Document
... • Oxidoreductases catalyze oxidation–reduction reactions of substrate molecules, most commonly addition or removal of oxygen or hydrogen. Because oxidation and reduction must occur together, these enzymes require coenzymes that are reduced or oxidized as the substrate is oxidized or reduced. ...
... • Oxidoreductases catalyze oxidation–reduction reactions of substrate molecules, most commonly addition or removal of oxygen or hydrogen. Because oxidation and reduction must occur together, these enzymes require coenzymes that are reduced or oxidized as the substrate is oxidized or reduced. ...
Chapter 8: Energy generation:glycolysis
... FAD and FMN act in a similar way, but reacting with two protons rather than one: FAD + 2H+ + 2e– Æ FADH2 FMN + 2H+ + 2e– Æ FMNH2 Both FAD and FMN, like NAD+, are involved in the energy-generating pathway. ...
... FAD and FMN act in a similar way, but reacting with two protons rather than one: FAD + 2H+ + 2e– Æ FADH2 FMN + 2H+ + 2e– Æ FMNH2 Both FAD and FMN, like NAD+, are involved in the energy-generating pathway. ...
Nutrition, Metabolism, and Body Temperature Regulation
... Proteins are important structural materials of the body, including, for example, keratin in skin, collagen and elastin in connective tissues, and muscle proteins. In addition, functional proteins such as enzymes and some hormones regulate an incredible variety of body functions. Whether amino acids ...
... Proteins are important structural materials of the body, including, for example, keratin in skin, collagen and elastin in connective tissues, and muscle proteins. In addition, functional proteins such as enzymes and some hormones regulate an incredible variety of body functions. Whether amino acids ...
Interval Training Interval Training Understand Energy Systems to
... System Type of recovery Work:rest 1:1/2 Recovery: light activity Types of activities >5 Km run Triathlon z 1500 m swim z z ...
... System Type of recovery Work:rest 1:1/2 Recovery: light activity Types of activities >5 Km run Triathlon z 1500 m swim z z ...
Metabolism
... and metal ions such as iron (Fe2+), copper (Cu2+), and magnesium (Mg2+). coenzymes Organic compounds, often B vitamin derivatives, that combine with an inactive enzyme to form an active enzyme. Coenzymes associate closely with these enzymes, allowing them to catalyze certain metabolic reactions in ...
... and metal ions such as iron (Fe2+), copper (Cu2+), and magnesium (Mg2+). coenzymes Organic compounds, often B vitamin derivatives, that combine with an inactive enzyme to form an active enzyme. Coenzymes associate closely with these enzymes, allowing them to catalyze certain metabolic reactions in ...
Bio102 Problems
... 12A. If we forgot to add any NaCl when preparing the PBS, what would happen to the white blood cells? Why? 12B. If we accidentally added 280mM NaCl while preparing the PBS, what would happen to the white blood cells? Why? 12C. After they have lived in the test tube for several hours, we are able to ...
... 12A. If we forgot to add any NaCl when preparing the PBS, what would happen to the white blood cells? Why? 12B. If we accidentally added 280mM NaCl while preparing the PBS, what would happen to the white blood cells? Why? 12C. After they have lived in the test tube for several hours, we are able to ...
BTEC National in Applied Science Unit 01 Sample redacted web
... An ammonium ion contains a dative bond (see Figure 1.8). When ammonia reacts with hydrochloric acid, a hydrogen ion from the acid is transferred to the ammonia molecule. A lone pair of electrons on the nitrogen atom forms a dative covalent bond with the hydrogen ion. ...
... An ammonium ion contains a dative bond (see Figure 1.8). When ammonia reacts with hydrochloric acid, a hydrogen ion from the acid is transferred to the ammonia molecule. A lone pair of electrons on the nitrogen atom forms a dative covalent bond with the hydrogen ion. ...
Lec 12: Fatty acid biosynthesis
... for fatty acid biosynthesis. This is often believed to be the limiting step of fatty acid biosynthesis ACCase is a multifunctional enzyme with 3 enzymatic activities: • Bacterial ACCase is complex of 3 proteins (one of them is complex of 2 polypeptides). For example: E. coli ACCase is coded by gene ...
... for fatty acid biosynthesis. This is often believed to be the limiting step of fatty acid biosynthesis ACCase is a multifunctional enzyme with 3 enzymatic activities: • Bacterial ACCase is complex of 3 proteins (one of them is complex of 2 polypeptides). For example: E. coli ACCase is coded by gene ...
Study of the distribution of autotrophic CO2 fixation
... carboxylase as the carboxylating enzyme has been identified for (micro)aerobic members of the Sulfolobales. The dicarboxylate/4-hydroxybutyrate cycle using oxygen-sensitive pyruvate synthase and phosphoenolpyruvate carboxylase as carboxylating enzymes has been found in members of the anaerobic Desul ...
... carboxylase as the carboxylating enzyme has been identified for (micro)aerobic members of the Sulfolobales. The dicarboxylate/4-hydroxybutyrate cycle using oxygen-sensitive pyruvate synthase and phosphoenolpyruvate carboxylase as carboxylating enzymes has been found in members of the anaerobic Desul ...
Sulfur Metabolism and Sulfur-Containing Amino Acids
... liberating pyrophosphate (PPi). The product of this reaction, APS is thus the activated form of SO42- [11]. In plants, S metabolism starts with soil SO42- assimilatory reduction pathways forming organic species, firstly the S-AA Cysteine (Cys), which then produces Methionine (Met) and all other bioo ...
... liberating pyrophosphate (PPi). The product of this reaction, APS is thus the activated form of SO42- [11]. In plants, S metabolism starts with soil SO42- assimilatory reduction pathways forming organic species, firstly the S-AA Cysteine (Cys), which then produces Methionine (Met) and all other bioo ...
ЛЕКЦІЯ 2
... membrane in the same way that action potentials travel along nerve fiber membranes. 6. The action potential depolarizes the muscle membrane, and fiber. Here it causes the sarcoplasmic reticulum to release large quantities of calcium ions that have been stored within this reticulum. 7. The calcium io ...
... membrane in the same way that action potentials travel along nerve fiber membranes. 6. The action potential depolarizes the muscle membrane, and fiber. Here it causes the sarcoplasmic reticulum to release large quantities of calcium ions that have been stored within this reticulum. 7. The calcium io ...
Alcohol Metabolism
... 1st step – oxidation by alcohol dehydrogenase – rate-limiting and zero-order (constant amount/unit time) EtOH oxidation requires NAD+ and its availability: o is rate-limiting factor (limits EtOH metabolism to about 8g/h) o leads to competition between EtOH and other metabolic substrates for avai ...
... 1st step – oxidation by alcohol dehydrogenase – rate-limiting and zero-order (constant amount/unit time) EtOH oxidation requires NAD+ and its availability: o is rate-limiting factor (limits EtOH metabolism to about 8g/h) o leads to competition between EtOH and other metabolic substrates for avai ...
Mitochondrial dysfunction and molecular pathways of
... contain riboflavin, iron-sulfur clusters, copper centers, or ironcontaining heme moieties. Ubiquinone shuttles electrons from complexes I and II to complex III. Cytochrome c, an iron-containing heme protein with a binuclear center of a copper ion (Hunter et al., 2000), transfers electrons from compl ...
... contain riboflavin, iron-sulfur clusters, copper centers, or ironcontaining heme moieties. Ubiquinone shuttles electrons from complexes I and II to complex III. Cytochrome c, an iron-containing heme protein with a binuclear center of a copper ion (Hunter et al., 2000), transfers electrons from compl ...
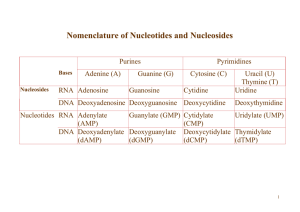
Nomenclature of Nucleotides and Nucleosides
... Nucleoside phosphorylases catalyze the phosphorolysis of nucleosides to free bases and ribose-1 -phosphate or deoxyribose-1 -phosphate. 1. In phosphorolysis, a bond is cleaved by the addition of inorganic phosphate (Pi) across it: Inosine + Pi ↔ Hypoxanthine + Ribose-1-phosphate Deoxyinosine + Pi ↔ ...
... Nucleoside phosphorylases catalyze the phosphorolysis of nucleosides to free bases and ribose-1 -phosphate or deoxyribose-1 -phosphate. 1. In phosphorolysis, a bond is cleaved by the addition of inorganic phosphate (Pi) across it: Inosine + Pi ↔ Hypoxanthine + Ribose-1-phosphate Deoxyinosine + Pi ↔ ...
22. pyruvate oxidation and citric acid cycle
... Energy charge and the metabolic rate The energy charge is one if the total adenine nucleotide pool is fully phosphorylated to ATP High concentrations of ATP inhibit the relative rates of (i.e., it is all ATP) and zero if the adenine a typical ATP-generating (= catabolic) pathway and nucleotides are ...
... Energy charge and the metabolic rate The energy charge is one if the total adenine nucleotide pool is fully phosphorylated to ATP High concentrations of ATP inhibit the relative rates of (i.e., it is all ATP) and zero if the adenine a typical ATP-generating (= catabolic) pathway and nucleotides are ...
8)Discuss the roles of cofactors and coenzymes in enzyme activity.
... 8) Some enzymes are multienzyme or multifunctional complexes. ...
... 8) Some enzymes are multienzyme or multifunctional complexes. ...
Document
... at higher levels of glucose, this hexokinase will allow the muscle to rapidly metabolize glucose, which will facilitate glucose removal from the blood. 6. (2 pts) Why are high HDL levels considered to be good for health? Give 2 reasons. High HDL means more removal of cholesterol from peripheral tiss ...
... at higher levels of glucose, this hexokinase will allow the muscle to rapidly metabolize glucose, which will facilitate glucose removal from the blood. 6. (2 pts) Why are high HDL levels considered to be good for health? Give 2 reasons. High HDL means more removal of cholesterol from peripheral tiss ...
PDF
... Maris et al., 2004b). The high intracellular pH ensures that the majority of lactic acid is present as the lactate anion, which is incapable of diffusing across the plasma membrane. Therefore, analogous to the export of other weak organic acid anions (Piper et al., 1998; Fernandes et al., 2005), it ...
... Maris et al., 2004b). The high intracellular pH ensures that the majority of lactic acid is present as the lactate anion, which is incapable of diffusing across the plasma membrane. Therefore, analogous to the export of other weak organic acid anions (Piper et al., 1998; Fernandes et al., 2005), it ...
幻灯片 1
... the fatty acids from which they are derived but they make up for this deficiency by serving as “water-soluble lipids” that can be more readily transported in the blood plasma. During starvation, ketone bodies are produced in large amounts becoming substitutes for glucose as the principal fuel for br ...
... the fatty acids from which they are derived but they make up for this deficiency by serving as “water-soluble lipids” that can be more readily transported in the blood plasma. During starvation, ketone bodies are produced in large amounts becoming substitutes for glucose as the principal fuel for br ...
Proton-Coupled Electron Flow in Protein Redox Machines
... McConnell superexchange coupling model58 that accounts for rate scatter attributable to protein structural complexity. In this tunneling-pathway model, the medium between D and A is decomposed to smaller subunits linked by covalent bonds, hydrogen bonds, and through-space jumps. More elaborate compu ...
... McConnell superexchange coupling model58 that accounts for rate scatter attributable to protein structural complexity. In this tunneling-pathway model, the medium between D and A is decomposed to smaller subunits linked by covalent bonds, hydrogen bonds, and through-space jumps. More elaborate compu ...
CHOLESTEROL SYNTHESIS
... multiple reactions that involve the removal of three methyl groups, hydrogenation of the double bond in the side-chain, and a shift of the double bond from position 8,9 to 5,6 in ring B. ...
... multiple reactions that involve the removal of three methyl groups, hydrogenation of the double bond in the side-chain, and a shift of the double bond from position 8,9 to 5,6 in ring B. ...
Additional data file
... 3-ketoacyl-CoA thiolase with broad chain length specificity, cleaves 3-ketoacylCoA into acyl-CoA and acetyl-CoA during beta-oxidation of fatty acids Beta-isopropylmalate dehydrogenase, catalyzes the third step in the leucine biosynthesis pathway Cytoplasmic aldehyde dehydrogenase, involved in beta-a ...
... 3-ketoacyl-CoA thiolase with broad chain length specificity, cleaves 3-ketoacylCoA into acyl-CoA and acetyl-CoA during beta-oxidation of fatty acids Beta-isopropylmalate dehydrogenase, catalyzes the third step in the leucine biosynthesis pathway Cytoplasmic aldehyde dehydrogenase, involved in beta-a ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.