ATOMS AND ELEMENTS
... A. Electrons travel around the nucleus in the electron cloud. B. Electrons follow paths called energy levels or energy shells. C. All elements have at least 1 energy level. D. The period number (or the rows) on the Periodic Table tells you the number of occupied energy shells that element has. E. El ...
... A. Electrons travel around the nucleus in the electron cloud. B. Electrons follow paths called energy levels or energy shells. C. All elements have at least 1 energy level. D. The period number (or the rows) on the Periodic Table tells you the number of occupied energy shells that element has. E. El ...
Study Guide Matter: Building Blocks of the Universe
... have 7 valence electrons are active nonmetals usually combined w/ other elements * Know that there is a difference between fission and fusion: fusion- put atoms together with enormous amounts of energy released fission- splitting atoms- energy released- not as much as fusion- may occur in a chain re ...
... have 7 valence electrons are active nonmetals usually combined w/ other elements * Know that there is a difference between fission and fusion: fusion- put atoms together with enormous amounts of energy released fission- splitting atoms- energy released- not as much as fusion- may occur in a chain re ...
Chapter 4.1
... 1. Protons – positive particles in the nucleus -charge is +1 -# protons = atomic # 2. Electrons – negative particles on orbits around the nucleus -charge is -1 -# electons = # protons= atomic # 3. Neutrons – neutral particles in the nucleus -charge is 0 -#neutrons= mass-atomic # ...
... 1. Protons – positive particles in the nucleus -charge is +1 -# protons = atomic # 2. Electrons – negative particles on orbits around the nucleus -charge is -1 -# electons = # protons= atomic # 3. Neutrons – neutral particles in the nucleus -charge is 0 -#neutrons= mass-atomic # ...
Regents Chemistry Review
... together in the 12 o’clock position, continue placing electrons in the remaining positions (3, 6 & 9 o’clock), one at a time, until you have two in each; the max is an OCTET. ...
... together in the 12 o’clock position, continue placing electrons in the remaining positions (3, 6 & 9 o’clock), one at a time, until you have two in each; the max is an OCTET. ...
The topic that fascinated me the most in my Science lessons this
... The topic that fascinated me the most in my Science lessons this year is the Periodic Table and its power of predicting the existence and properties of elements yet to be discovered. Dimitri Mendeleev placed the 65 known elements of his time into a grid table and observed gaps in the table. Based on ...
... The topic that fascinated me the most in my Science lessons this year is the Periodic Table and its power of predicting the existence and properties of elements yet to be discovered. Dimitri Mendeleev placed the 65 known elements of his time into a grid table and observed gaps in the table. Based on ...
8th Grade Science Notes Chapter 2
... Niels Bohr - proposed an atomic model that placed electrons in circular orbits called energy levels. Electron Cloud - the modern atomic model. Electrons move in an area represented as a cloud around the nucleus. Quarks - smaller particles that make up protons and neutrons. There are six: up, down, c ...
... Niels Bohr - proposed an atomic model that placed electrons in circular orbits called energy levels. Electron Cloud - the modern atomic model. Electrons move in an area represented as a cloud around the nucleus. Quarks - smaller particles that make up protons and neutrons. There are six: up, down, c ...
Chapter 4 Study Guide
... Know how to read the periodic table (what information is given to you in each element square?). ...
... Know how to read the periodic table (what information is given to you in each element square?). ...
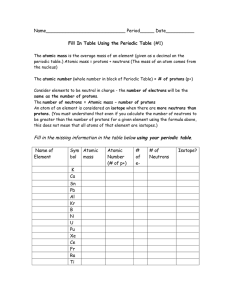
Periodic Table Fill in Table 1
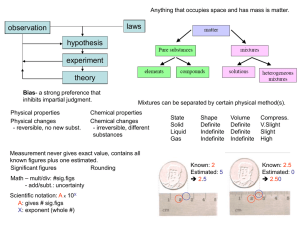
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
UNIT 5 REVIEW PROBLEMS
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
First Semester Honors Chemistry Exam Review (2011
... surrounded by how many electrons? 69. What do you need to know to draw a Lewis structure? 70. Multiple covalent bonds may occur in atoms that contain carbon, nitrogen, or… 71. Explain the valence electrons in metals. 72. Malleability and ductility are characteristic of substances with what type of b ...
... surrounded by how many electrons? 69. What do you need to know to draw a Lewis structure? 70. Multiple covalent bonds may occur in atoms that contain carbon, nitrogen, or… 71. Explain the valence electrons in metals. 72. Malleability and ductility are characteristic of substances with what type of b ...
Atomic History notes
... Excited State: When an atom absorbs energy from an outside source, the energy may cause an electron or electrons to move to a higher energy level. Electrons do NOT remain at these higher levels for very long. The electrons return (or fall back) to the original ground state. When they do, the electro ...
... Excited State: When an atom absorbs energy from an outside source, the energy may cause an electron or electrons to move to a higher energy level. Electrons do NOT remain at these higher levels for very long. The electrons return (or fall back) to the original ground state. When they do, the electro ...
Atomic Structure and the Periodic Table
... nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
... nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
Unit 1: Atomic Structure AP Chemistry
... proportions, but his data used percentages instead of weights. ...
... proportions, but his data used percentages instead of weights. ...
1 - cloudfront.net
... 19. How many protons, electrons, and neutrons does an atom with atomic number 50 and mass number 125 contain? 20. Who was the man who lived from 460 B.C. – 370 B.C., and was among the first to suggest the idea of atoms? 21. A fictitious element “X” has 10.0 % of the isotope with mass 55 amu, 20.0 % ...
... 19. How many protons, electrons, and neutrons does an atom with atomic number 50 and mass number 125 contain? 20. Who was the man who lived from 460 B.C. – 370 B.C., and was among the first to suggest the idea of atoms? 21. A fictitious element “X” has 10.0 % of the isotope with mass 55 amu, 20.0 % ...
File
... The smallest atomic unit The process of combining two light nuclei to form a heavier more stable nucleus The process of using a neutron to split a heavy nucleus into two nuclei with smaller mass numbers Brittle versus soft Stretchable Metal-like but does not contain all metal characteristics A posit ...
... The smallest atomic unit The process of combining two light nuclei to form a heavier more stable nucleus The process of using a neutron to split a heavy nucleus into two nuclei with smaller mass numbers Brittle versus soft Stretchable Metal-like but does not contain all metal characteristics A posit ...
I can describe an atom and its components I can relate energy levels
... electrons have similar properties. ○ ex) A sodium ion (atomic # 11) and a potassium ion (atomic # 19) each have one valence electron. ○ Both are highly reactive metals, and easily combine with other elements ...
... electrons have similar properties. ○ ex) A sodium ion (atomic # 11) and a potassium ion (atomic # 19) each have one valence electron. ○ Both are highly reactive metals, and easily combine with other elements ...