Blue Flashcards (CR) - mvhs
... NADH & FADH2 give up __________ to power active pumping of ____ through transmembrane proteins from ____________________ (location) to ____________________ (location). This pumping forms a concentration gradient. ______ is the final electron acceptor. ...
... NADH & FADH2 give up __________ to power active pumping of ____ through transmembrane proteins from ____________________ (location) to ____________________ (location). This pumping forms a concentration gradient. ______ is the final electron acceptor. ...
Energy Transfer and Glycolysis Cellular Respiration • Remember
... Remember that there are four stages that occur in three different places within the cell 1. Glycolysis: occurs in the cytoplasm 2. Pyruvate Oxidation: occurs in the matrix of the mitochondrion 3. Kreb Cycle: occurs in the matrix of the mitochondrion 4. Electron Transport and Chemiosmosis: occur on ...
... Remember that there are four stages that occur in three different places within the cell 1. Glycolysis: occurs in the cytoplasm 2. Pyruvate Oxidation: occurs in the matrix of the mitochondrion 3. Kreb Cycle: occurs in the matrix of the mitochondrion 4. Electron Transport and Chemiosmosis: occur on ...
A-Level Biology Unit 4: Chapter 2 ATP Common Exam
... Give two ways in which the properties of ATP make it a suitable source (this means RELEASE not storage) of energy in biological processes. Energy released in small/suitable amounts; ...
... Give two ways in which the properties of ATP make it a suitable source (this means RELEASE not storage) of energy in biological processes. Energy released in small/suitable amounts; ...
Stage 4 Digestion: Electron Transport Chain
... • allows H+ gradient to rush through using energy to make ATP ADP + P ATP ATP Conversions • 1 cytosolic NADH = 2 ATP • 1 mitochondrial NADH = 3 ATP • 1 mitochondrial FADH = 2 ATP Net ATP from 1 Glucose • 2 ATP (G) • 2 NADH2 (G) = 2 FADH2 (transport) • 2NADH2 (K prep) • 2 x 3 NADH2 (K) • 2 x FADH2 (K ...
... • allows H+ gradient to rush through using energy to make ATP ADP + P ATP ATP Conversions • 1 cytosolic NADH = 2 ATP • 1 mitochondrial NADH = 3 ATP • 1 mitochondrial FADH = 2 ATP Net ATP from 1 Glucose • 2 ATP (G) • 2 NADH2 (G) = 2 FADH2 (transport) • 2NADH2 (K prep) • 2 x 3 NADH2 (K) • 2 x FADH2 (K ...
Ch. 7 Study Guide
... □ I can explain how glucose is oxidized during glycolysis and the Krebs Cycle to produce reducing power in NADH and FADH ...
... □ I can explain how glucose is oxidized during glycolysis and the Krebs Cycle to produce reducing power in NADH and FADH ...
Metabolism - California Science Teacher
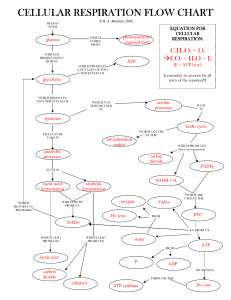
... During respiration most energy flows in a sequence: glucose, NADH, electron transport chain, proton, motive force, ATP. ...
... During respiration most energy flows in a sequence: glucose, NADH, electron transport chain, proton, motive force, ATP. ...
AP Biology: Chapter 9
... 7. What is the role of NAD+ & FAD+2 in respiration? 8. Explain why respiration is considered exergonic. 9. Glycolysis starts with _____________________ and produces 10. The Kreb’s cycle takes place in the: 11. Pyruvate is converted to ___________________________________ before the Krebs cycle. 12. T ...
... 7. What is the role of NAD+ & FAD+2 in respiration? 8. Explain why respiration is considered exergonic. 9. Glycolysis starts with _____________________ and produces 10. The Kreb’s cycle takes place in the: 11. Pyruvate is converted to ___________________________________ before the Krebs cycle. 12. T ...
Name ENERGY AND LIFE 8-1 pp 201
... 1. Which molecule stores more than 90 times the energy in an ATP molecule ? A. ADP B. water C. glucose D. adenine 2. All organisms get the ENERGY they need to regenerate ATP from __________________________ A. phosphates B. foods like glucose C. organelles D. ADP 3. CIRCLE ALL THE STATEMENTS THAT ARE ...
... 1. Which molecule stores more than 90 times the energy in an ATP molecule ? A. ADP B. water C. glucose D. adenine 2. All organisms get the ENERGY they need to regenerate ATP from __________________________ A. phosphates B. foods like glucose C. organelles D. ADP 3. CIRCLE ALL THE STATEMENTS THAT ARE ...
Redox (Reduction / Oxidation) Reaction: It is a great way of
... Oxidative phosphorylation - The process of taking something in a phosphate group and adding it onto another molecule Substrate level phosphorylation – The process of taking something in a phosphate group out of a molecule This is because the human body needs efficiency due to the high powering brain ...
... Oxidative phosphorylation - The process of taking something in a phosphate group and adding it onto another molecule Substrate level phosphorylation – The process of taking something in a phosphate group out of a molecule This is because the human body needs efficiency due to the high powering brain ...
Slide 1
... electron carriers. These electron carriers pass electrons from NADH and FADH to one another down a red-ox stairway. The net result of this series of step-wise electron exchanges is to pump H+ (protons) out of the matrix into the outer compartment between the outer and inner membrane of the mitochond ...
... electron carriers. These electron carriers pass electrons from NADH and FADH to one another down a red-ox stairway. The net result of this series of step-wise electron exchanges is to pump H+ (protons) out of the matrix into the outer compartment between the outer and inner membrane of the mitochond ...
Key Terms and Ideas: Fill in the blanks or provide a definition in your
... 2. ____________________________ is a simple transfer of a phosphate group from the substrate molecule to the ADP. 3. Glycolysis is an ____________ process; no oxygen is required for this process to occur. 4. The electron transport chain occurs on the ____________________ of the mitochondria. 5. ____ ...
... 2. ____________________________ is a simple transfer of a phosphate group from the substrate molecule to the ADP. 3. Glycolysis is an ____________ process; no oxygen is required for this process to occur. 4. The electron transport chain occurs on the ____________________ of the mitochondria. 5. ____ ...
Review 3
... (deoxy)ribonucleotides • Carbamoyl phosphate and urea • Pyruvate, oxaloacetate, a-ketoglutarate • PRPP ...
... (deoxy)ribonucleotides • Carbamoyl phosphate and urea • Pyruvate, oxaloacetate, a-ketoglutarate • PRPP ...
Advanced Cellular Respiration Worksheet
... 6. How many carbon dioxide molecules (CO2) are generated per pyruvate in the transition reaction? in the citric acid cycle? So therefore how many CO2 are produced per glucose? 7. How many NADH molecules are generated per glucose in a. glycolysis b. transition reaction ...
... 6. How many carbon dioxide molecules (CO2) are generated per pyruvate in the transition reaction? in the citric acid cycle? So therefore how many CO2 are produced per glucose? 7. How many NADH molecules are generated per glucose in a. glycolysis b. transition reaction ...
Chapter 18 - King William County Public Schools
... Reacts with alcohols to produce aldehydes and NADH ...
... Reacts with alcohols to produce aldehydes and NADH ...
Title - Iowa State University
... ATP ________ is the protein involved in ATP production. The process by which H+ is pumped back across the membrane to create ATP is called _____________. The amount of ATP produced is based on how many molecules of ______ are oxidized. ATP synthase works as a ________ machine, where H+ chang ...
... ATP ________ is the protein involved in ATP production. The process by which H+ is pumped back across the membrane to create ATP is called _____________. The amount of ATP produced is based on how many molecules of ______ are oxidized. ATP synthase works as a ________ machine, where H+ chang ...
SBI 4U Cellular Respiration Review Game2
... 20. What is the overall equation of aerobic cellular respiration? 21. How many NADH, FADH2 and ATP are produced per glucose molecule in the Kreb’s Cycle? 22. What is the final electron acceptor in the electron transport chain and describe what happens? 23. Where does the ETC occur in the cell? 24. H ...
... 20. What is the overall equation of aerobic cellular respiration? 21. How many NADH, FADH2 and ATP are produced per glucose molecule in the Kreb’s Cycle? 22. What is the final electron acceptor in the electron transport chain and describe what happens? 23. Where does the ETC occur in the cell? 24. H ...
Chapter-4 part-2 Energy Metabolism
... • Hydrolysis-dehydration • Add H20 • Remove H20 • Transfer of groups ...
... • Hydrolysis-dehydration • Add H20 • Remove H20 • Transfer of groups ...
How much ATP is produced in this cycle?
... Absorb certain wavelengths of light while reflecting others. ...
... Absorb certain wavelengths of light while reflecting others. ...
Glycolysis - Centre College
... • Charge repulsion of phosphates • Increase in entropy (number of molecules increases) • Resonance stabilization of product ...
... • Charge repulsion of phosphates • Increase in entropy (number of molecules increases) • Resonance stabilization of product ...
18.3 Important Coenzymes
... • These are nucleotide molecules • accept/deliver electrons for redox reactions • accept/delivers phosphates to generate ATP ...
... • These are nucleotide molecules • accept/deliver electrons for redox reactions • accept/delivers phosphates to generate ATP ...
PHOTOSYNTHESIS – The anabolic reduction of CO2 to form sugar.
... Note: the force that is driving the ETC is the pull on electrons exerted by the very highly electronegative O2. thus, “oxidative phosphorylation.” * NUMBERS ARE PER GLUCOSE MOLECULE Chemiosmosis, or chemiosmotic phosphorylation operates in both photosynthesis and cellular respiration, and is referre ...
... Note: the force that is driving the ETC is the pull on electrons exerted by the very highly electronegative O2. thus, “oxidative phosphorylation.” * NUMBERS ARE PER GLUCOSE MOLECULE Chemiosmosis, or chemiosmotic phosphorylation operates in both photosynthesis and cellular respiration, and is referre ...
Week 4:
... Fermentation: when oxygen is taken away, yields only the energy associated with glycolysis (2ATP) and yields one of several products that are generally harmful to living organisms at high concentrations: e.g. ethanol, lactic acid. Tuesday 4/26: We mentioned the terms oxidation, reduction, and “redox ...
... Fermentation: when oxygen is taken away, yields only the energy associated with glycolysis (2ATP) and yields one of several products that are generally harmful to living organisms at high concentrations: e.g. ethanol, lactic acid. Tuesday 4/26: We mentioned the terms oxidation, reduction, and “redox ...
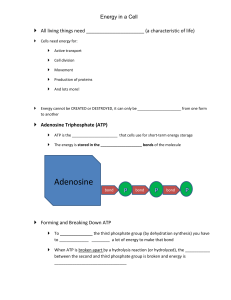
Adenosine triphosphate
Adenosine triphosphate (ATP) is a nucleoside triphosphate used in cells as a coenzyme often called the ""molecular unit of currency"" of intracellular energy transfer.ATP transports chemical energy within cells for metabolism. It is one of the end products of photophosphorylation, cellular respiration, and fermentation and used by enzymes and structural proteins in many cellular processes, including biosynthetic reactions, motility, and cell division. One molecule of ATP contains three phosphate groups, and it is produced by a wide variety of enzymes, including ATP synthase, from adenosine diphosphate (ADP) or adenosine monophosphate (AMP) and various phosphate group donors. Substrate-level phosphorylation, oxidative phosphorylation in cellular respiration, and photophosphorylation in photosynthesis are three major mechanisms of ATP biosynthesis.Metabolic processes that use ATP as an energy source convert it back into its precursors. ATP is therefore continuously recycled in organisms: the human body, which on average contains only 250 grams (8.8 oz) of ATP, turns over its own body weight equivalent in ATP each day.ATP is used as a substrate in signal transduction pathways by kinases that phosphorylate proteins and lipids. It is also used by adenylate cyclase, which uses ATP to produce the second messenger molecule cyclic AMP. The ratio between ATP and AMP is used as a way for a cell to sense how much energy is available and control the metabolic pathways that produce and consume ATP. Apart from its roles in signaling and energy metabolism, ATP is also incorporated into nucleic acids by polymerases in the process of transcription. ATP is the neurotransmitter believed to signal the sense of taste.The structure of this molecule consists of a purine base (adenine) attached by the 9' nitrogen atom to the 1' carbon atom of a pentose sugar (ribose). Three phosphate groups are attached at the 5' carbon atom of the pentose sugar. It is the addition and removal of these phosphate groups that inter-convert ATP, ADP and AMP. When ATP is used in DNA synthesis, the ribose sugar is first converted to deoxyribose by ribonucleotide reductase.ATP was discovered in 1929 by Karl Lohmann, and independently by Cyrus Fiske and Yellapragada Subbarow of Harvard Medical School, but its correct structure was not determined until some years later. It was proposed to be the intermediary molecule between energy-yielding and energy-requiring reactions in cells by Fritz Albert Lipmann in 1941. It was first artificially synthesized by Alexander Todd in 1948.