The Modern Nuclear Atom
... and protons but different numbers of neutrons. • Atomic number = number of protons • Mass number = the sum of the protons and neutrons in an atom’s nucleus ...
... and protons but different numbers of neutrons. • Atomic number = number of protons • Mass number = the sum of the protons and neutrons in an atom’s nucleus ...
Quantum Mechanics
... measure either the energy of anything at the same instant as time or the momentum at the same instant of position This means that if something is moving, there is a real possibility that you do not know where it actually is This is typically a very small number (10-30) for ordinary objects, but ...
... measure either the energy of anything at the same instant as time or the momentum at the same instant of position This means that if something is moving, there is a real possibility that you do not know where it actually is This is typically a very small number (10-30) for ordinary objects, but ...
Chapter 5 Sec. 2 Bohr`s Model and the Quantum Mechanical Model
... o In 1924, a French physics student named Louis de Broglie explained the fixed energy levels of Bohr’s model. He explained that electrons can act like _____________________________. He also showed that electrons on circular orbits can only have _____________________ numbers of wavelengths. o de ...
... o In 1924, a French physics student named Louis de Broglie explained the fixed energy levels of Bohr’s model. He explained that electrons can act like _____________________________. He also showed that electrons on circular orbits can only have _____________________ numbers of wavelengths. o de ...
Modern physics 2330
... 5- (..) A black hole is an object of sufficiently high density can trap light forever. 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed of drop. 7- (..) Thompson cloud chamber experiment is inaccurate because the theory applies only to a single particl ...
... 5- (..) A black hole is an object of sufficiently high density can trap light forever. 6- ( ) Millikan’s oil drop experiment is inaccurate because it is difficult to measure the speed of drop. 7- (..) Thompson cloud chamber experiment is inaccurate because the theory applies only to a single particl ...
R - University of St Andrews
... When viewed at high resolution, transitions split. Transitions between any two Bohr energy states involve several spectral lines. This is known as fine structure. Explanation: each energy level actually consists of several distinct states with almost the same energy. The first theory that justified ...
... When viewed at high resolution, transitions split. Transitions between any two Bohr energy states involve several spectral lines. This is known as fine structure. Explanation: each energy level actually consists of several distinct states with almost the same energy. The first theory that justified ...
Powerpoint handout
... Bohr derived a more general formula to predict the observed energies of light: Each electron’s energy is determined by which level it is in. The levels are designated by whole numbers, n. ...
... Bohr derived a more general formula to predict the observed energies of light: Each electron’s energy is determined by which level it is in. The levels are designated by whole numbers, n. ...
Introduction to Chemistry
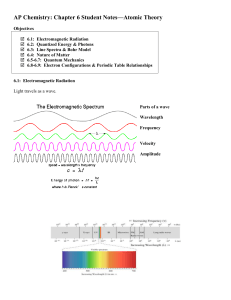
... Learning Objectives (from Zumdahl Resource Guide): (3-4 days lecture/discussion) To characterize electromagnetic radiation in terms of wavelength, frequency, and speed. To introduce the concept of quantized energy. To show that light has both wave and particulate properties. To describe how diffract ...
... Learning Objectives (from Zumdahl Resource Guide): (3-4 days lecture/discussion) To characterize electromagnetic radiation in terms of wavelength, frequency, and speed. To introduce the concept of quantized energy. To show that light has both wave and particulate properties. To describe how diffract ...
Chapter 2: Data Analysis
... quantum – quantity of energy gained or lost by an atom when electrons are excited photon – a quantum of light ground state – lowest energy level of an atom excited state – a heightened state of energy in an atom Electrons of hydrogen circle the nucleus in orbits 1. orbits have a fixed amount of ener ...
... quantum – quantity of energy gained or lost by an atom when electrons are excited photon – a quantum of light ground state – lowest energy level of an atom excited state – a heightened state of energy in an atom Electrons of hydrogen circle the nucleus in orbits 1. orbits have a fixed amount of ener ...
Lesson 2 - The Bohr and Quantum Mechanical Model of the Atom
... • how electrons occupy energy levels • maximum number of electrons at each level ...
... • how electrons occupy energy levels • maximum number of electrons at each level ...
Set #4 - comsics
... mass, which lies halfway between them. (a) If such a system were a normal atom, how would its emission spectrum compared to that of hydrogen atom? (b) What would be the electron-positron separation, r, in the ground state orbit of positronium? ...
... mass, which lies halfway between them. (a) If such a system were a normal atom, how would its emission spectrum compared to that of hydrogen atom? (b) What would be the electron-positron separation, r, in the ground state orbit of positronium? ...
The Quantum Atom (section 18)
... electromagnetic radiation and lose energy. Why does the electron not do this? Excited gases emit line spectra: light at certain characteristic frequencies, not continuous spectra. They also absorb light only at these frequencies. Why? ...
... electromagnetic radiation and lose energy. Why does the electron not do this? Excited gases emit line spectra: light at certain characteristic frequencies, not continuous spectra. They also absorb light only at these frequencies. Why? ...
e - Purdue Physics - Purdue University
... The Bohr Model for the electronic energies in atomic Hydrogen To better understand the origin of Balmer’s empirical formula, Bohr formulated a theory of the hydrogen atom in 1911. ...
... The Bohr Model for the electronic energies in atomic Hydrogen To better understand the origin of Balmer’s empirical formula, Bohr formulated a theory of the hydrogen atom in 1911. ...
(n=1).
... • Bohr’s Model gives accurate values for electron energy levels... • But Quantum Mechanics is needed to describe electrons in atom. • Electrons jump between states by emitting or absorbing photons of the appropriate energy. • Each state has specific energy and is labeled by 4 quantum numbers (next t ...
... • Bohr’s Model gives accurate values for electron energy levels... • But Quantum Mechanics is needed to describe electrons in atom. • Electrons jump between states by emitting or absorbing photons of the appropriate energy. • Each state has specific energy and is labeled by 4 quantum numbers (next t ...
spectral lines
... This led to the classic model of the atom- similar to the solar system Distant electrons orbit a massive nucleus due to electrical forces of attraction. Rutherford’s model was very appealing but there were some “minor” problems that had to be solved. What held the nucleus together to be so s ...
... This led to the classic model of the atom- similar to the solar system Distant electrons orbit a massive nucleus due to electrical forces of attraction. Rutherford’s model was very appealing but there were some “minor” problems that had to be solved. What held the nucleus together to be so s ...
1to7
... their orbit. The further out the electron orbits, more energy the electron has. Orbits are fixed distances. ...
... their orbit. The further out the electron orbits, more energy the electron has. Orbits are fixed distances. ...
Radiation and quantised orbits
... But in his model, Bohr directly assumed that the orbits in which electrons revolve are NON RADIATING. According to him, the orbits are quantised, but then what happened to classical theory of electrodynamics? ...
... But in his model, Bohr directly assumed that the orbits in which electrons revolve are NON RADIATING. According to him, the orbits are quantised, but then what happened to classical theory of electrodynamics? ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.