Molecules of Life
... Substrate ( reactant) fits into the active site of the enzyme 2) The enzyme breaks/forms bonds and releases the products 3) The enzyme can then be used again with another substrate ...
... Substrate ( reactant) fits into the active site of the enzyme 2) The enzyme breaks/forms bonds and releases the products 3) The enzyme can then be used again with another substrate ...
Metabolism
... • Split to form 2 Glyceraldehyde 3phosphate • Final Products are: – 2 Pyruvic Acid (C3H4O3) • Compare to original glucose - C6H12O6 ...
... • Split to form 2 Glyceraldehyde 3phosphate • Final Products are: – 2 Pyruvic Acid (C3H4O3) • Compare to original glucose - C6H12O6 ...
Sample Exam #1 ( file)
... B. Prefers to give up 3 electrons to form ions. C. Has 2 electrons in its outer shell so it forms 2 chemical bonds. D. Is never found in inorganic molecules but always found in organic molecules. ...
... B. Prefers to give up 3 electrons to form ions. C. Has 2 electrons in its outer shell so it forms 2 chemical bonds. D. Is never found in inorganic molecules but always found in organic molecules. ...
3 sources of energy during excercise
... oxygen during excercise... *Body compensates for the lack of oxygen by a process called Anaerobic fermentation that carries out a series of chemical reactions that produce ATP from glucose in the absence of O 2 *Fermentation allows glycolysis to continue making ATP when oxygen is not available ...
... oxygen during excercise... *Body compensates for the lack of oxygen by a process called Anaerobic fermentation that carries out a series of chemical reactions that produce ATP from glucose in the absence of O 2 *Fermentation allows glycolysis to continue making ATP when oxygen is not available ...
ch 9ppt
... electrons passed from one electron carrier to next in mitochondrial membrane (ETC) transport proteins in membrane pump H+ across inner membrane to intermembrane space ...
... electrons passed from one electron carrier to next in mitochondrial membrane (ETC) transport proteins in membrane pump H+ across inner membrane to intermembrane space ...
Metabolism Part II: The tricarboxylic acid (TCA), citric acid, or Krebs
... fact a cycle of reactions that was responsible for the aerobic oxidation of fuel molecules. Concern over whether citric acid (or more accurately the citrate ion) was the first product of this cycle led Krebs to propose calling this sequence of enzyme-catalyzed reactions the tricarboxylic acid cycle. ...
... fact a cycle of reactions that was responsible for the aerobic oxidation of fuel molecules. Concern over whether citric acid (or more accurately the citrate ion) was the first product of this cycle led Krebs to propose calling this sequence of enzyme-catalyzed reactions the tricarboxylic acid cycle. ...
Document
... a. number of electrons b. number of valence electrons c. number of protons d. number of neutrons _____ 16. Which group on the periodic table contains elements that do not normally form chemical bonds? a. Group 2 c. Group 10 b. Group 6 d. Group 18 17. The outermost energy level of an atom is consider ...
... a. number of electrons b. number of valence electrons c. number of protons d. number of neutrons _____ 16. Which group on the periodic table contains elements that do not normally form chemical bonds? a. Group 2 c. Group 10 b. Group 6 d. Group 18 17. The outermost energy level of an atom is consider ...
Cellular Respiration
... cycle is a series of reactions that produce energy-storing molecules during aerobic respiration. •Electron Transport Chain During aerobic respiration, large amounts of ATP are made in an electron transport chain. ...
... cycle is a series of reactions that produce energy-storing molecules during aerobic respiration. •Electron Transport Chain During aerobic respiration, large amounts of ATP are made in an electron transport chain. ...
Cellular Respiration What is Cellular Respiration?
... cycle is a series of reactions that produce energy-storing molecules during aerobic respiration. •Electron Transport Chain During aerobic respiration, large amounts of ATP are made in an electron transport chain. ...
... cycle is a series of reactions that produce energy-storing molecules during aerobic respiration. •Electron Transport Chain During aerobic respiration, large amounts of ATP are made in an electron transport chain. ...
Enzymes - NVHSIntroBioPiper1
... Question: How do enzymes speed up reactions? Answer: They lower activation energy, making it easier for the chemical reaction to occur ...
... Question: How do enzymes speed up reactions? Answer: They lower activation energy, making it easier for the chemical reaction to occur ...
First of all, do you know any methods to check
... Error in AES: analysis: < 15%, Error within a few % can be achieved with better standards and calibration. Take care Sensitivities Si for peak to peak height of differentiated Auger peak different from the one for original Auger peak(with background subtraction) ...
... Error in AES: analysis: < 15%, Error within a few % can be achieved with better standards and calibration. Take care Sensitivities Si for peak to peak height of differentiated Auger peak different from the one for original Auger peak(with background subtraction) ...
H 2 O
... The Warburg Effect • In oncology, the Warburg effect is that most cancer cells predominantly produce energy by a high rate of glycolysis followed by lactic acid fermentation in the cytosol, rather than by a comparatively low rate of glycolysis followed by oxidation of pyruvate in mitochondria like ...
... The Warburg Effect • In oncology, the Warburg effect is that most cancer cells predominantly produce energy by a high rate of glycolysis followed by lactic acid fermentation in the cytosol, rather than by a comparatively low rate of glycolysis followed by oxidation of pyruvate in mitochondria like ...
File
... • Anion—atom that gains electrons (net negative charge) • Cation—atom that loses an electron (net positive charge) • Ions with opposite charges are attracted to each other Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... • Anion—atom that gains electrons (net negative charge) • Cation—atom that loses an electron (net positive charge) • Ions with opposite charges are attracted to each other Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
Photosynthesis Cellular Respiration
... • The process in which one molecule of glucose is broken in half producing two molecules of pyruvic acid. Energy yield from glycolysis is small. • Doesn’t require oxygen. • Occurs in the cytoplasm. ...
... • The process in which one molecule of glucose is broken in half producing two molecules of pyruvic acid. Energy yield from glycolysis is small. • Doesn’t require oxygen. • Occurs in the cytoplasm. ...
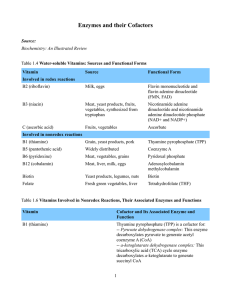
Enzymes and their Cofactors Source: Biochemistry: An Illustrated
... degrades odd-numbered fatty acids -- Methionine synthase/homocysteine methyltransferase: This enzyme transfers a methyl group from 5-methyltetrahydrofolate (N5-methylTHF) onto homocysteine to form methionine. Methionine reacts with adenosine triphosphate (ATP) to generate S-adenosyl methionine (SAM) ...
... degrades odd-numbered fatty acids -- Methionine synthase/homocysteine methyltransferase: This enzyme transfers a methyl group from 5-methyltetrahydrofolate (N5-methylTHF) onto homocysteine to form methionine. Methionine reacts with adenosine triphosphate (ATP) to generate S-adenosyl methionine (SAM) ...
An Overview of Cellular Respiration 2017
... 3. The first stage of cellular respiration is glycolysis. It occurs in the cytoplasm and produces 2 net ATP molecules. ...
... 3. The first stage of cellular respiration is glycolysis. It occurs in the cytoplasm and produces 2 net ATP molecules. ...
Mitochondria, Chloroplasts, and Peroxisomes
... molecular oxygen is coupled to the transfer of protons from the mitochondrial matrix to the intermembrane space. Since protons are charged particles, this transfer establishes an electric potential across the inner membrane, with the matrix being negative. During protein import, this electric potent ...
... molecular oxygen is coupled to the transfer of protons from the mitochondrial matrix to the intermembrane space. Since protons are charged particles, this transfer establishes an electric potential across the inner membrane, with the matrix being negative. During protein import, this electric potent ...
First test material Study guide
... What are the two alternative routes of fatty acid oxidation? Give a brief overview of each and explain when they may come in to use. Answer: 1. Peroxisomal B-Oxidation of Fatty Acids Very-long chain fatty acids (24-26 C) are oxidized exclusively in peroxisomes. This process is very similar to mitoch ...
... What are the two alternative routes of fatty acid oxidation? Give a brief overview of each and explain when they may come in to use. Answer: 1. Peroxisomal B-Oxidation of Fatty Acids Very-long chain fatty acids (24-26 C) are oxidized exclusively in peroxisomes. This process is very similar to mitoch ...
Metabolic Model Describing Growth of Substrate Uptake
... medium is considerably greater to that of the salts medium. The observed specific oxygen uptake rate k3 does not reach a maximum for higher concentration of of oxygen because the growth is performed under limiting conditions of oxygen(0.5%). ...
... medium is considerably greater to that of the salts medium. The observed specific oxygen uptake rate k3 does not reach a maximum for higher concentration of of oxygen because the growth is performed under limiting conditions of oxygen(0.5%). ...
Ch. 5 Enzyme Review
... a. Higher temperatures allow greater contact between enzymes and substrates; therefore, the higher the temperature the better the enzyme will function. b. Enzymes cannot function at a pH lower than 6. c. Most coenzymes are inorganic substances such as ions of iron or potassium. d. Excessive salt ion ...
... a. Higher temperatures allow greater contact between enzymes and substrates; therefore, the higher the temperature the better the enzyme will function. b. Enzymes cannot function at a pH lower than 6. c. Most coenzymes are inorganic substances such as ions of iron or potassium. d. Excessive salt ion ...
... 12. (12 pts) Please do one of the following two choices. Please indicate your choice: Choice A: A fictitious enzyme can utilize the energy associate with a glucose gradient across the cell membrane to synthesize ATP from ADP and Pi. The standard free energy for hydrolysis of ATP is -30 kJ/mol. i) Wh ...
Metabolic Model Describing Growth of Substrate Uptake
... medium is considerably greater to that of the salts medium. The observed specific oxygen uptake rate k3 does not reach a maximum for higher concentration of of oxygen because the growth is performed under limiting conditions of oxygen(0.5%). ...
... medium is considerably greater to that of the salts medium. The observed specific oxygen uptake rate k3 does not reach a maximum for higher concentration of of oxygen because the growth is performed under limiting conditions of oxygen(0.5%). ...
Biology Answer Key
... Why and where is aldosterone produced? What factors stimulate its secretion? From Adrenal cortex, so that it causes reabsorption of Na+ and water from the distal parts of the tubule. This increases GFR and blood pressure Low GFR activates JG cells to release rennin which converts angiotensinogen to ...
... Why and where is aldosterone produced? What factors stimulate its secretion? From Adrenal cortex, so that it causes reabsorption of Na+ and water from the distal parts of the tubule. This increases GFR and blood pressure Low GFR activates JG cells to release rennin which converts angiotensinogen to ...
ppt - UCLA Chemistry and Biochemistry
... • Electrostatic catalysis: the enzyme uses charge-charge interactions in catalysis • Preferential binding of transition state: binding interactions between the enzyme and TS are maximized; they are greater than those in the enzyme-substrate or enzyme-product complexes • General acid and general base ...
... • Electrostatic catalysis: the enzyme uses charge-charge interactions in catalysis • Preferential binding of transition state: binding interactions between the enzyme and TS are maximized; they are greater than those in the enzyme-substrate or enzyme-product complexes • General acid and general base ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.