CARBOHYDRATE METABOLISM
... Pyruvate is converted to acetyl CoA by oxidative decarboxylation. This is an irreversible reaction, catalysed by a multi enzyme complex, known as pyruvate dehydrogenase complex (PDH), which is found only in the mitochondria. The enzyme PDH requires five cofactors (coenzymes), namely1. ...
... Pyruvate is converted to acetyl CoA by oxidative decarboxylation. This is an irreversible reaction, catalysed by a multi enzyme complex, known as pyruvate dehydrogenase complex (PDH), which is found only in the mitochondria. The enzyme PDH requires five cofactors (coenzymes), namely1. ...
Light-independent reactions - Mrs Jones A
... Fortunately, some molecules which are found in these reactions can be used to synthesise other molecules which are of use to organisms: one of every six molecules of TP/GALP produced are not recycled as RuBP, but are actually used to make other biological molecules such as saccharides – two molecu ...
... Fortunately, some molecules which are found in these reactions can be used to synthesise other molecules which are of use to organisms: one of every six molecules of TP/GALP produced are not recycled as RuBP, but are actually used to make other biological molecules such as saccharides – two molecu ...
1 - Lone Star College System
... Ions form when an atom gains or loses electrons in its outer energy level to become stable 1) Positive ion—has lost electrons; indicated by superscript positive sign, as in Na+ or Ca2+ 2) Negative ion—has gained electrons; indicated by superscript negative sign, as in Cl– or O2b. An ionic bond is fo ...
... Ions form when an atom gains or loses electrons in its outer energy level to become stable 1) Positive ion—has lost electrons; indicated by superscript positive sign, as in Na+ or Ca2+ 2) Negative ion—has gained electrons; indicated by superscript negative sign, as in Cl– or O2b. An ionic bond is fo ...
Enzymes I – What Is an Enzyme?
... As we saw in the previous lesson on thermodynamics, chemical reactions do not only involve structural changes in molecules, they also involve changes in the amount of energy of reactants and products. Chemical reactions releasing energy, for example, are termed exergonic reactions, while chemical re ...
... As we saw in the previous lesson on thermodynamics, chemical reactions do not only involve structural changes in molecules, they also involve changes in the amount of energy of reactants and products. Chemical reactions releasing energy, for example, are termed exergonic reactions, while chemical re ...
Document
... Sample Problem 22.7 Pathways for Pyruvate Identify the pathway(s) that has each of the following characteristics: a. In this pathway, NAD+ is reduced to NADH + H+. b. The product of this pathway contains three carbon atoms. c. NADH is the oxidizing agent in this anaerobic pathway to give a two-carb ...
... Sample Problem 22.7 Pathways for Pyruvate Identify the pathway(s) that has each of the following characteristics: a. In this pathway, NAD+ is reduced to NADH + H+. b. The product of this pathway contains three carbon atoms. c. NADH is the oxidizing agent in this anaerobic pathway to give a two-carb ...
19 Dr. Nafez Abu Tarboosh Qusai Al Sharef
... So in the solution (cytoplasm) you can find NAD+ or NADH only. -The catalitic part of NAD+ is a carbon in the ring that H- adds to, so it takes electrons. -Where can we find them? Since it takes up a hydrogen we can find it in most dehydrogenases which donates electrons (in the form of hydride ion) ...
... So in the solution (cytoplasm) you can find NAD+ or NADH only. -The catalitic part of NAD+ is a carbon in the ring that H- adds to, so it takes electrons. -Where can we find them? Since it takes up a hydrogen we can find it in most dehydrogenases which donates electrons (in the form of hydride ion) ...
Cellular Respiration and Photosynthesis Notes
... • It powers all cell functions such as building proteins and opening ion channels for active transport Q: How does it work? ...
... • It powers all cell functions such as building proteins and opening ion channels for active transport Q: How does it work? ...
Redox - Plusnet
... Rules for assigning: (these rarely change) F is always -1 O is -2, except in OF2 Group 7 are -1, except with O or F Group 1 metals are +1 Group 2 metals are +2 H is +1, except in hydrides, e.g. NaH Al is +3 The total for an ion is its charge (e.g. -1 for CN-) More electronegative atoms get negative ...
... Rules for assigning: (these rarely change) F is always -1 O is -2, except in OF2 Group 7 are -1, except with O or F Group 1 metals are +1 Group 2 metals are +2 H is +1, except in hydrides, e.g. NaH Al is +3 The total for an ion is its charge (e.g. -1 for CN-) More electronegative atoms get negative ...
Lecture_12
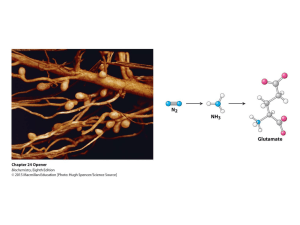
... The reductase and the nitrogenase are iron-sulfur proteins. The reductase, also called the Fe protein, transfers electrons from ferredoxin to the nitrogenase. A site on the nitrogenase called the P cluster accepts the electrons. From the P cluster, the electrons flow to the FeMo cofactor, the site ...
... The reductase and the nitrogenase are iron-sulfur proteins. The reductase, also called the Fe protein, transfers electrons from ferredoxin to the nitrogenase. A site on the nitrogenase called the P cluster accepts the electrons. From the P cluster, the electrons flow to the FeMo cofactor, the site ...
control of intermediary metabolism
... AEROBIC METABOLISM PYRUVIC ACID (3 C FRAGMENT) ENTERS MITOCHONDRIA COMBINES WITH COENZYME A LOOSING A CO2 AND BECOMING ACETYL COENZYME A (2 C FRAGMENT) THIS FRAGMENT ENTERS A CYCLIC REACTION SCHEME, THE CITRIC ACID CYCLE, ATP IS PRODUCED PRODUCTS OF THE CITRIC ACID CYCLE ENTER THE ELECTRON ...
... AEROBIC METABOLISM PYRUVIC ACID (3 C FRAGMENT) ENTERS MITOCHONDRIA COMBINES WITH COENZYME A LOOSING A CO2 AND BECOMING ACETYL COENZYME A (2 C FRAGMENT) THIS FRAGMENT ENTERS A CYCLIC REACTION SCHEME, THE CITRIC ACID CYCLE, ATP IS PRODUCED PRODUCTS OF THE CITRIC ACID CYCLE ENTER THE ELECTRON ...
emboj7601444-sup
... initiated by the addition of IV-CoA. The rate of the acyltransfer reaction at 30°C was determined by monitoring the decline of absorbance at 340 nm. All kinetic data were fitted by nonlinear regression analysis using KaleidaGraph (Synergy Software, Essex Junction, VT). ...
... initiated by the addition of IV-CoA. The rate of the acyltransfer reaction at 30°C was determined by monitoring the decline of absorbance at 340 nm. All kinetic data were fitted by nonlinear regression analysis using KaleidaGraph (Synergy Software, Essex Junction, VT). ...
14 - Ch 22 Respiration Exercise Multiple-choice questions (p. 22-35)
... Pyruvate is the product of glycolysis. (1) As the production of pyruvate is greatly reduced after treating with drug X. (1) Glycolysis was inhibited in this case. (1) (b) drug Y inhibited Krebs cycle (1) when the respiratory pathway is halted at Krebs cycle, pyruvate will not be metabolised (1) but ...
... Pyruvate is the product of glycolysis. (1) As the production of pyruvate is greatly reduced after treating with drug X. (1) Glycolysis was inhibited in this case. (1) (b) drug Y inhibited Krebs cycle (1) when the respiratory pathway is halted at Krebs cycle, pyruvate will not be metabolised (1) but ...
Coenzymes
... Cytochromes • Heme-containing coenzymes whose Fe(III) undergoes reversible one-electron reduction • Cytochromes a,b and c have different visible absorption spectra and heme prosthetic groups • Electron transfer potential varies among different cytochromes due to the different protein environment of ...
... Cytochromes • Heme-containing coenzymes whose Fe(III) undergoes reversible one-electron reduction • Cytochromes a,b and c have different visible absorption spectra and heme prosthetic groups • Electron transfer potential varies among different cytochromes due to the different protein environment of ...
1. What is the collective term for all of the chemical processes
... 42. Which of the following is the proper order of DNA Replication/Protein Synthesis A) Transcription, Translation, Proteins to form new DNA from existing DNA B) Protein placement, Transcription, Translation C) Translation, Transcription, DNA polymerase formation D) Proteins to form new DNA from exis ...
... 42. Which of the following is the proper order of DNA Replication/Protein Synthesis A) Transcription, Translation, Proteins to form new DNA from existing DNA B) Protein placement, Transcription, Translation C) Translation, Transcription, DNA polymerase formation D) Proteins to form new DNA from exis ...
New Reaction Chemistries
... Safety BOC/CBZ/Carbamate Cleavage Redesign protecting groups – enzyme cleavable (penicillin acylase) Aqueous versus organic (recycle) Addition to imines (oxidation) Alternative to Coenzyme A C-C bond formation ...
... Safety BOC/CBZ/Carbamate Cleavage Redesign protecting groups – enzyme cleavable (penicillin acylase) Aqueous versus organic (recycle) Addition to imines (oxidation) Alternative to Coenzyme A C-C bond formation ...
Chapter 8: An Introduction to Metabolism - Biology E
... …and energy is released. For the exergonic reaction ADP + Pi g ATP + H2O, ΔG is 7.3 kcal/mol (30.5 kJ/mol). 13. What is energy coupling? Energy coupling is the use of an exergonic process to drive an endergonic one. ATP is responsible for mediating most energy coupling in cells, and in most cases i ...
... …and energy is released. For the exergonic reaction ADP + Pi g ATP + H2O, ΔG is 7.3 kcal/mol (30.5 kJ/mol). 13. What is energy coupling? Energy coupling is the use of an exergonic process to drive an endergonic one. ATP is responsible for mediating most energy coupling in cells, and in most cases i ...
... acetyl CoA cannot be used to produce pyruvate which could be used, via gluconeogenesis, to produce glucose. 12. (14 pts) Pick any coordinately regulated step in glucogen or glucose metabolism and briefly describe (use the back of the previous page if you need additional room): i) How it is regulated ...
Anaerobic Fermentation
... Very slow energy production Produces 36 ATP per glucose Anaerobic Fermentation (AF) Doesn't require O 2 Fast energy production Produces only 2 ATP per glucose ...
... Very slow energy production Produces 36 ATP per glucose Anaerobic Fermentation (AF) Doesn't require O 2 Fast energy production Produces only 2 ATP per glucose ...
Review for Chapter 1
... What are the different types of lipids and how are they different? What makes up a protein and what do they look like? What bonds the parts together? What is an enzyme? What makes up a nucleic acid and what do they look like? What is a nucleotide made of? What’s the difference between DNA and RNA? W ...
... What are the different types of lipids and how are they different? What makes up a protein and what do they look like? What bonds the parts together? What is an enzyme? What makes up a nucleic acid and what do they look like? What is a nucleotide made of? What’s the difference between DNA and RNA? W ...
Pathways of Carbohydrate and Lipid Metabolism Glycolysis • Is the
... • One thing which the diagram does not show is the production of 2 CO2 molecules for each acetylCoA that enters the cycle. They are release at the same time/place as the first two NADH molecules ...
... • One thing which the diagram does not show is the production of 2 CO2 molecules for each acetylCoA that enters the cycle. They are release at the same time/place as the first two NADH molecules ...
Media:ATPsynthase
... • Hydrophobic pocket between beta and gamma subunit • Gamma – Because beta most likely more highly conserved – Alpha, beta in larger scale project ...
... • Hydrophobic pocket between beta and gamma subunit • Gamma – Because beta most likely more highly conserved – Alpha, beta in larger scale project ...
Ch. 5 The Working Cell
... and yields products rich in potential energy – The reactants contain little energy in the beginning, but ...
... and yields products rich in potential energy – The reactants contain little energy in the beginning, but ...
Test Review Answers - Northwest ISD Moodle
... 13. What element do nucleic acids and proteins contain that carbs and lipids do not? Phosphorous 14. Explain how energy is released from an ATP molecule. In order to release energy, bonds must be broken between Atoms. This breaking releases energy! 15. Lipids and carbohydrates both contain energy. W ...
... 13. What element do nucleic acids and proteins contain that carbs and lipids do not? Phosphorous 14. Explain how energy is released from an ATP molecule. In order to release energy, bonds must be broken between Atoms. This breaking releases energy! 15. Lipids and carbohydrates both contain energy. W ...
K,Mg,Ca,Na… 0,4%
... 6.2. Nutrient uptake Passive (simple) diffusion Nutrients moving from a region of higher concentration to one of lower (influence of concentration gradient) – ions, glycerol, O2, CO2 – no energy consumption Osmosis Some solvent molecules and water move across membrane Isotonic – Hypotonic - Hyp ...
... 6.2. Nutrient uptake Passive (simple) diffusion Nutrients moving from a region of higher concentration to one of lower (influence of concentration gradient) – ions, glycerol, O2, CO2 – no energy consumption Osmosis Some solvent molecules and water move across membrane Isotonic – Hypotonic - Hyp ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.