chemical changes are represented by a balanced chemical equation that identifies the ratios with which reactants react and products form. Essential knowledge 3.A.1: A chemical change may be represented by a molecular, ionic, or net ionic equation. Enduring u ...
... AP Chemistry: Enduring understanding 3.A: Chemical changes are represented by a balanced chemical equation that identifies the ratios with which reactants react and products form. Essential knowledge 3.A.1: A chemical change may be represented by a molecular, ionic, or net ionic equation. Enduring u ...
Sample % Sulfate Absolute Deviation A 44.02 B 44.11 C 43.98 D
... In the late 1700s, French and English scientists measured how the mass of products of chemical reaction related to the masses of the reactants, with special interest in reactions by which different elements are reacted together to form compounds, or by which compounds are decomposed (usually by heat ...
... In the late 1700s, French and English scientists measured how the mass of products of chemical reaction related to the masses of the reactants, with special interest in reactions by which different elements are reacted together to form compounds, or by which compounds are decomposed (usually by heat ...
Chapter One Outline
... Physical properties can be observed and measured without changing the composition of a substance. Examples include temperature, mass, density, etc. Density is the ratio of an objects mass to its volume; D = m/v Chemical Properties A substances chemical properties describe the kinds of chemical react ...
... Physical properties can be observed and measured without changing the composition of a substance. Examples include temperature, mass, density, etc. Density is the ratio of an objects mass to its volume; D = m/v Chemical Properties A substances chemical properties describe the kinds of chemical react ...
Chemistry is a material science
... Chemical changes are chemical __________ and are more difficult to ________ than physical changes. In a chemical reaction new _________ are formed with a new and different set of physical and chemical properties. For example, when H2 and O2 react to ...
... Chemical changes are chemical __________ and are more difficult to ________ than physical changes. In a chemical reaction new _________ are formed with a new and different set of physical and chemical properties. For example, when H2 and O2 react to ...
Efficiently Studying Organic Chemistry
... chemistry, nor a vast amount of usually less important information. It was written to accompany the preparation for an examination in organic chemistry (covering basic courses as given in a bachelor education). It contains (more or less) everything one needs to know in an examination and not much mo ...
... chemistry, nor a vast amount of usually less important information. It was written to accompany the preparation for an examination in organic chemistry (covering basic courses as given in a bachelor education). It contains (more or less) everything one needs to know in an examination and not much mo ...
The structure of Matter
... O The electron may be more strongly attracted to one of the atoms and spend more time revolving around that nucleus instead of the other. O This results in a positive and negative end for the compound (the end with the electron most of the time is negative…). O This is called a polar bond. ...
... O The electron may be more strongly attracted to one of the atoms and spend more time revolving around that nucleus instead of the other. O This results in a positive and negative end for the compound (the end with the electron most of the time is negative…). O This is called a polar bond. ...
Organic compounds are covalent compounds composed of carbon
... composed of carbonbased molecules. More than 90% of all compounds belong to this group. ...
... composed of carbonbased molecules. More than 90% of all compounds belong to this group. ...
Exam 2
... 6. In the reduction of 2-butanone to (2)-butanol using the (S)-CBS reagent (2-methyloxazaborolidine + BH3), what is transferred in the critical step in the reaction mechanism? a) a hydride ion, H- b) a hydrogen radical, H c) a proton, H+ d) both hydrogens simultaneously as molecular hydrogen, H2 e) ...
... 6. In the reduction of 2-butanone to (2)-butanol using the (S)-CBS reagent (2-methyloxazaborolidine + BH3), what is transferred in the critical step in the reaction mechanism? a) a hydride ion, H- b) a hydrogen radical, H c) a proton, H+ d) both hydrogens simultaneously as molecular hydrogen, H2 e) ...
Stoichiometry
... Here there will be change in the oxidation state of ions during the reaction. One element will be oxidized; that means that it will lose electrons and become more positive. One element will be reduced; that means that it will gain electrons and become more negative. A balanced equation may r ...
... Here there will be change in the oxidation state of ions during the reaction. One element will be oxidized; that means that it will lose electrons and become more positive. One element will be reduced; that means that it will gain electrons and become more negative. A balanced equation may r ...
Chapter 4: Chemical Reactions Elements can be characterized as
... Zn(s) + CuSO4(aq) -> ZnSO4(aq) + Cu(s) Active metal + non-oxidizing acid -> Hydrogen + salt of Acid Zn(s) + H2SO4 Metathesis Reactions – a reaction in which the positive ion and the negative ion change partners AX + BY -> AY + BX or AgNO3(aq) + NaCl -> AgCl(s) + NaNO3 (AgCl precipitates) Metathesis ...
... Zn(s) + CuSO4(aq) -> ZnSO4(aq) + Cu(s) Active metal + non-oxidizing acid -> Hydrogen + salt of Acid Zn(s) + H2SO4 Metathesis Reactions – a reaction in which the positive ion and the negative ion change partners AX + BY -> AY + BX or AgNO3(aq) + NaCl -> AgCl(s) + NaNO3 (AgCl precipitates) Metathesis ...
Lecture 7
... Going down the group the cations formed get bigger. This means that the positive charge is spread over a larger volume and so is less concentrated. A small ion with its concentrated charge has a high charge density. This high charge density gives the ion the ability to distort or polarize nearby ani ...
... Going down the group the cations formed get bigger. This means that the positive charge is spread over a larger volume and so is less concentrated. A small ion with its concentrated charge has a high charge density. This high charge density gives the ion the ability to distort or polarize nearby ani ...
Basic Chemistry notes
... Molecules and Compounds ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
... Molecules and Compounds ______________________—two or more like atoms combined chemically ______________________—two or more different atoms combined chemically ...
Chapter 6: Chemistry in Biology
... Substances that release hydrogen ions ( H ) when dissolved in water are called __________. Substances that release hydroxide ions ( OH ) when dissolved in water are called __________. pH and Buffers: The measure of concentration of H in a solution is called __________. ...
... Substances that release hydrogen ions ( H ) when dissolved in water are called __________. Substances that release hydroxide ions ( OH ) when dissolved in water are called __________. pH and Buffers: The measure of concentration of H in a solution is called __________. ...
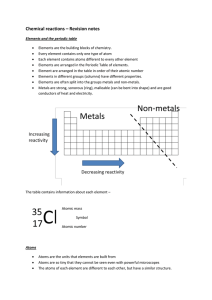
Chemical reactions revision
... Elements in different groups (columns) have different properties. Elements are often split into the groups metals and non-metals. Metals are strong, sonorous (ring), malleable (can be bent into shape) and are good conductors of heat and electricity. ...
... Elements in different groups (columns) have different properties. Elements are often split into the groups metals and non-metals. Metals are strong, sonorous (ring), malleable (can be bent into shape) and are good conductors of heat and electricity. ...
Chemistry
... Exothermic and endothermic processes; the Joule as a unit of energy. Thermochemical equations and the ∆H notation. Energy changes accompanying neutralisation, solution, combustion and atomisation reactions. Experimental determination of energy changes not required. Calorific value of fuels and food; ...
... Exothermic and endothermic processes; the Joule as a unit of energy. Thermochemical equations and the ∆H notation. Energy changes accompanying neutralisation, solution, combustion and atomisation reactions. Experimental determination of energy changes not required. Calorific value of fuels and food; ...
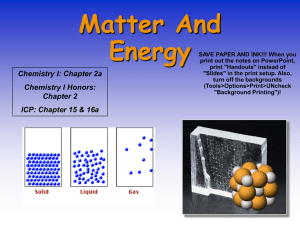
Matter and Energy
... 6. Which of the following are types of matter? (1) elements only (2) compounds only (3) mixtures only (4) all of these 7. Which of the following is a type of mixture? (1) elements only(2) compounds only (3) solutions only (4) elements and Compounds 8. Which of the following is NOT composed of two or ...
... 6. Which of the following are types of matter? (1) elements only (2) compounds only (3) mixtures only (4) all of these 7. Which of the following is a type of mixture? (1) elements only(2) compounds only (3) solutions only (4) elements and Compounds 8. Which of the following is NOT composed of two or ...
AP Chemistry Syllabus
... either would like to earn college credit (by AP examination) or would like to prepare for college chemistry while in high school. This is accomplished through an intensive, in-depth approach. It is highly recommended that the student take the College Board's Advance Placement test in Chemistry. The ...
... either would like to earn college credit (by AP examination) or would like to prepare for college chemistry while in high school. This is accomplished through an intensive, in-depth approach. It is highly recommended that the student take the College Board's Advance Placement test in Chemistry. The ...
CHEMISTRY IM 06 SYLLABUS 1
... Exothermic and endothermic processes; the Joule as a unit of energy. Thermochemical equations and the H notation. Energy changes accompanying neutralisation, solution, combustion and atomisation reactions. Experimental determination of energy changes not required. Calorific value of fuels and food; ...
... Exothermic and endothermic processes; the Joule as a unit of energy. Thermochemical equations and the H notation. Energy changes accompanying neutralisation, solution, combustion and atomisation reactions. Experimental determination of energy changes not required. Calorific value of fuels and food; ...
Inorganic chemistry

Inorganic chemistry deals with the synthesis and behavior of inorganic and organometallic compounds. This field covers all chemical compounds except the myriad organic compounds (carbon based compounds, usually containing C-H bonds), which are the subjects of organic chemistry. The distinction between the two disciplines is far from absolute, and there is much overlap, most importantly in the sub-discipline of organometallic chemistry. It has applications in every aspect of the chemical industry–including catalysis, materials science, pigments, surfactants, coatings, medicine, fuel, and agriculture.