Spectroscopy
... The constant R is called the Rydberg constant. Planck’s constant is h; the speed of light is c. In the Bohr Model, the Rydberg constant is predicted to be R 1.0975 x10 7 m 1 . We shall determine R experimentally by observing the emission spectrum of Hydrogen. The four bright spectral lines in the ...
... The constant R is called the Rydberg constant. Planck’s constant is h; the speed of light is c. In the Bohr Model, the Rydberg constant is predicted to be R 1.0975 x10 7 m 1 . We shall determine R experimentally by observing the emission spectrum of Hydrogen. The four bright spectral lines in the ...
Chapter 5 Electrons In Atoms 5.1 Models of the Atom The
... The Development of Atomic Models The model for the atom consisted of protons and neutrons making up a nucleus surrounded by electrons. The Bohr Model Bohr proposed that an ______________ is found only in specific circular paths, or orbits, around the nucleus. The fixed energies an electron can have ...
... The Development of Atomic Models The model for the atom consisted of protons and neutrons making up a nucleus surrounded by electrons. The Bohr Model Bohr proposed that an ______________ is found only in specific circular paths, or orbits, around the nucleus. The fixed energies an electron can have ...
the Bohr`s atom model - Latin-American Journal of Physics Education
... around the 1913 whereas De Broglie published his ideas approximately 10 years later. Bohr himself used the quantization of the angular moment as an ad hoc condition to reproduce the formula of Balmer in the first part of his famous trilogy [9]. In both following parts he tried to look for a justific ...
... around the 1913 whereas De Broglie published his ideas approximately 10 years later. Bohr himself used the quantization of the angular moment as an ad hoc condition to reproduce the formula of Balmer in the first part of his famous trilogy [9]. In both following parts he tried to look for a justific ...
Early Quantum Theory and Models of the Atom
... • Bohr studied Rutherford’s planetary model and found it had validity • But to make it work the newly developing quantum theory would have to be incorporated • Plank and Einstein had shown that in heated solids, the energy of oscillating electric charges must change from one discrete energy state to ...
... • Bohr studied Rutherford’s planetary model and found it had validity • But to make it work the newly developing quantum theory would have to be incorporated • Plank and Einstein had shown that in heated solids, the energy of oscillating electric charges must change from one discrete energy state to ...
The Quantum Mechanical Behavior of Light and Matter
... energy/area/time (energy flux) at distance r = L/4πr ...
... energy/area/time (energy flux) at distance r = L/4πr ...
Chapter 28 Atoms
... planetary picture of an atom as in the Bohr model. In particular, the radius of the electron orbit is not like the radius of the orbit of a planet about the sun. In wave particle nature matter means that it is impossible to know both the position and momentum of an electron at the same time. Thus th ...
... planetary picture of an atom as in the Bohr model. In particular, the radius of the electron orbit is not like the radius of the orbit of a planet about the sun. In wave particle nature matter means that it is impossible to know both the position and momentum of an electron at the same time. Thus th ...
Matter Unit
... distances from the nucleus. RULE 2: Atoms radiate energy when an electron jumps from a higher-energy orbit to a lower-energy orbit. Also, an atom absorbs energy when an electron gets boosted from a low- ...
... distances from the nucleus. RULE 2: Atoms radiate energy when an electron jumps from a higher-energy orbit to a lower-energy orbit. Also, an atom absorbs energy when an electron gets boosted from a low- ...
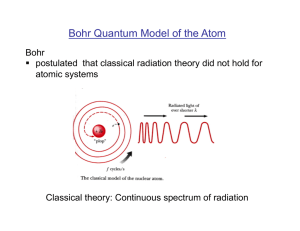
Bohr Quantum Model of the Atom
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
... § postulated that the electron orbital momentum is quantized Justification of Bohr’s postulates: comparison with experimental observations! ...
Ch 6 Outline
... A laser used in eye surgery to fuse detached retinas produces radiation with a wavelength of 640.0 nm. Calculate the frequency of this radiation. ...
... A laser used in eye surgery to fuse detached retinas produces radiation with a wavelength of 640.0 nm. Calculate the frequency of this radiation. ...
Electron Orbits
... on its orbit: potential energy: total energy: binding energy experimental result for the binding energy of Hydrogen (H): estimate of the radius of the electron orbit: ...
... on its orbit: potential energy: total energy: binding energy experimental result for the binding energy of Hydrogen (H): estimate of the radius of the electron orbit: ...
Class 27: The Bohr model for the atom
... which reveals how the shortest possible wavelength of photons emitted by a transition in a hydrogen atom is related to the circumference of the orbit in the ground state. One physical interpretation of the fine structure constant arises from consideration of the electron. The electron is a point-lik ...
... which reveals how the shortest possible wavelength of photons emitted by a transition in a hydrogen atom is related to the circumference of the orbit in the ground state. One physical interpretation of the fine structure constant arises from consideration of the electron. The electron is a point-lik ...
Chapter 2 Part 1 ppt
... • 4r2R2: probability of finding electron at a given distance from nucleus, summed over all angles • Probability of finding the electron at a certain distance from the nucleus is not equal to the probability of finding the electron at a certain point at that distance from the nucleus. • There is a w ...
... • 4r2R2: probability of finding electron at a given distance from nucleus, summed over all angles • Probability of finding the electron at a certain distance from the nucleus is not equal to the probability of finding the electron at a certain point at that distance from the nucleus. • There is a w ...
Arrangement of Electrons In Atoms
... Arrangement of Electrons In Atoms Objectives 1. Explain relationship between speed, wavelength, frequency of electromagnetic radiation 2. Discuss the wave-particle duality of light 3. How the photoelectric effect and line-emission spectrum of hydrogen helped develop the atomic model 4. Describe the ...
... Arrangement of Electrons In Atoms Objectives 1. Explain relationship between speed, wavelength, frequency of electromagnetic radiation 2. Discuss the wave-particle duality of light 3. How the photoelectric effect and line-emission spectrum of hydrogen helped develop the atomic model 4. Describe the ...
Bohr model
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
Atomic Structure
... 1. The Bohr model of the atom was the first quantum mechanical model of the atom. a. Bohr postulated that a hydrogen atom could only exist without radiating in one of a set of stationary states. Explain what is meant by this postulate. b. Bohr related his postulate to the classical picture of a hydr ...
... 1. The Bohr model of the atom was the first quantum mechanical model of the atom. a. Bohr postulated that a hydrogen atom could only exist without radiating in one of a set of stationary states. Explain what is meant by this postulate. b. Bohr related his postulate to the classical picture of a hydr ...
3.3 Review Name________________________________ Period_______Date_____________________
... contribution to the quantum theory listed below. Each name may be used more than once. Planck ...
... contribution to the quantum theory listed below. Each name may be used more than once. Planck ...
Document
... contribution to the quantum theory listed below. Each name may be used more than once. Planck ...
... contribution to the quantum theory listed below. Each name may be used more than once. Planck ...
Chapter 13 – Electrons in Atoms
... Atoms/elements emit light when the electrons are excited (first absorb then emit energy in the form of light) at specific frequencies. ...
... Atoms/elements emit light when the electrons are excited (first absorb then emit energy in the form of light) at specific frequencies. ...
Quantum Mechanical Model of the Atom
... MODEL OF THE ATOM ESSENTIAL QUESTION: WHAT IS THE CURRENT MODEL OF THE ATOM? ...
... MODEL OF THE ATOM ESSENTIAL QUESTION: WHAT IS THE CURRENT MODEL OF THE ATOM? ...
Tutorial 7
... Energy of the transition from the first energy level to the third energy level in a hydrogen atom. ...
... Energy of the transition from the first energy level to the third energy level in a hydrogen atom. ...
Bohr`s Model of the Atom - Mr. Walsh`s AP Chemistry
... into a single theory. Bohr hypothesized that electrons moved around the nucleus as in Rutherford’s model, but that these electrons had only certain allowed quantum values of energy, which could be described by a quantum number (n). The value of that quantum number was the same n as in Rydberg’s equa ...
... into a single theory. Bohr hypothesized that electrons moved around the nucleus as in Rutherford’s model, but that these electrons had only certain allowed quantum values of energy, which could be described by a quantum number (n). The value of that quantum number was the same n as in Rydberg’s equa ...
$doc.title
... knocked out. For high Z atoms, these are very tightly bound states (K shells), so require high energies (many keV) to eject them Spectrum shows sharp peaks, due to emission of photons by outer electrons falling to vacated core states. Energy (frequency) is characteristic of element. N.B. Lower ene ...
... knocked out. For high Z atoms, these are very tightly bound states (K shells), so require high energies (many keV) to eject them Spectrum shows sharp peaks, due to emission of photons by outer electrons falling to vacated core states. Energy (frequency) is characteristic of element. N.B. Lower ene ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.