10 - LifeSciTRC
... 2. NADPH, Xanthine Oxidase, Complex I & III 3. Inhibition of Ox-Phos allows ubiquinone to form ubisemiubiquinone. ...
... 2. NADPH, Xanthine Oxidase, Complex I & III 3. Inhibition of Ox-Phos allows ubiquinone to form ubisemiubiquinone. ...
Understanding Our Environment
... Plants w (has high affinity to CO2 and is not sensitive to oxygen).ith Kranz Anatomy produce oxaloacetic acid (4-carbon compound). Phosphoenolpyruvate (PEP) and carbon dioxide are combined in mesophyll cells with the aid of PEP carboxylase. PEP carboxylase has high affinity to CO2 and is not sen ...
... Plants w (has high affinity to CO2 and is not sensitive to oxygen).ith Kranz Anatomy produce oxaloacetic acid (4-carbon compound). Phosphoenolpyruvate (PEP) and carbon dioxide are combined in mesophyll cells with the aid of PEP carboxylase. PEP carboxylase has high affinity to CO2 and is not sen ...
oxidation
... Stage 3: Oxidative phosphorylation – involves electrons carried by NADH and FADH2, – shuttles these electrons to the electron transport chain embedded in the inner mitochondrial membrane, – involves chemiosmosis, and – generates ATP through oxidative phosphorylation associated with chemiosmosis. ...
... Stage 3: Oxidative phosphorylation – involves electrons carried by NADH and FADH2, – shuttles these electrons to the electron transport chain embedded in the inner mitochondrial membrane, – involves chemiosmosis, and – generates ATP through oxidative phosphorylation associated with chemiosmosis. ...
Chapter 9 Cellular Respiration (working)
... chemiosmosis couples electron transport to ATP synthesis • Following glycolysis and the citric acid cycle, NADH and FADH2 account for most of the energy extracted from food • These two electron carriers donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosph ...
... chemiosmosis couples electron transport to ATP synthesis • Following glycolysis and the citric acid cycle, NADH and FADH2 account for most of the energy extracted from food • These two electron carriers donate electrons to the electron transport chain, which powers ATP synthesis via oxidative phosph ...
What happened to my cousin Patrick O’Neill?
... B: His muscles are not functioning properly. C: He cannot efficiently break down food for energy. D: All of the above are possible causes. ...
... B: His muscles are not functioning properly. C: He cannot efficiently break down food for energy. D: All of the above are possible causes. ...
Bio AP chp 9 notes
... Under aerobic respiration, a molecule of glucose yields 38 ATP, but the same molecule of glucose yields only 2 ATP under anaerobic respiration. ...
... Under aerobic respiration, a molecule of glucose yields 38 ATP, but the same molecule of glucose yields only 2 ATP under anaerobic respiration. ...
cellular-respiration 1
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
Chapter 8 Cellular Respiration Dr. Harold Kay Njemanze 8.1
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
Chapter 8 Cellular Respiration 8.1 Cellular Respiration 1. Cellular
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
... c. Some of the protein carriers are cytochrome molecules, complex carbon rings with a heme (iron) group in the center. 4. Cycling of Carriers a. By the time electrons are received by O2, three ATP have been made. b. When FADH2 delivers electrons to the electron transport system, two ATP are formed b ...
Lecture_10_F11
... Cellular Respiration: the big picture • process in which cells consume O2 and produce CO2 ...
... Cellular Respiration: the big picture • process in which cells consume O2 and produce CO2 ...
Plant Respiration Exchange of Gases in Plants - E
... Answer: The metabolic pathway through which the electron passes from one carrier to another, is called the electron transport system (ETS). This pathway is present in the inner mitochondrial membrane. Electrons from NADH (produced in the mitochondria matrix) are oxidized by an NADH dehydrogenase (Co ...
... Answer: The metabolic pathway through which the electron passes from one carrier to another, is called the electron transport system (ETS). This pathway is present in the inner mitochondrial membrane. Electrons from NADH (produced in the mitochondria matrix) are oxidized by an NADH dehydrogenase (Co ...
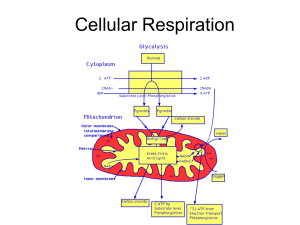
Cellular Respiration
... 3. Write the equation for photosynthesis. 4. Write the equation for cell respiration…notice they are opposites! 5. Which step of cell respiration breaks down glucose? 6. The Acetyl CoA enzyme from step 1 enters step 2 of the Citric Acid Cycle / Krebs Cycle and forms ___ & ___. 7. The electrons from ...
... 3. Write the equation for photosynthesis. 4. Write the equation for cell respiration…notice they are opposites! 5. Which step of cell respiration breaks down glucose? 6. The Acetyl CoA enzyme from step 1 enters step 2 of the Citric Acid Cycle / Krebs Cycle and forms ___ & ___. 7. The electrons from ...
AP Biology - mvhs
... Redox reactions – LEO, GER; where do electrons originate, what pulls them away Light- Dependent Reactions – location, purpose; role of chlorophyll, water and photosystems; how is ATP/NADPH produced; reason for noncyclic vs. cyclic electron flow Calvin Cycle – location, purpose, dependence on light r ...
... Redox reactions – LEO, GER; where do electrons originate, what pulls them away Light- Dependent Reactions – location, purpose; role of chlorophyll, water and photosystems; how is ATP/NADPH produced; reason for noncyclic vs. cyclic electron flow Calvin Cycle – location, purpose, dependence on light r ...
Table of Contents - Milan Area Schools
... • The respiratory chain uses the reducing agents generated by pyruvate oxidation and the citric acid cycle. • The flow of electrons in a series of redox reactions causes the active transport of protons across the inner mitochondrial membrane, creating a proton concentration gradient. • The protons t ...
... • The respiratory chain uses the reducing agents generated by pyruvate oxidation and the citric acid cycle. • The flow of electrons in a series of redox reactions causes the active transport of protons across the inner mitochondrial membrane, creating a proton concentration gradient. • The protons t ...
Electrontransfer proteins
... - In the case of cytochrome a and b the hem is bound strongly but not covalently to the protein. In the case of cytochrome c the hem and the protein bind covalently. Most of the cytochromes are 1:1 units (1 hem + 1 protein), but there are cytochromes with multiple hem units. - The cytochromes always ...
... - In the case of cytochrome a and b the hem is bound strongly but not covalently to the protein. In the case of cytochrome c the hem and the protein bind covalently. Most of the cytochromes are 1:1 units (1 hem + 1 protein), but there are cytochromes with multiple hem units. - The cytochromes always ...
Chapter 8 THE ENERGY CONSUMING PROCESS OF RESPIRATION
... Each (8) gains one(9)_____________ group from the cytoplasm, then (10)_____________ atoms and electrons from each PGAL are transferred to NAD, changing this coenzyme to NADH. At the same time, two (11) _____________ molecules form by substrate-level phosphorylation; the cell’s energy investment is p ...
... Each (8) gains one(9)_____________ group from the cytoplasm, then (10)_____________ atoms and electrons from each PGAL are transferred to NAD, changing this coenzyme to NADH. At the same time, two (11) _____________ molecules form by substrate-level phosphorylation; the cell’s energy investment is p ...
Evolution of the citric acid cycle and respiratory
... only to ensure redox balance (various other accessory oxidants including certain inorganic compounds could have functioned in similar fashion [10]). I have proposed [1] that the soluble fumarate reduction mechanism gradually evolved by addition of intermediary electron carriers and incorporation of ...
... only to ensure redox balance (various other accessory oxidants including certain inorganic compounds could have functioned in similar fashion [10]). I have proposed [1] that the soluble fumarate reduction mechanism gradually evolved by addition of intermediary electron carriers and incorporation of ...
Cell Respiration powerpoint slides
... We get energy from ATP by breaking the bonds between the last two phosphates in the ATP chain. Then the cycle starts all over again! ...
... We get energy from ATP by breaking the bonds between the last two phosphates in the ATP chain. Then the cycle starts all over again! ...
Answer Key for the Supplemental Problem Set #1
... 3. What are the three metabolically irreversible steps of glycolysis? What general type of reaction is catalyzed by these enzymes? Why are these reactions irreversible? Glucose phosphorylation catalyzed by hexokinase; fructose-6-phosphate phosphorylation catalyzed by phosphofructokinase; and phospho ...
... 3. What are the three metabolically irreversible steps of glycolysis? What general type of reaction is catalyzed by these enzymes? Why are these reactions irreversible? Glucose phosphorylation catalyzed by hexokinase; fructose-6-phosphate phosphorylation catalyzed by phosphofructokinase; and phospho ...
Unit 2: Biochem Notes
... - A solution with a pH __________ 7, has more OH- ions than H+ ions, and is basic. - A solution with a pH _________ 7, has more H+ ions than OH- ions, and is acidic. b. buffer – Weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH. Buffers make acidic ...
... - A solution with a pH __________ 7, has more OH- ions than H+ ions, and is basic. - A solution with a pH _________ 7, has more H+ ions than OH- ions, and is acidic. b. buffer – Weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH. Buffers make acidic ...
2401_Ch2.pdf
... Electrically neutral atoms have an equal number of protons and electrons Atomic number: number of protons in an atom (written 11Na) Atomic weight: equal to number of protons and neutrons Isotopes – atoms having the same # of protons & electrons but different # of neutrons (e.g. 12C and 14C) ...
... Electrically neutral atoms have an equal number of protons and electrons Atomic number: number of protons in an atom (written 11Na) Atomic weight: equal to number of protons and neutrons Isotopes – atoms having the same # of protons & electrons but different # of neutrons (e.g. 12C and 14C) ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.