Note sheet Chap 5, Sect 3
... __3__ NADH; __2__ ATP; and __1___ FADH2 (another electron carrier) are produced. The __NADH___ and ___FADH2____ are electron carriers and store energy. Electron Transport Chain Electrons donated by __NADH__ and __FADH2__ pass to an electron transport chain in the _folds__ of the __mitochondrion_____ ...
... __3__ NADH; __2__ ATP; and __1___ FADH2 (another electron carrier) are produced. The __NADH___ and ___FADH2____ are electron carriers and store energy. Electron Transport Chain Electrons donated by __NADH__ and __FADH2__ pass to an electron transport chain in the _folds__ of the __mitochondrion_____ ...
Respiration
... What happens to the NADH produced in glycolysis and the Krebs cycle * joins with pyruvate to make glucose passes electrons to the ETC releases H+ ions into the stroma passes electrons to acetyl CoA Which part of cellular respiration produces the most ATP? * glycolysis ...
... What happens to the NADH produced in glycolysis and the Krebs cycle * joins with pyruvate to make glucose passes electrons to the ETC releases H+ ions into the stroma passes electrons to acetyl CoA Which part of cellular respiration produces the most ATP? * glycolysis ...
Anaerobic metabolism is the production of ATP with oxygen
... 2. True or False: An enzyme is not changed by the reaction it causes. 3. True or False: An enzyme does not need to fit precisely with the reactant to catalyze the reaction. 4. True or False: The electron transport system is where most of the ATP is produced during aerobic metabolism. 5. True or Fals ...
... 2. True or False: An enzyme is not changed by the reaction it causes. 3. True or False: An enzyme does not need to fit precisely with the reactant to catalyze the reaction. 4. True or False: The electron transport system is where most of the ATP is produced during aerobic metabolism. 5. True or Fals ...
Energy and Life 8-1
... • Energy stored in ATP is released when ATP is converted into ADP and a phosphate group. • Because a cell can add and subtract a third phosphate group, it has a way of storing and releasing energy as needed. • The characteristic of ATP make it an exceptionally useful molecule that is used by ALL typ ...
... • Energy stored in ATP is released when ATP is converted into ADP and a phosphate group. • Because a cell can add and subtract a third phosphate group, it has a way of storing and releasing energy as needed. • The characteristic of ATP make it an exceptionally useful molecule that is used by ALL typ ...
Diagrams to Review 1
... Chloroplasts convert radiant energy from the sun into chemical energy the plant can use. Its inner membrane is composed of flattened sacs called thylakoids. A stack of these is called a granum. The thylakoid membrane have a green pigment called chlorophyll in them. Chloroplasts are part of a larger ...
... Chloroplasts convert radiant energy from the sun into chemical energy the plant can use. Its inner membrane is composed of flattened sacs called thylakoids. A stack of these is called a granum. The thylakoid membrane have a green pigment called chlorophyll in them. Chloroplasts are part of a larger ...
Photosynthesis and Cellular Respiration
... Write the complete overall chemical equation for cellular respiration using chemical symbols instead of words: ____________ + _______________ → ________________+_________________+________________ Compare this reaction to the one you learned about last chapter for PHOTOSYNTHSIS (6 H2O + 6 CO2 + light ...
... Write the complete overall chemical equation for cellular respiration using chemical symbols instead of words: ____________ + _______________ → ________________+_________________+________________ Compare this reaction to the one you learned about last chapter for PHOTOSYNTHSIS (6 H2O + 6 CO2 + light ...
outline File
... Energy yield can fluctuate. ***Your textbook provides a modified energy yield of 32 ATP due to alternate calculations of ATP generated from electron carriers. We will discuss these alternate calculations in class*** 7.7 Regulation of Aerobic Respiration feedback inhibition 7.8 Oxidation Without Oxyg ...
... Energy yield can fluctuate. ***Your textbook provides a modified energy yield of 32 ATP due to alternate calculations of ATP generated from electron carriers. We will discuss these alternate calculations in class*** 7.7 Regulation of Aerobic Respiration feedback inhibition 7.8 Oxidation Without Oxyg ...
Cells and Energy Lesson Quiz B Completion LESSON 4
... Directions: On each line, write the term that correctly completes each sentence. ...
... Directions: On each line, write the term that correctly completes each sentence. ...
Cellular Respiration Chapter 7- Cfe Higher Human Biology
... PROTEINS AS RESPIRATORY SUBSTRATES Proteins in the diet are broken down to their component amino acids by the action of digestive enzymes. Amino acids in excess of the body’s requirements for protein synthesis undergo deamination, forming urea and respiratory pathway intermediates as shown opposite ...
... PROTEINS AS RESPIRATORY SUBSTRATES Proteins in the diet are broken down to their component amino acids by the action of digestive enzymes. Amino acids in excess of the body’s requirements for protein synthesis undergo deamination, forming urea and respiratory pathway intermediates as shown opposite ...
Ch. 4: ATP and Cellular Respiration
... compounds. • Compounds that store energy: ATP, NADH and FADH2. • When bonds are broken, energy is released. ...
... compounds. • Compounds that store energy: ATP, NADH and FADH2. • When bonds are broken, energy is released. ...
REVIEW - CELL RESPIRATION
... NAME: __________________________________________ DATE: ___________ PERIOD: _____ ...
... NAME: __________________________________________ DATE: ___________ PERIOD: _____ ...
-The oxygen consumed during cellular respiration is involved
... -In chemiosmosis, what is the most direct source of energy that is used to convert ADP + ...
... -In chemiosmosis, what is the most direct source of energy that is used to convert ADP + ...
Chapter 18 Electron Transport and Oxidative Phosphorylation
... The Hazards of the electron transport chain. An oxygen that is not completely Reduced to water will form free radicals. ...
... The Hazards of the electron transport chain. An oxygen that is not completely Reduced to water will form free radicals. ...
Review game
... 100 Where does the electron transport chain take place? Cristae or inner membrane of mitochondria 200 What is the final electron acceptor in the electron transport chain? Oxygen 300 Define chemiosmosis. Process of making ATP by movement of protons to provide energy so phosphorylation can occur 400 W ...
... 100 Where does the electron transport chain take place? Cristae or inner membrane of mitochondria 200 What is the final electron acceptor in the electron transport chain? Oxygen 300 Define chemiosmosis. Process of making ATP by movement of protons to provide energy so phosphorylation can occur 400 W ...
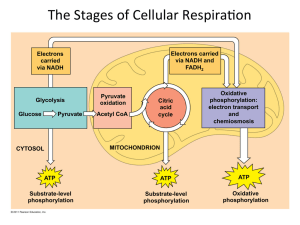
The Stages of Cellular RespiraWon
... (citric acid cycle) Cristae or inner membrane: electron transport chain and oxida3ve phosphoryla3on ...
... (citric acid cycle) Cristae or inner membrane: electron transport chain and oxida3ve phosphoryla3on ...
chapters-6-8-filled
... Cellular respiration begins with glycolysis, the breakdown of glucose to pyruvate. This consists of an energy investment phase and an energy payoff phase with a net gain of two molecules of ATP. In the presence of oxygen, pyruvate is broken down into carbon dioxide and an acetyl group. With the hel ...
... Cellular respiration begins with glycolysis, the breakdown of glucose to pyruvate. This consists of an energy investment phase and an energy payoff phase with a net gain of two molecules of ATP. In the presence of oxygen, pyruvate is broken down into carbon dioxide and an acetyl group. With the hel ...
Cell Respiration ch. 9
... Inner membrane proteins (cytochromes) carry electrons from NADH & FADH2 down to oxygen Chemiosmosis: energy coupling mechanism ATP synthase: produces ATP by using the H+ gradient (proton-motive force) pumped into the inner membrane space from the electron transport chain; this enzyme harnesses the f ...
... Inner membrane proteins (cytochromes) carry electrons from NADH & FADH2 down to oxygen Chemiosmosis: energy coupling mechanism ATP synthase: produces ATP by using the H+ gradient (proton-motive force) pumped into the inner membrane space from the electron transport chain; this enzyme harnesses the f ...
Cellular Respiration
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
... • The mitochondria are the engines of our cells where sugar is burned for fuel and the exhaust is CO2 and H2O. ...
How do we get energy?
... ATP is like your wallet filled with money, you can only spend what you have, then it has to be refilled. ATP carries chemical energy that cells use for building molecules and moving materials. ...
... ATP is like your wallet filled with money, you can only spend what you have, then it has to be refilled. ATP carries chemical energy that cells use for building molecules and moving materials. ...
Ch 31 reading guide
... 1.What is nitrogen fixation? How much ATP does it cost to form two molecuels of ammonia one molecule of N2? 2. Plants and micororganisms can incorporate ammonia into amino acids by reductive amination of a-ketoglutarate to form __________________. 3. Ammonia can also be incorporated into glutamate t ...
... 1.What is nitrogen fixation? How much ATP does it cost to form two molecuels of ammonia one molecule of N2? 2. Plants and micororganisms can incorporate ammonia into amino acids by reductive amination of a-ketoglutarate to form __________________. 3. Ammonia can also be incorporated into glutamate t ...
CELLULAR RESPIRATION
... Cellular Respiration - Cells transfer energy from food molecules to ATP, the byproduct is water and carbon dioxide ...
... Cellular Respiration - Cells transfer energy from food molecules to ATP, the byproduct is water and carbon dioxide ...
Chapter 13
... 3. Most metabolisms are carried out in specific organelles. For example, glycolysis takes place in cytosol, whereas the citric acid cycle and the oxidative phosphorylation processes occur in mitochondria. Important metabolic functions are listed in Table 15-2. 4. ATP is the most important cellular e ...
... 3. Most metabolisms are carried out in specific organelles. For example, glycolysis takes place in cytosol, whereas the citric acid cycle and the oxidative phosphorylation processes occur in mitochondria. Important metabolic functions are listed in Table 15-2. 4. ATP is the most important cellular e ...
Adenosine triphosphate
Adenosine triphosphate (ATP) is a nucleoside triphosphate used in cells as a coenzyme often called the ""molecular unit of currency"" of intracellular energy transfer.ATP transports chemical energy within cells for metabolism. It is one of the end products of photophosphorylation, cellular respiration, and fermentation and used by enzymes and structural proteins in many cellular processes, including biosynthetic reactions, motility, and cell division. One molecule of ATP contains three phosphate groups, and it is produced by a wide variety of enzymes, including ATP synthase, from adenosine diphosphate (ADP) or adenosine monophosphate (AMP) and various phosphate group donors. Substrate-level phosphorylation, oxidative phosphorylation in cellular respiration, and photophosphorylation in photosynthesis are three major mechanisms of ATP biosynthesis.Metabolic processes that use ATP as an energy source convert it back into its precursors. ATP is therefore continuously recycled in organisms: the human body, which on average contains only 250 grams (8.8 oz) of ATP, turns over its own body weight equivalent in ATP each day.ATP is used as a substrate in signal transduction pathways by kinases that phosphorylate proteins and lipids. It is also used by adenylate cyclase, which uses ATP to produce the second messenger molecule cyclic AMP. The ratio between ATP and AMP is used as a way for a cell to sense how much energy is available and control the metabolic pathways that produce and consume ATP. Apart from its roles in signaling and energy metabolism, ATP is also incorporated into nucleic acids by polymerases in the process of transcription. ATP is the neurotransmitter believed to signal the sense of taste.The structure of this molecule consists of a purine base (adenine) attached by the 9' nitrogen atom to the 1' carbon atom of a pentose sugar (ribose). Three phosphate groups are attached at the 5' carbon atom of the pentose sugar. It is the addition and removal of these phosphate groups that inter-convert ATP, ADP and AMP. When ATP is used in DNA synthesis, the ribose sugar is first converted to deoxyribose by ribonucleotide reductase.ATP was discovered in 1929 by Karl Lohmann, and independently by Cyrus Fiske and Yellapragada Subbarow of Harvard Medical School, but its correct structure was not determined until some years later. It was proposed to be the intermediary molecule between energy-yielding and energy-requiring reactions in cells by Fritz Albert Lipmann in 1941. It was first artificially synthesized by Alexander Todd in 1948.