Corso di Fisica Moderna
... 2) Instead of the infinity of orbits which would be possible in classical mechanics, it is only possible for an electron to move in an orbit for which its orbital angular momentum L is and integra ...
... 2) Instead of the infinity of orbits which would be possible in classical mechanics, it is only possible for an electron to move in an orbit for which its orbital angular momentum L is and integra ...
Unit 06 Chapter 7 Notes
... a. When ∆E is negative, then energy is released. Homework: Section 5: The Quantum Mechanical Model of the Atom 1) Developed by: Heisenberg, de Broglie, and Schrodinger 2) Standing Wave: waves that are stationary, that do not gravel down the length of the string, similar to the electron’s motion arou ...
... a. When ∆E is negative, then energy is released. Homework: Section 5: The Quantum Mechanical Model of the Atom 1) Developed by: Heisenberg, de Broglie, and Schrodinger 2) Standing Wave: waves that are stationary, that do not gravel down the length of the string, similar to the electron’s motion arou ...
Document
... If an electron falls from the third orbit to the first, how much energy does it lose? ...
... If an electron falls from the third orbit to the first, how much energy does it lose? ...
Development of Quantum Mechanics Waves
... If light is a wave with particle properties, are electrons particles with wave properties? C. J. Davisson and L. H. Germier at Bell labs proved that electrons produce diffraction patterns and verified deBroglies hypothesis ...
... If light is a wave with particle properties, are electrons particles with wave properties? C. J. Davisson and L. H. Germier at Bell labs proved that electrons produce diffraction patterns and verified deBroglies hypothesis ...
Bohr Atom
... Bohr's condition, that the angular momentum is an integer multiple of ħ was later reinterpreted by de Broglie as a standing wave condition: the electron is described by a wave and a whole number of wavelengths must fit along the circumference of the electron's orbit: The Bohr model gives almost exac ...
... Bohr's condition, that the angular momentum is an integer multiple of ħ was later reinterpreted by de Broglie as a standing wave condition: the electron is described by a wave and a whole number of wavelengths must fit along the circumference of the electron's orbit: The Bohr model gives almost exac ...
Homework Set 1
... Use the initial conditions x(t = 0) ≡ x0 and p(t = 0) ≡ p0 to obtain the constants A and B in terms of m, ω, x0 and p0 . c. Now show that, although x(t) and p(t) clearly vary with time, the energy function H[x(t), p(t)] is actually a constant, i.e., a conserved quantity. Do this by plugging in the s ...
... Use the initial conditions x(t = 0) ≡ x0 and p(t = 0) ≡ p0 to obtain the constants A and B in terms of m, ω, x0 and p0 . c. Now show that, although x(t) and p(t) clearly vary with time, the energy function H[x(t), p(t)] is actually a constant, i.e., a conserved quantity. Do this by plugging in the s ...
Academic Chemistry Atomic History Study Guide 1. Identify and
... in World War I, contributed greatly to science prior to his death by discovering the atomic number. 11. According to the laws of classical physics, a charged particle traveling on a curved path will lose energy. This would cause an electron to drop into the nucleus, which would destroy the atom. ___ ...
... in World War I, contributed greatly to science prior to his death by discovering the atomic number. 11. According to the laws of classical physics, a charged particle traveling on a curved path will lose energy. This would cause an electron to drop into the nucleus, which would destroy the atom. ___ ...
Chapter 7 Student Learning Map
... duality explanation used to explain light and electrons? What is the relationship between the speed, frequency, and wavelength of electromagnetic radiation? What is the significance of the photoelectric effect in describing the behavior of the electron and light? ...
... duality explanation used to explain light and electrons? What is the relationship between the speed, frequency, and wavelength of electromagnetic radiation? What is the significance of the photoelectric effect in describing the behavior of the electron and light? ...
Unit 2 Intro Worksheet - Coral Gables Senior High
... 2. states the impossibility of knowing both velocity and position of a moving particle at the same time ...
... 2. states the impossibility of knowing both velocity and position of a moving particle at the same time ...
Bohr`s equation for the hydrogen atom Bohr derived an equation to
... The full treatment of the electron energy in terms of quantum numbers is not predicted by the simple Bohr model. Every electron within an atom is considered to have four quantum numbers: (a) the principal quantum number (n), representing the energy level; (b) the orbital quantum number (L), which ma ...
... The full treatment of the electron energy in terms of quantum numbers is not predicted by the simple Bohr model. Every electron within an atom is considered to have four quantum numbers: (a) the principal quantum number (n), representing the energy level; (b) the orbital quantum number (L), which ma ...
Chemistry 1 Concept 5 “Electrons in Atoms” Study Guide
... 5. What is the wavelength of microwaves of 4.0 x 109 Hz frequency? _______________ 6. An electron for which n = 4 has more _______ than an electron for which n = 2. 7. What is the frequency of a photon with an energy of 1.75 x 10 -19 J________________ 8. The number of sublevels within each energy le ...
... 5. What is the wavelength of microwaves of 4.0 x 109 Hz frequency? _______________ 6. An electron for which n = 4 has more _______ than an electron for which n = 2. 7. What is the frequency of a photon with an energy of 1.75 x 10 -19 J________________ 8. The number of sublevels within each energy le ...
Transcript - the Cassiopeia Project
... Yet the electron couldn’t be in an orbit circling the nucleus either. Circular motion requires constant acceleration of the circling body to keep it from flying away. But the electron has charge and charged particles radiate light when they are accelerating. So an electron in a circular orbit would ...
... Yet the electron couldn’t be in an orbit circling the nucleus either. Circular motion requires constant acceleration of the circling body to keep it from flying away. But the electron has charge and charged particles radiate light when they are accelerating. So an electron in a circular orbit would ...
Bohr Model and Principal Quantum Number
... developed his model Bohr postulated: 1. Electrons exist in circular orbits 2. Electrons exist only in allowed orbits 3. Electrons do not radiate energy within an orbit 4. Electrons can jump between orbits ...
... developed his model Bohr postulated: 1. Electrons exist in circular orbits 2. Electrons exist only in allowed orbits 3. Electrons do not radiate energy within an orbit 4. Electrons can jump between orbits ...
NIELS BOHR power point22222
... orbit and absorbed when it jumps from a lower to higher orbit. 4) The energy and frequency of light emitted or absorbed is given by the difference between the two orbit energies, e.g., ...
... orbit and absorbed when it jumps from a lower to higher orbit. 4) The energy and frequency of light emitted or absorbed is given by the difference between the two orbit energies, e.g., ...
mp2b-16 honors
... What was different about Planck's description of light compared to the previous theory of light? C. Know how to use the two equations of light - know the metric system! E=hx C=x D. What are the frequency, wavelength, and amplitude of a light wave? Be able to diagram. What happens when each is cha ...
... What was different about Planck's description of light compared to the previous theory of light? C. Know how to use the two equations of light - know the metric system! E=hx C=x D. What are the frequency, wavelength, and amplitude of a light wave? Be able to diagram. What happens when each is cha ...
Bohr model
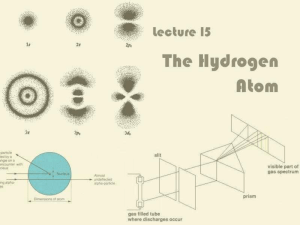
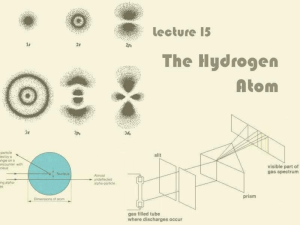
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.