Protein engineering of aldolase: Directed evolution
... and often do not exhibit properties required for in vitro applications. Protein engineering aims to create novel enzymes for use in medicine and industry by using the techniques of mutagenesis, protein expression, protein purification, and protein characterisation. We are interested in the protein e ...
... and often do not exhibit properties required for in vitro applications. Protein engineering aims to create novel enzymes for use in medicine and industry by using the techniques of mutagenesis, protein expression, protein purification, and protein characterisation. We are interested in the protein e ...
The Molecules of Cells
... – The shape of a protein molecule is critical to its function – Protein molecules have at least 3 levels of organization • Primary- shape held together by bonds between r groups chain of amino acids linked by peptide bonds • Secondary-coiling or folding of the primary structure to form a helix or a ...
... – The shape of a protein molecule is critical to its function – Protein molecules have at least 3 levels of organization • Primary- shape held together by bonds between r groups chain of amino acids linked by peptide bonds • Secondary-coiling or folding of the primary structure to form a helix or a ...
15.2 Electrons and Chemical Bonds
... was why elements combined with other elements only in particular ratios (or not at all). For example, why do two hydrogen atoms bond with one oxygen atom to make water? Why isn’t there a molecule with three (H3O) or even four (H4O) hydrogen atoms? Why does sodium chloride have a precise ratio of one ...
... was why elements combined with other elements only in particular ratios (or not at all). For example, why do two hydrogen atoms bond with one oxygen atom to make water? Why isn’t there a molecule with three (H3O) or even four (H4O) hydrogen atoms? Why does sodium chloride have a precise ratio of one ...
Increased expression of the F1Fo ATP synthase in response to iron
... (17). The dependence of the increase on iron availability may reflect the emergence of a primitive enzyme performing the oxidation/reduction of Fe2+ and evolving to carry out oxidative phosphorylation. The original high affinity of the enzyme for iron has been maintained in iron metabolism. The F1Fo ...
... (17). The dependence of the increase on iron availability may reflect the emergence of a primitive enzyme performing the oxidation/reduction of Fe2+ and evolving to carry out oxidative phosphorylation. The original high affinity of the enzyme for iron has been maintained in iron metabolism. The F1Fo ...
Bacterial enzymes that can deglycate glucose
... of the Schiff base resulting from the condensation of fructose 6phosphate and ammonia. Both glucoselysine and fructoselysine are compounds that are abundantly present in free form in vegetable and fruits, where their concentration may reach approx. 7 % of the fresh mass, i.e. 400 mM [15]. Thus it is ...
... of the Schiff base resulting from the condensation of fructose 6phosphate and ammonia. Both glucoselysine and fructoselysine are compounds that are abundantly present in free form in vegetable and fruits, where their concentration may reach approx. 7 % of the fresh mass, i.e. 400 mM [15]. Thus it is ...
CHAPTER 26: Lipid Metabolism - Richest energy source
... - we said acetyl CoA formed enters the Krebs cycle, or is used to form Ketone bodies - ketaone bodies are formed from the buildup of acetyl coA in the body with depletion of oxaloacetate = krebs cycle can’t start - caused by diabetes or diet low in carbohydrates (fatty acid oxidation increases to ...
... - we said acetyl CoA formed enters the Krebs cycle, or is used to form Ketone bodies - ketaone bodies are formed from the buildup of acetyl coA in the body with depletion of oxaloacetate = krebs cycle can’t start - caused by diabetes or diet low in carbohydrates (fatty acid oxidation increases to ...
Chapter 5: The Working Cell
... • Small polar uncharged molecules can pass through easily (e.g.: H2O , CO2) • Large polar uncharged molecules pass with difficulty (e.g.: glucose) 3. Ionic (Hydrophilic) Molecules • Charged ions or particles cannot get through (e.g.: ions such as Na+ , K+ , Cl- ) ...
... • Small polar uncharged molecules can pass through easily (e.g.: H2O , CO2) • Large polar uncharged molecules pass with difficulty (e.g.: glucose) 3. Ionic (Hydrophilic) Molecules • Charged ions or particles cannot get through (e.g.: ions such as Na+ , K+ , Cl- ) ...
I. Energy
... • Large polar uncharged molecules pass with difficulty (e.g.: glucose) 3. Ionic (Hydrophilic) Molecules • Charged ions or particles cannot get through (e.g.: ions such as Na+ , K+ , Cl- ) ...
... • Large polar uncharged molecules pass with difficulty (e.g.: glucose) 3. Ionic (Hydrophilic) Molecules • Charged ions or particles cannot get through (e.g.: ions such as Na+ , K+ , Cl- ) ...
Pentose Phosphate Pathway
... ENTNER-DUDOROFF PATHWAY • The Entner-Doudoroff pathway yields one ATP and two NADPH molecules from one glucose molecule. • Uses 4 enzymes that are different from EMP 1 Glucose 2 pyruvate + 1 ATP + 1 NADH + 1 NADPH Bacteria: Pseudomonas, Rhizobium, Azotobacter, Agrobacterium, Enterococcus faecalis ...
... ENTNER-DUDOROFF PATHWAY • The Entner-Doudoroff pathway yields one ATP and two NADPH molecules from one glucose molecule. • Uses 4 enzymes that are different from EMP 1 Glucose 2 pyruvate + 1 ATP + 1 NADH + 1 NADPH Bacteria: Pseudomonas, Rhizobium, Azotobacter, Agrobacterium, Enterococcus faecalis ...
The citric acid cycle is the
... intermediate and is utilized in this step of the TCA cycle to drive the phosphorylation of GDP to GTP (in mammals) or ADP to ATP (in plants and bacteria). • The reaction is catalyzed by succinylCoA synthetase, sometimes called succinate Thiokinase琥珀酸硫激酶. ...
... intermediate and is utilized in this step of the TCA cycle to drive the phosphorylation of GDP to GTP (in mammals) or ADP to ATP (in plants and bacteria). • The reaction is catalyzed by succinylCoA synthetase, sometimes called succinate Thiokinase琥珀酸硫激酶. ...
Chapter 8 Study Guide
... the pigment that produces the yellow and orange fall leaf colors these organisms get their energy from food by cellular respiration this is an enzyme assisted anaerobic process that breaks down glucose to pyruvate this are light absorbing substances this is a metabolic process that requires oxygen i ...
... the pigment that produces the yellow and orange fall leaf colors these organisms get their energy from food by cellular respiration this is an enzyme assisted anaerobic process that breaks down glucose to pyruvate this are light absorbing substances this is a metabolic process that requires oxygen i ...
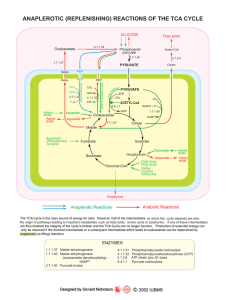
TCA (Krebs) Cycle
... or citric acid cycle: located in mitochondrion; common 8-Rx oxidative pathway for all fuels. Two major metabolic roles: energy production and biosynthesis. 4 oxidative steps: capture high DG e– in 3 NADH and 1 FADH2; transfer to ETS for ATP. Substrate level phosphorylation: 1 GTP. 8 reaction ...
... or citric acid cycle: located in mitochondrion; common 8-Rx oxidative pathway for all fuels. Two major metabolic roles: energy production and biosynthesis. 4 oxidative steps: capture high DG e– in 3 NADH and 1 FADH2; transfer to ETS for ATP. Substrate level phosphorylation: 1 GTP. 8 reaction ...
Bacterial Physiology and Metabolism
... culture, but is found in sediments in the coastal waters of Namibia measuring up to 0.75 mm in size, which is about 100 times bigger than a normal sized bacterium. In addition to the acidophilic and neutrophilic sulfur bacteria, alkalophilic sulfur bacteria thrive in alkaline soda lakes. These are s ...
... culture, but is found in sediments in the coastal waters of Namibia measuring up to 0.75 mm in size, which is about 100 times bigger than a normal sized bacterium. In addition to the acidophilic and neutrophilic sulfur bacteria, alkalophilic sulfur bacteria thrive in alkaline soda lakes. These are s ...
Document
... • katal (International system of units SI) – the amount of enzyme that converts 1 mole of substrate per second ...
... • katal (International system of units SI) – the amount of enzyme that converts 1 mole of substrate per second ...
respiration
... • Net Reaction Appears as the Reverse of PS • The individual reactions that occur to achieve the net effect are entirely different ...
... • Net Reaction Appears as the Reverse of PS • The individual reactions that occur to achieve the net effect are entirely different ...
Answer Set 3
... oxaloacetate from pyruvate and CO2 by pyruvate carboxylase and (2) the formation of phosphoenolpyruvate from oxaloacetate and GTP by phosphoenolpyruvate carboxykinase. The formation of fructose-1,6-bisphosphate by phosphofructokinase is bypassed by fructose-1,6-bisphosphatase in gluconeogenesis, whi ...
... oxaloacetate from pyruvate and CO2 by pyruvate carboxylase and (2) the formation of phosphoenolpyruvate from oxaloacetate and GTP by phosphoenolpyruvate carboxykinase. The formation of fructose-1,6-bisphosphate by phosphofructokinase is bypassed by fructose-1,6-bisphosphatase in gluconeogenesis, whi ...
Week 1 Pre-Lecture Slides
... What assumptions are you making in your calculation? • Why is RNA more like protein than DNA? • Why is it more like DNA than protein? • What extra work must you do to complete a Bio200 exam? • Is this a good or bad exam system? Why? ...
... What assumptions are you making in your calculation? • Why is RNA more like protein than DNA? • Why is it more like DNA than protein? • What extra work must you do to complete a Bio200 exam? • Is this a good or bad exam system? Why? ...
Ch. 5 Presentation
... 5.5 Water balance between cells and their surroundings is crucial to organisms How will animal cells be affected when placed into solutions of various tonicities? When an animal cell is placed into – an isotonic solution, the concentration of solute is the same on both sides of a membrane, and th ...
... 5.5 Water balance between cells and their surroundings is crucial to organisms How will animal cells be affected when placed into solutions of various tonicities? When an animal cell is placed into – an isotonic solution, the concentration of solute is the same on both sides of a membrane, and th ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.