Effect of Temperature Increasing the temperature increases the
... • Proteins take on the 3-D structure with lowest potential energy - increases their stability • Increased energy causes increased motion within the molecule as well as between molecules • Weak bonds in the tertiary structure (hydrogen bonds) are broken and new bonds form in different positions • New ...
... • Proteins take on the 3-D structure with lowest potential energy - increases their stability • Increased energy causes increased motion within the molecule as well as between molecules • Weak bonds in the tertiary structure (hydrogen bonds) are broken and new bonds form in different positions • New ...
Engineering of metabolic pathways by artificial enzyme channels
... or contradictory reactions such as synthesis (anabolism) and degradation (catabolism) or oxidation and reduction to take place within the same cell at the same time. Compartmentation also allows detoxification of toxic pathway intermediates without harming the cell. In peroxisomes, for example, the ...
... or contradictory reactions such as synthesis (anabolism) and degradation (catabolism) or oxidation and reduction to take place within the same cell at the same time. Compartmentation also allows detoxification of toxic pathway intermediates without harming the cell. In peroxisomes, for example, the ...
Early bioenergetic evolution
... pathway becomes endergonic at low H2 partial pressures, with DG80 ¼ þ29 kJ mol21 at approximately 10 Pa H2 [73]. Equation (3.1) entails the thiol group of coenzyme A (CoASH) rather than CH3SH, and it is the line reaction of the acetyl-CoA or Wood– Ljungdahl [74,75] pathway of microbial CO2 fixation. ...
... pathway becomes endergonic at low H2 partial pressures, with DG80 ¼ þ29 kJ mol21 at approximately 10 Pa H2 [73]. Equation (3.1) entails the thiol group of coenzyme A (CoASH) rather than CH3SH, and it is the line reaction of the acetyl-CoA or Wood– Ljungdahl [74,75] pathway of microbial CO2 fixation. ...
Lesson 8. Enzymes
... reactions proceed too slowly on their own to sustain life. Hence catalysts are required to greatly accelerate the rates of these chemical reactions. In nature enzymes posses the catalytic power to facilitate life processes in essentially all life-forms from viruses to man. Most of the enzymes retain ...
... reactions proceed too slowly on their own to sustain life. Hence catalysts are required to greatly accelerate the rates of these chemical reactions. In nature enzymes posses the catalytic power to facilitate life processes in essentially all life-forms from viruses to man. Most of the enzymes retain ...
Calvin Cycle Flux, Pathway Constraints, and
... all substrates, excreted organic acids were eventually consumed. Since fumarate and malate have the same oxidation state, we grouped them as one metabolite to account for their simultaneous consumption and to better reflect the effects of intermediary metabolic fluxes on H2 yield. If we were to show ...
... all substrates, excreted organic acids were eventually consumed. Since fumarate and malate have the same oxidation state, we grouped them as one metabolite to account for their simultaneous consumption and to better reflect the effects of intermediary metabolic fluxes on H2 yield. If we were to show ...
1 ENZYME KINETICS [APPLICATION OF UV
... molecules collide and bind with them. The temperature effect can be explained in terms of collision theory within a certain temperature range: molecular motion and hence collisions between enzyme and substrate speed up as the temperature rises but as enzymes are proteins, there is an upper limit bey ...
... molecules collide and bind with them. The temperature effect can be explained in terms of collision theory within a certain temperature range: molecular motion and hence collisions between enzyme and substrate speed up as the temperature rises but as enzymes are proteins, there is an upper limit bey ...
Modification of halogen specificity of a vanadium‐dependent
... residue located at the chlorine binding site of various amylases (Machius et al. 1995). In the case of BPO from C. pilulifera, the substituted tryptophan or phenylalanine residues at position 397 could participate in chloride binding. In the native BPO enzyme the active site cavity provides the corr ...
... residue located at the chlorine binding site of various amylases (Machius et al. 1995). In the case of BPO from C. pilulifera, the substituted tryptophan or phenylalanine residues at position 397 could participate in chloride binding. In the native BPO enzyme the active site cavity provides the corr ...
The CoFactor database: organic cofactors in enzyme catalysis
... representations of the cofactor’s distribution over enzyme reaction space and its chemical profile, based on the enzyme classification (NC-IUBMB and Webb, 1992). ...
... representations of the cofactor’s distribution over enzyme reaction space and its chemical profile, based on the enzyme classification (NC-IUBMB and Webb, 1992). ...
25-2 Carbohydrate Metabolism
... substrate in citric acid cycle by NAD • Pumps 6 hydrogen ions into intermembrane space • Reentry into matrix generates 3 molecules of ATP ...
... substrate in citric acid cycle by NAD • Pumps 6 hydrogen ions into intermembrane space • Reentry into matrix generates 3 molecules of ATP ...
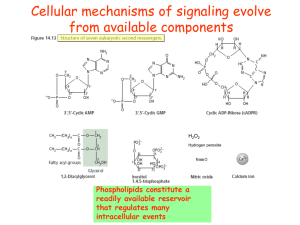
Phospholipid signaling
... Phospholipid signaling under salt stress, drought, cold, or ABA. Osmotic stress, cold, and ABA activate several types of phospholipases that cleave phospholipids to generate lipid messengers (e.g., PA, DAG, and IP3), which ...
... Phospholipid signaling under salt stress, drought, cold, or ABA. Osmotic stress, cold, and ABA activate several types of phospholipases that cleave phospholipids to generate lipid messengers (e.g., PA, DAG, and IP3), which ...
1 ENZYME KINETICS [APPLICATION OF UV
... molecules collide and bind with them. The temperature effect can be explained in terms of collision theory within a certain temperature range: molecular motion and hence collisions between enzyme and substrate speed up as the temperature rises but as enzymes are proteins, there is an upper limit bey ...
... molecules collide and bind with them. The temperature effect can be explained in terms of collision theory within a certain temperature range: molecular motion and hence collisions between enzyme and substrate speed up as the temperature rises but as enzymes are proteins, there is an upper limit bey ...
Document
... addition of a phosphate group to adenosine diphosphate (ADP) • The energy to phosphorylate ADP comes from catabolic reactions in the cell • The ATP cycle is a revolving door through which energy passes during its transfer from catabolic to anabolic pathways ...
... addition of a phosphate group to adenosine diphosphate (ADP) • The energy to phosphorylate ADP comes from catabolic reactions in the cell • The ATP cycle is a revolving door through which energy passes during its transfer from catabolic to anabolic pathways ...
Crystal Structure of 4-Chlorobenzoate:CoA Ligase/Synthetase in the
... thioester formation from adenylated short or long chain fatty acids or adenylated benzoic acid derivatives. The nonribosomal peptide synthetases (NRPSs) are composed of numerous catalytic domains linked in a modular fashion, often within a single polypeptide that can be thousands of residues in leng ...
... thioester formation from adenylated short or long chain fatty acids or adenylated benzoic acid derivatives. The nonribosomal peptide synthetases (NRPSs) are composed of numerous catalytic domains linked in a modular fashion, often within a single polypeptide that can be thousands of residues in leng ...
Chapter 6 Power Points
... addition of a phosphate group to adenosine diphosphate (ADP) • The energy to phosphorylate ADP comes from catabolic reactions in the cell • The ATP cycle is a revolving door through which energy passes during its transfer from catabolic to anabolic pathways ...
... addition of a phosphate group to adenosine diphosphate (ADP) • The energy to phosphorylate ADP comes from catabolic reactions in the cell • The ATP cycle is a revolving door through which energy passes during its transfer from catabolic to anabolic pathways ...
Gelatinization of Starch
... Hydrocolloids are substances that will form a gel or add viscosity on addition of water. ...
... Hydrocolloids are substances that will form a gel or add viscosity on addition of water. ...
on the potential efficiency of converting solar radiation to phytoenergy
... After briefly considering the spectral properties of solar radiation and how effectively plants can absorb it (the top ‘half’ of Fig. 1a,b), the analysis turns to quantitative biochemistry. This includes summing up the reactions that convert CO2 to photosynthate (i.e. sucrose and starch) to quantify ...
... After briefly considering the spectral properties of solar radiation and how effectively plants can absorb it (the top ‘half’ of Fig. 1a,b), the analysis turns to quantitative biochemistry. This includes summing up the reactions that convert CO2 to photosynthate (i.e. sucrose and starch) to quantify ...
ExamView - Test 2 Ch 5-9 Take Home Exam DUE IN CLASS NO
... b. mitochondrial outer membrane c. mitochondrial inner membrane d. mitochondrial intermembrane space e. cytosol The oxygen consumed during cellular respiration is involved directly in which process or event? a. glycolysis b. accepting electrons at the end of the electron transport chain c. the citri ...
... b. mitochondrial outer membrane c. mitochondrial inner membrane d. mitochondrial intermembrane space e. cytosol The oxygen consumed during cellular respiration is involved directly in which process or event? a. glycolysis b. accepting electrons at the end of the electron transport chain c. the citri ...
PowerPoint
... – E. In the fourth stage, the electron transport system (ETS), electrons of the hydrogen molecules released in the earlier stages are passed through a series of acceptors. As they travel, they go through chemical reactions in which oxidation (the combining with oxygen) and reduction (the removing of ...
... – E. In the fourth stage, the electron transport system (ETS), electrons of the hydrogen molecules released in the earlier stages are passed through a series of acceptors. As they travel, they go through chemical reactions in which oxidation (the combining with oxygen) and reduction (the removing of ...
Ketoglutarate Dehydrogenase Complex
... a diaphorase, carrying out single-electron transfers from a variety of electron donors to a variety of electron acceptors.28 The R-enantiomer is >25-fold more active as a substrate than the S-enantiomer.34 The amount of E3 found in mitochondria typically exceeds the amount required for the activity ...
... a diaphorase, carrying out single-electron transfers from a variety of electron donors to a variety of electron acceptors.28 The R-enantiomer is >25-fold more active as a substrate than the S-enantiomer.34 The amount of E3 found in mitochondria typically exceeds the amount required for the activity ...
Free Radicals, Oxidative Stress, and Diseases
... - chemotherapeutic agents, such as quinones _______________________________________________________________________________ ...
... - chemotherapeutic agents, such as quinones _______________________________________________________________________________ ...
Lecture 32: Protein (Part-I)
... Introduction: Proteins perform multiple functions in a cell and they are the factors to control several events.They are the building blocks and work as enzyme to participate in metabolic reactions of the organism. Peptide Bonds: Proteins are polymers of amino acids, joined by the covalent bonds, kno ...
... Introduction: Proteins perform multiple functions in a cell and they are the factors to control several events.They are the building blocks and work as enzyme to participate in metabolic reactions of the organism. Peptide Bonds: Proteins are polymers of amino acids, joined by the covalent bonds, kno ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.