* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemical Equilibrium - local.brookings.k12.sd.us
Marcus theory wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Al-Shifa pharmaceutical factory wikipedia , lookup
Host–guest chemistry wikipedia , lookup
Chemical weapon proliferation wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Safety data sheet wikipedia , lookup
Chemical weapon wikipedia , lookup
Chemical plant wikipedia , lookup
Chemical industry wikipedia , lookup
Chemical Corps wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Pseudo Jahn–Teller effect wikipedia , lookup
Colloidal crystal wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Chemical reaction wikipedia , lookup
Crystallization wikipedia , lookup
Planck's law wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Stoichiometry wikipedia , lookup
Chemical potential wikipedia , lookup
Thermomechanical analysis wikipedia , lookup
Rate equation wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
Thermodynamics wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Transition state theory wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Chemical Equilibrium I. A State of Dynamic Balance 1N2 (g) + 3H2(g) The concentrations of the reactants decrease at first… …while the concentration of the product increases 2NH3(g) ΔG0 = -33.1 kJ But, then, before the reactants are used up, all concentrations become constant The reaction is spontaneous under standard conditions Chemical Equilibrium I. A State of Dynamic Balance -when a ________ reaction results in the almost ________ complete conversion of ________ reactants to ________, products the 1N2 (g) + 3H2(g) 2NH3(g) ________ reaction is said to go to completion but _____ most _________ reactions __________, 1N2 (g) + 3H2(g) 2NH3(g) ___ do ____ not go to __________, completion most reactions are __________ reversible 1N2 (g) + 3H2(g) 2NH3(g) _________ At first, only the reactants are present, so only the forward reaction can occur 1N2 (g) + 3H2(g) 2NH3(g) Chemical Equilibrium I. A State of Dynamic Balance -as soon as the ________ forward ________ reaction begins, the concentrations ____________ of the _________ reactants go _____, down and the reaction _____ rate goes _____ down as the _________ collisions per unit number of __________ ____ time goes _____ down As soon as the products begin forming, the forward reaction rate slows and the reverse reaction begins 1N2 (g) + 3H2(g) 2NH3(g) Chemical Equilibrium I. A State of Dynamic Balance -as the _________ reaction proceeds, the ____ rate of the ________ forward _________ reaction continues to ________ decrease and the ____ rate reverse ________ reaction continues of the ________ increase until the two _____ rates are to ________ _____, equal and the system has reached a chemical __________ equilbrium 1N2 (g) + 3H2(g) 2NH3(g) state of ________ Chemical Equilibrium I. A State of Dynamic Balance -at ___________, equilibrium the concentrations ____________ of the ________ reactants and ________ products are not _____, equal but _______, constant because the rate of _________ formation of the ________ products ____ equal to the ____ rate of _________ formation is _____ of the ________ reactants The Golden Gate Bridge If all other roads leading connects San Francisco in and out of the two cities to Sausalito. were closed… …and the number of vehicles crossing the bridge per hour in one direction equaled the number of vehicles crossing the bridge in the opposite direction… What is true of the number of vehicles in each city throughout the day? Are there the same number of vehicles in each city? Chemical Equilibrium II. Equilibrium Expressions -while _____ some chemical systems have and Constants little tendency to _____, react and _____ some chemical systems _____ react readily and go to __________, completion _____ most chemical ___ state of __________, equilibrium systems reach a _____ leaving varying amounts of ________ reactant ____________ unconsumed Waage -in 1864, Norwegian chemists ______ Guldberg proposed the _______ Law of and _________ ___________________, Chemical Equilibrium which states, at a given ___________, temperature a chemical state in which a system may reach a _____ ratio of _______ reactant and particular _____ product ____________ concentrations has a constant _______ _______ value Cato Maximilian Guldberg 1836-1902 Peter Waage 1833-1900 Chemical Equilibrium II. Equilibrium Expressions -the _______ general ________ equation for a _______ reaction and Constants at __________ equilibrium can be written ______________________________, aA + bB cC + dD A and __ B are ________, reactants __ C and where __ D are ________, products __, a __, b __, c and __ d are __ the ___________ coefficients in the ________ balanced ________, equation and the __________ equilibrium _______ constant __________ expression is [C]c [D]d Keq = [A]a [B]b equilibrium ________ mixtures with ___ Keq -___________ > __ 1 contain more ________ products values __ reactants at ___________, equilibrium while than ________ __________ equilibrium ________ mixtures with ___ Keq values 1 contain more ________ __ < __ reactants than products at __________ equilibrium ________ Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the homogeneous equilibrium for the synthesis of ammonia from nitrogen and hydrogen. 1N2 (g) + 3H2(g) Keq = Keq 2NH3(g) [C]c [D]d The equilibrium is homogeneous because all the reactants and products are in the same physical state (gas) [A]a [B]b c [C] = [A]a [B]b = [NH3]c [N2]a[H2]b = [NH3]2 [N2]1[H2]3 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the equilibrium for the synthesis of Hydrogen iodide from iodine and hydrogen. 1H2 (g) + 1I2(g) Keq = Keq 2HI(g) [C]c [D]d The equilibrium is homogeneous because all the reactants and products are in the same physical state (gas) [A]a [B]b c [C] = [A]a [B]b = [HI]c [H2]a[I2]b = [HI]2 [H2]1 [I2]1 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the equilibrium for the decomposition of Dinitrogen tetroxide into Nitrogen dioxide. 1N2O4 (g) Keq = Keq = 2NO2(g) [C]c [D]d The equilibrium is homogeneous because all the reactants and products are in the same physical state (gas) [A]a [B]b [C]c [A]a = [NO2]c [N2O4]a = [NO2]2 [N2O4]1 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the equilibrium for the reaction of Carbon monoxide and Hydrogen which produces methane (Tetrahydrogen monocarbide) and water. 1CO(g) + 3H2 (g) Keq [C]c [D]d = [A]a [B]b Keq c [D]d [C] = [A]a [B]b = 1CH4(g) + [CH4]c[H2O]d = [CO]a [H2]b 1H2O (g) [CH4]1[H2O]1 [CO]1 [H2]3 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the equilibrium for the decomposition of Dihydrogen monosulfide into diatomic hydrogen and diatomic sulfur. 2H2S (g) Keq [C]c [D]d = [A]a [B]b Keq c [D]d [C] = [A]a = 2H2 (g) + [H2]c [S2]d [H2S]a = 1S2 (g) [H2]2 [S2]1 [H2S]2 Chemical Equilibrium II. Equilibrium Expressions -_________ equilibria in which all ________ reactants and and Constants ________ products are in the same ________ physical _____ state are ____________, homogeneous but reactions with _________ reactants and ________ products in _____ more than ___ one ________ physical ________ _____ state result in _____________ heterogeneous gaseous _________ equilibria ethanol 1C2H5OH (l) Keq 1C2H5OH (g) [C]c [D]d = [A]a [B]b Keq = [C2H5OH (g)]1 [C2H5OH (l)]1 Keq = [C2H5OH (g)]1 liquid ethanol Chemical Equilibrium II. Equilibrium Expressions -since ______ liquid and _____ solid ________ reactants and and Constants ________ products don’t change ___________, concentration (which is really their ______), density if the temperature remains ________, constant ___________ equilibrium _______ constant then in the ___________ __________ expression for a ____________ heterogeneous ___________, equilibrium the ___________ equilibrium ________ constant only depends on the concentrations of the ________ reactants and ______________ products in the gaseous ________ _______ state of matter Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the heterogeneous equilibrium for the decomposition of Sodium Hydrogen carbonate into Sodium carbonate, Carbon dioxide, and water. 2NaHCO3 (s) Keq = 1Na2CO3 (s) + 1CO2 (g) + 1H2O (g) [C]c [D]d [E]e [A]a Keq = [D]d [E]e = [CO2]d [H2O]e = The equilibrium is heterogeneous because the reactants and products are in different physical states (gas and solid) [CO2]1[H2O]1 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the heterogeneous equilibrium for the decomposition of Calcium carbonate into Calcium oxide and Carbon dioxide. 1CaCO3 (s) Keq = 1CaO (s) + 1CO2 (g) The equilibrium is heterogeneous because the reactants and products are in different physical states (gas and solid) [C]c [D]d [A]a Keq = [D]d = [CO2]d = [CO2]1 Chemical Equilibrium II. Equilibrium Expressions and Constants Write the complete, balanced thermochemical equation and equilibrium constant expression for the heterogeneous equilibrium for the reaction of monatomic Sulfur and fluorine gas, which produces Sulfur tetrafluoride gas and Sulfur hexafluoride gas. 2S (s) Keq = + 5F2 (g) [SF4]1 [SF6]1 [F2]5 1SF4 (g) + 1SF6 (g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the complete, balanced thermochemical equation and equilibrium constant expression for the homogeneous equilibrium for the reaction of hydrazine (Tetrahydrogen dinitride) and Nitrogen dioxide, which produces nitrogen and water. 2N2H4 (g) + Keq 2NO2 (g) 3[H O]4 [N ] 2 2 = [N2H4]2[NO2]2 3N2 (g) + 4H2O (g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the complete, balanced thermochemical equation and equilibrium constant expression for the homogeneous equilibrium for the reaction of Sulfur trioxide and Carbon dioxide, which produces Carbon disulfide and oxygen. 2SO3 (g) + 1CO2 (g) Keq = [CS2]1 [O2]4 [SO3]2 [CO2]1 1CS2 (g) + 4O2 (g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the complete, balanced thermochemical equation and equilibrium constant expression for the heterogeneous equilibrium for the reaction of monatomic Sulfur and fluorine gas, which produces Sulfur tetrafluoride gas and Sulfur hexafluoride gas. 2S (s) Keq = + 5F2 (g) [SF4]1 [SF6]1 [F2]5 1SF4 (g) + 1SF6 (g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the complete, balanced thermochemical equation and equilibrium constant expression for the heterogeneous equilibrium for the reaction of magnatite (Fe3O4) and hydrogen gas, which produces iron and water vapor. 1Fe3O4 (s) + Keq = 4H2 (g) [H2O]4 [H2]4 3Fe (s) + 4H2O (g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the homogeneous equilibrium for the synthesis of ammonia and calculate the value of Keq when [NH3] = 0.933 M, [N2] = 0.533 M, and [H2] = 1.600 M. 1N2 (g) + 3H2(g) Keq = [NH3]2 [N2]1[H2]3 Keq = [0.933]2 [0.533]1 [1.600]3 Keq = 0.399 2NH3(g) Chemical Equilibrium II. Equilibrium Expressions and Constants Write the equilibrium constant expression for the homogeneous equilibrium for the decomposition of Sulfur trioxide into Sulfur dioxide and oxygen gas, and calculate the value of Keq when [SO3] = 0.0160 M, [SO2] = 0.00560 M, and [O2] = 0.00210 M. 2SO3 (g) Keq = [SO2]2[O2]1 [SO3]2 Keq = [0.00560]2 [0.00210]1 [0.0160]2 Keq = 2.58 x 10-4 2SO2 (g) + 1O2 (g) Chemical Equilibrium III. Le Châtelier’s Principle 1. Hypothesis: What is the effect of temperature on equilibrium? 2. Prediction: 3. Gather Data: A. Safety: The surfaces of the hot plates and the water will be hot enough to cause burns. Use caution. Cobalt(II) chloride hexahydrate is toxic, with an LD50 = 80mg/kg Avoid ingestion (don’t eat or drink it). Wash hands thoroughly with soap and water before leaving lab. Ethanol is extremely flammable. No open flame. B. Procedure: 1. Pick up a sheet of white construction paper and an artist’s paintbrush. Chemical Equilibrium III. Le Châtelier’s Principle 3. Gather Data: B. Procedure: 2. With a partner, using a top-loading electronic balance, mass 0.3 grams of CoCl2·6H2O, crush it into a fine powder using a mortar and pestle, and place it in a test tube. 3. Add 10 mL of ethanol to the test tube, cap, and shake vigorously until CoCl2·6H2O dissolves. If the solution is not light pink, add water dropwise until it turns light pink. 4. Use the solution to paint a winter scene on your white construction paper, including a pink-colored field of snow. 5. To simulate the coming of spring, warm your painting over the hotplate in the fume hood. Record your observations. Chemical Equilibrium III. Le Châtelier’s Principle 4. Analyze Data: + heat 1Co(H2O)62+ (aq) + Hexahydrate Co2+ ion (pink) 1Co(H2O)62+ (aq) + Hexahydrate Co2+ ion (pink) 5. Draw Conclusions: 4Cl- (aq) chloride ion 4Cl- (aq) chloride ion 1CoCl42-(aq) + 6H2O (l) Tetrachlorocobaltate ion (blue) 1CoCl42-(aq) + 6H2O (l) Tetrachlorocobaltate ion (blue) Chemical Equilibrium III. Le Châtelier’s Principle -________ reactions that reach __________ equilibrium instead of going to __________ completion do not ________ produce as much -in 1888, ________________________ Henry-Louis Le Châtelier discovered that there are ways to _______ control _________ equilibria in order to make reactions more __________ productive _________ -____________________ Le Châtelier’s Principle states that if a stress (like a ______ change in ______ temperature is applied to a system __________) equilibrium the system _____ shifts in at __________, the ________ direction that _______ relieves the _____ stress Henry-Louis Le Châtelier 1850-1936 Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium A. Changes in Concentration Write the equilibrium constant expression for the equilibrium for the reaction of Carbon monoxide and Hydrogen to produce methane and water. Then, calculate the Keq value when [CO] = 0.30000 M, [H2] = 0.10000 M, and [CH4] = 0.05900 M, and [H2O] = 0.02000 M. 1CO(g) + 3H2 (g) Keq = Keq 1CH4(g) + 1H2O (g) [CH4]1[H2O]1 [CO]1 [H2]3 1 [0.02000]1 [0.05900] = [0.30000]1[0.10000]3 = 3.933 = first equilibrium position Chemical Equilibrium III. Le Châtelier’s Principle A. Changes in Concentration -_________ increasing the ____________ concentration of CO _________ increases the _______ number of ___ _________ ___ and collisions between CO ___, H2 _________ increasing the _____ rate of the forward _______ reaction _______ 1CO(g) + 3H2 (g) 1CH4(g) + 1H2O (g) -the system responds to the ______ stress of the addition of reactant by forming more _______ product to bring the system _______ back into equilbrium Chemical Equilibrium III. Le Châtelier’s Principle A. Changes in Concentration 1CO(g) + 3H2 (g) 0.99254 M Keq = Keq 0.07762 M 1CH4(g) + 1H2O (g) 0.06648 M 0.02746 M [CH4]1[H2O]1 [CO]1 [H2]3 1 [0.02746]1 [0.06648] = [0.99254]1[0.07762]3 = 3.933 = second equilibrium position Chemical Equilibrium III. Le Châtelier’s Principle A. Changes in Concentration -_________ increasing the ____________ concentration of reactant causes __________ equilbrium a ________ to _____ _______ shift to the ____ right to increase the ____ ______ rate of formation of product 1CO(g) + 3H2 (g) 1CH4(g) + 1H2O (g) 1CO(g) + 3H2 (g) 1CH4(g) + 1H2O (g) decreasing the ____________ concentration of -_________ product causes __________ equilbrium a ________ to _____ _______ shift to the ____ right to increase the ____ ______ rate of formation of product Chemical Equilibrium III. Le Châtelier’s Principle A. Changes in Concentration Predict what should happen to the following equilibrium if hydrogen bonding due to the addition of acetone binds water and effectively removes it from the products. 1Co(H2O)62+ (aq) + Hexahydrate Co2+ ion (pink) 4Cl- (aq) chloride ion 1CoCl42-(aq) + 6H2O (l) Tetrachlorocobaltate ion (blue) Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium A. Changes in Volume -_________ decreasing the volume ______ of the _______ reaction container, according to ______, Boyle increases the ________, pressure which in turn ________ 1CO(g) + 3H2 (g) increases the _____ rate of _________ collision ________ particles of the between the ________ ________, reactants _________ increasing the _____ rate of the ________ forward _______ reaction 1CH4(g) + 1H2O (g) shift in the equilibrium -the _____ _________ causes the stress on the system to be _______ relieved as _____ for every __ 4 _____ moles of _______ gaseous _______ reactant _________, _____ of _______ consumed only __ 2 moles gaseous product are _________, produced which, _______ ½ the according to Avogadro ________, occupies __ volume decreases the pressure ______, which _________ ________ Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium Use Le Châtelier’s Principle to predict how each of these changes would affect the ammonia equilibrium system. 1N2 (g) + 3H2(g) 2NH3(g) a. removing hydrogen from the system __________________________ equilibrium shifts to the left 1N2 (g) + 3H2(g) 2NH3(g) b. adding ammonia to the system _______________________________ equilibrium shifts to the left 1N2 (g) + 3H2(g) 2NH3(g) Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium Use Le Châtelier’s Principle to predict how each of these changes would affect the ammonia equilibrium system. 1N2 (g) + 3H2(g) 2NH3(g) c. adding hydrogen to the system _______________________________ equilibrium shifts to the right 1N2 (g) + 3H2(g) 2NH3(g) Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium How would decreasing the volume of the reaction container affect each of these equilibria? a. 2SO2 (g) + 1O2(g) 2SO3(g) _________________________ equilibrium shifts to the right b. 1H2 (g) + 1Cl2(g) 2HCl(g) _____________________________ stress has no effect on equilibrium c. 2NOBr(g) 2NO(g) + 1Br2(g) _________________________ equilibrium shifts to the left Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium A. Changes in Temperature -while _______ changes in _____________ concentration and ________ changes in _______ volume cause ______ shifts in _________, equilibria they ___ do not _______ change the __________ equilibrium ___ constant but a ______ change in _______, ___________ temperature causes ______ change in both the __________ equilibrium ________ position equilibrium _______ constant and the __________ Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium A. Changes in Temperature -since the _______ reaction for making _______ methane has a _______ negative ____, ΔH0 the ________ _______ is _________, forward reaction exothermic 1CO(g) + 3H2 (g) reverse _______ reaction is and the _______ endothermic heat can be __________, so ____ thought of as a _______ product in the ________ forward _______ reaction and a _______ reactant reverse reaction in the _______ _______ 1CH4(g) + 1H2O (g) + heat ΔH0 = -206.5 kJ Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium A. Changes in Temperature -_________ increasing the temperature __________ is like _______ adding more _______ reactant to the _______ reaction in which _____ heat acts as a reactant and is _____ used ___, up in this _______ endothermic _______ reverse case, the __________ _______ reaction 1CO(g) + 3H2 (g) 1CH4(g) + 1H2O (g) + heat equilibrium shifts to the _____, left -__________ _________ decreasing the ___________ concentration of _______ methane because _______ methane is a reactant reverse _______ reaction _______ in the _______ Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium In the following equilibrium, would you raise or lower the temperature to get the following results? 1C2H2 (g) + 1H2O(g) 1CH3CHO(g) ΔH0 = -151 kJ a. increase the amount of CH3CHO______________________________ lower the temperature 1C2H2 (g) + 1H2O(g) 1CH3CHO(g) + heat b. decrease the amount of C2H2 ________________________________ lower the temperature 1C2H2 (g) + 1H2O(g) 1CH3CHO(g) + heat Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium In the following equilibrium, would you raise or lower the temperature to get the following results? 1C2H2 (g) + 1H2O(g) 1CH3CHO(g) ΔH0 = -151 kJ c. increase the amount of H2O _________________________________ raise the temperature 1C2H2 (g) + 1H2O(g) 1CH3CHO(g) + heat Chemical Equilibrium III. Le Châtelier’s Principle -stressors that cause a shift in equilibrium In the following equilibrium, what effect does decreasing the volume of the reaction vessel have? 1CO(g) + 1Fe3O4(s) 1CO2 (g) + 3FeO(s) __________________________________________________________ None. The solids do not change their concentrations, and the number __________________________________________________________ of moles of gaseous reactant equals the number of moles of gaseous __________________________________________________________ product In the following equilibrium, what effect does simultaneously increasing the temperature and the pressure have? 1CO (g) + 1Cl2(g) 1COCl2(g) + heat ΔH0 = -151 kJ __________________________________________________________ Cannot predict. An increase in temperature causes a shift in the __________________________________________________________ equilibrium to the left, while an increase in pressure causes a shift in __________________________________________________________ equilibrium to the right. Chemical Equilibrium III. Le Châtelier’s Principle 1. Hypothesis: What is the effect of a change in concentration of reactants and a change in temperature on equilibrium? 2. Prediction: 3. Gather Data: A. Safety: The surfaces of the hot plate will be hot enough to cause burns. Use caution. Cobalt(II) chloride hexahydrate is toxic, with an LD50 = 80mg/kg Avoid ingestion (don’t eat or drink it). Wash hands thoroughly with soap and water before leaving lab. Concentrated Hydrochloric acid is extremely corrosive. Avoid contact with eyes, skin, and clothing. Goggles, aprons, and gloves mandatory. Chemical Equilibrium III. Le Châtelier’s Principle 3. Gather Data: B. Procedure: 1. With a partner, measure out 2 mL of 0.1 M CoCl2 solution into a test tube. Record initial color. __________ 2. Add 3 mL (60 drops) of concentrated HCl to the test tube. Record color. _____________ 3. Add water dropwise to the test tube until a color change occurs. Record color. ______________ 4. Add 2 mL of 0.1 M CoCl2 solution to another test tube. Add concentrated HCl dropwise until the solution turns purple. If the solution turns blue, add water until it turns purple. Chemical Equilibrium III. Le Châtelier’s Principle 3. Gather Data: B. Procedure: 5. Place the test tube in an ice bath. Record color. ________ 6. Place the test tube in a hot water bath. Record color. ___________ 4. Analyze Data: A. The equation for the reversible reaction in this experiment is: + heat 1Co(H2O)62+ (aq) + Hexahydrate Co2+ ion (pink) 4Cl- (aq) chloride ion 1CoCl42-(aq) + 6H2O (l) Tetrachlorocobaltate ion (blue) Chemical Equilibrium III. Le Châtelier’s Principle 4. Analyze Data: A. Use the equation to explain the colors of the solution in steps 1, 2, and 3 In Step 1, the solution is initially a pink color, because the reaction arrives at an equilibrium in which the concentration of the pink-colored 1Co(H2O)62+(aq) is at a higher concentration than the blue-colored 1CoCl42-(aq). In Step 2, the addition of HCl increases the concentration of Cl-, shifting the equilibrium to the right to favor the formation of the blue 1CoCl42-(aq), so the solution turns blue. In Step 3, the increase in concentration of water shifts the equilibrium left, re-establishing a new equilbrium where the concentration of 1CoCl42-(aq) is higher than it was orginally, so the purple color shows more of a balance of pink and blue. Chemical Equilibrium III. Le Châtelier’s Principle 4. Analyze Data: B. Explain how the equilibrium shifts when heat energy is added or removed. In Step 5, since heat acts like a product in the exothermic reverse reaction, removing heat by lowering the temperature causes the equilibrium to shift to the left, increasing the rate of the reverse reaction and causing the solution to turn pink. In Step 6, since heat acts like a reactant in the endothermic forward reaction, adding heat by increasing the temperature causes the equilibrium to shift to the right, increasing the rate of the forward reaction and causing the solution to turn blue. 5. Draw Conclusions: Chemical Equilibrium IV. Using Equilibrium Constants -when a ________ reaction has a _____ large Keq the __________ ___, equilibrium _______ mixture contains _____ more ________ products than reactants at __________ equilibrium ________ -when a ________ reaction has a _____ small Keq the __________ ___, equilibrium _______ mixture contains _____ more ________ reactants than products at __________ equilibrium ________ A. Calculating Equilibrium Concentrations -__________ equilibrium ________ constants can also be used to ________ calculate the equilibrium __________ ____________ concentration of any ________ substance in the _______ reaction Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations At 1200 K, the Keq for the following reaction equals 3.933. What is the concentration of the methane produced, if [CO] = 0.850 M, [H2] = 1.333 M, and [H2O] = 0.286 M? 1CO(g) + 3H2 (g) 0.850 M Keq = 3.933 = 1.333 M [CH4]1[H2O]1 [CO]1 [H2]3 [CH4]1[0.286]1 [0.850]1[1.333]3 [CH4] = 27.7 M 1CH4(g) + ?M 1H2O (g) 0.286 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations At 1405 K, the Keq for the following reaction equals 2.27 x 10-3. What is the concentration of the Hydrogen gas produced, if [S2] = 0.0540 M, and [H2S] = 0.184 M? 2H2S (g) 0.184 M Keq = 2.27 x 10-3 [H2] [H2]2 [S2]1 [H2S]2 [H2]2 [0.0540]1 [0.184]2 = = 0.0377 M 2H2(g) + ?M 1S2 (g) 0.0540 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations If Keq for the following reaction equals 10.5, what is the equilibrium concentration of Carbon monoxide, if [H2] = 0.933 M, and [CH3OH] = 1.32 M? 1CO(g) + 2H2 (g) ?M Keq = 10.5 = 0.933 M [CH3OH]1 [CO]1 [H2]2 [1.32]1 [CO]1 [0.933]2 [CO] = 0.144 M 1CH3OH (g) 1.32 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) If the Keq for the following reaction equals 64.0, what are the equilibrium concentrations of I2, H2, and HI, if [I2]0 = 0.200 M, [H2]0 = 0.200 M and [HI] = 0.000 M? 1I2(g) + 1H2 (g) ?M Initial Change 2HI(g) ?M ?M [H2] [I2] [HI] 0.200 0.200 0.000 -1x -1x +2x 0.200 - 1x 2x Equilibrium 0.200 - 1x Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 1I2(g) + 1H2 (g) Initial Change [H2] [I2] [HI] 0.200 0.200 0.000 -1x -1x +2x 0.200 - 1x 2x Equilibrium 0.200 - 1x Keq = [HI]2 [I2]1 [H2]1 2HI(g) 8.00 = 2 [2x] 64.0 = [0.200 – 1x]1 [0.200 – 1x]1 [2x] [0.200 – 1x]1 x [H2] [I2] [HI] = = = = 0.160 0.040 M 0.040 M 0.320 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) If the Keq for the following reaction equals 16.0, what are the equilibrium concentrations of PCl3, Cl2, and PCl5, if [PCl5]0 = 1.00 M? 1PCl5(g) 1PCl3(g) + 1Cl2 (g) ?M ?M ?M [PCl3] [Cl2] [PCl5] Initial 0.00 0.00 1.00 Change +1x +1x -1x 0.00 + 1x 1.00 – 1x Equilibrium 0.00 + 1x Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 1PCl5(g) 1PCl3(g) + 1Cl2 (g) [PCl3] [Cl2] [PCl5] Initial 0.00 0.00 1.00 Change +1x +1x -1x 0.00 + 1x 1.00 – 1x Equilibrium 0.00 + 1x 1 [Cl ]1 [PCl ] 3 2 Keq = x2 = 16.0 – 16.0x x = -b ± √b2 – 4ac [PCl5]1 x2 + 16.0x - 16.0 = 0 2a 1 [x]1 [x] ax2 + bx + c = 0 16.0 = [1.00 - x]1 Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) x2 + 16.0x - 16.0 = 0 ax2 + bx + c = 0 x = -b ± √b2 – 4ac 2a x = -16.0 ± √(-16.0)2 – 4(1.00)(-16.0) 2(1.00) x = 0.950 (but not -17.0) [PCl3] = 0.950 M [Cl2] = 0.950 M [PCl5] = 0.05 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) If the Keq for the following reaction equals 0.680, what are the equilibrium concentrations of COCl2, CO, and Cl2, if [CO]0 = 0.500 M and [Cl2]0 = 1.00 M? 1CO (g) ?M + 1Cl2 (g) 1COCl2 (g) ?M ?M [COCl2] [CO] [Cl2] Initial 0.00 0.500 1.00 Change +1x -1x -1x 0.500 - 1x 1.00 – 1x Equilibrium 0.00 + 1x Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 1CO (g) + 1Cl2 (g) [COCl2] [CO] [Cl2] Initial 0.00 0.500 1.00 Change +1x -1x -1x 0.500 - 1x 1.00 – 1x Equilibrium 0.00 + 1x Keq = 0.680 = 1COCl2 (g) [COCl2]1 [CO]1 [Cl2]1 x = 0.340 - 0.340x – 0.680x + x2 0.680x2 - 2.02x + 0.340 = 0 [x]1 [0.500 - x]1 [1.00 - x]1 ax2 + bx + c = 0 Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 0.680x2 - 2.02x + 0.340 = 0 ax2 + bx + c = 0 x = x = -b ± √b2 – 4ac 2a 2.02 ± √(-2.02)2 – 4(0.680)(0.340) 2(0.680) x = 2.79 or 0.176 [COCl2] = 0.176 M [CO] = 0.324 M [Cl2] = 0.82 M x = 0.176 Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) If the Keq for the following reaction equals 36.0, what are the equilibrium concentrations of H2, Br2, and HBr, if [H2]0 = 0.250 M and [Br2]0 = 0.250 M? 1H2(g) ?M + 1Br2 (g) 2HBr(g) ?M ?M [H2] [Br2] [HBr] Initial 0.250 0.250 0.000 Change -1x -1x +2x 0.250 - 1x 0.000 + 2x Equilibrium 0.250 - 1x Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 1H2 (g) + 1Br2 (g) [H2] [Br2] [HBr] Initial 0.250 0.250 0.000 Change -1x -1x +2x 0.250 - 1x 0.000 + 2x Equilibrium 0.250 - 1x Keq = 36.0 = 2HBr (g) [HBr]2 [H2]1 [Br2]1 [2x]2 [0.250 - x]1 [0.250 - x]1 6.00 = 2x (0.250 – x) x = 0.188 Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 32x2 - 18.0x + 2.25 = 0 ax2 + bx + c = 0 x = -b ± √b2 – 4ac 2a x = 18.0 ± √(-18.0)2 – 4(32)(2.25) 2(32) x = 0.375 or 0.188 [H2] = 0.062 M [Br2] = 0.062 M [HBr] = 0.376 M Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) If the Keq for the following reaction equals 20.0, what are the equilibrium concentrations of H2, Cl2, and HCl, if [H2]0 = 1.00 M and [Cl2]0 = 2.00 M? 1H2(g) ?M + 1Cl2 (g) 2HCl(g) ?M ?M [H2] [Cl2] [HCl] Initial 1.00 2.00 0.00 Change -1x -1x +2x Equilibrium 1.00 - 1x 2.00 - 1x 0.00 + 2x Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 1H2 (g) 20.0 = 2HCl (g) [H2] [Cl2] [HCl] Initial 1.00 2.00 0.00 Change -1x -1x +2x Equilibrium Keq = + 1Cl2 (g) [HCl]2 [H2]1 [Cl2]1 1.00 - 1x 2.00 - 1x 0.00 + 2x 4.00x2 = 40.0 – 60.0x + 20.0x2 16.0x2 - 60.0x + 40.0 = 0 [2x]2 [1.00 - x]1 [2.00 - x]1 ax2 + bx + c = 0 Chemical Equilibrium IV. Using Equilibrium Constants A. Calculating Equilibrium Concentrations from Initial Concentrations Using ICE (Initial, Change, Equilibrium) 16.0x2 - 60.0x + 40.0 = 0 ax2 + bx + c = 0 x = -b ± √b2 – 4ac 2a x = 60.0 ± √(-60.0)2 – 4(16.0)(40.0) 2(16.0) x = 2.88 or 0.869 [H2] = 0.13 M [Cl2] = 1.13 M [HCl] = 1.74 M Chemical Equilibrium V. Solubility Equilibria -like a few _________ chemical _________ reactions that go to completion _________, upon __________, dissolving some ______ ionic __________ compounds _________ dissociate ions completely into _____ 1Na+ (aq) + 1NaCl (s) 1Cl- (aq) -some _____ ionic __________, compounds however, are sparingly _______, soluble and quickly reach only ________ solubility __________ equilibrium a ________ 1BaSO4 (s) Keq = 1Ba2+ (aq) + [Ba2+ ]1 [SO42-]1 [BaSO4]1 1SO42- (aq) Chemical Equilibrium V. Solubility Equilibria -in the __________ equilibrium constant _______ __________, expression ______ Barium ______ sulfate is a _____, solid so the _______ [BaSO4] is _______, constant and can be combined with the Keq value to form the ________ solubility _______ product ___ constant _______ Keq x [BaSO4]1 = [Ba2+ ]1 [SO42-]1 Ksp = [Ba2+ ]1 [SO42-]1 Write the solubility constant expression for the following solubility equilibrium: 1Mg(OH)2 (s) 1Mg2+ (aq) + 2OH- (aq) Ksp = [Mg2+ ]1 [OH-]2 Chemical Equilibrium V. Solubility Equilibria A. Calculating Solubilities from Solubility Product Constants What is the solubility, in M, of Silver iodide at 298 K? 1Ag+ (aq) + 1I- (aq) 1AgI (s) Ksp = [Ag+ ]1 [I-]1 8.5 x 10-17 = [Ag+ ]1 [I-]1 8.5 x 10-17 = s2 √ 8.5 x 10-17 = √ s2 s = 9.2 x 10-9 M Chemical Equilibrium V. Solubility Equilibria A. Calculating Solubilities from Solubility Product Constants What is the solubility, in M, of Copper(II) carbonate at 298 K? 1CuCO3 (s) 1Cu2+ (aq) + 1CO32- (aq) Ksp = [Cu2+ ]1 [CO32-]1 2.5 x 10-10 = [Cu2+ ]1[CO32-]1 2.5 x 10-10 = s2 √ 2.5 x 10-10 = √ s2 s = 1.6 x 10-5 M Chemical Equilibrium V. Solubility Equilibria B. Calculating Ion Concentration from Ksp What is [OH-] at 298 K in a saturated solution of Mg(OH)2 at equilibrium? 1Mg(OH)2 (s) 1Mg2+ (aq) + 2OH- (aq) Ksp = [Mg2+ ]1 [OH-]2 let x = [Mg2+ ] so 2x = [OH-] 5.6 x 10-12 = (x)(2x)2 5.6 x 10-12 = 4x3 1.4 x 10-12 = x3 3 √1.4 x 10-12 = x 1.1 x 10-4 = x 2.2 x 10-4 = 2x = [OH-] Chemical Equilibrium V. Solubility Equilibria B. Calculating Ion Concentration from Ksp What is [Ag+] at 298 K in a saturated solution of AgBr at equilibrium? 1Ag+ (aq) + 1AgBr (s) 1Br- (aq) Ksp = [Ag+ ]1 [Br-]1 let x = [Ag+ ] so x = [Br-] 5.4 x 10-13 = x2 √5.4 x 10-13 = x 7.3 x 10-7 = x = [Ag+] Chemical Equilibrium V. Solubility Equilibria B. Calculating Ion Concentration from Ksp What is [F-] at 298 K in a saturated solution of CaF2 at equilibrium? 1Ca2+ (aq) + 1CaF2 (s) 2F- (aq) Ksp = [Ca2+ ]1 [F-]2 3.5 x 10-11 = (x)(2x)2 3.5 x 10-11 = 4x3 8.8 x 10-12 = x3 3 √8.8 x 10-12 = x 2.1 x 10-4 = x 4.2 x 10-4 = 2x = [F-] Chemical Equilibrium V. Solubility Equilibria C. Predicting Precipitates -besides being used to calculate the _________ solubility of an _____ ionic _________ compound and the ___________ concentration of ____ ions in a saturated _______, solution ___ Ksp values can _________ predict if a _________ precipitate be used to _______ will form if two ___ _____ ionic __________ compounds are mixed Predict whether PbCl2 will form as a precipitate if 100 mL of 0.0100 M NaCl is added to 100 mL of 0.0200 M Pb(NO3)2: initial -the concentrations ____________ of the ______ solutions ________ allow you to calculate _______ the ____________ concentrations of ____ Pb2+ and ___ Cl- ions in the _____ mixed _________, solutions which multiplied together, when _________ ion _______, product or ___ Qsp determine the ___ Chemical Equilibrium V. Solubility Equilibria C. Predicting Precipitates Predict whether PbCl2 will form as a precipitate if 100 mL of 0.0100 M NaCl is added to 100 mL of 0.0200 M Pb(NO3)2: 1Pb2+ (aq) + 1PbCl2 (s) 2Cl- (aq) Qsp = [Pb2+]1 [Cl-]2 [Pb2+] = [Cl-] = 0.0200 M 2 0.0100 M = 0.0100 M = 0.00500 M 2 Qsp = [0.0100]1 [0.00500]2 Qsp = 2.50 x 10-7 < 1.7 x 10-5 = Ksp Chemical Equilibrium V. Solubility Equilibria C. Predicting Precipitates -if the ___ Qsp is ___ < the ___, Ksp the _______ is __________, solution unsaturated and a _________ precipitate ____ will ___ not ____, form and if Qsp is ___ = the ___, Ksp the _______ solution the ___ saturated and ___ no change is _________ ______ will Qsp is ___ Ksp a occur, but if ___ > the ___, __________ precipitate will form, reducing the Qsp ___ Ksp ___ ion ___________ concentration until ___ = ___, and the system arrives at equilibrium and the _______ solution __________ becomes ________ saturated Qsp = 2.50 x 10-7 < 1.7 x 10-5 = No precipitate should form Ksp Chemical Equilibrium V. Solubility Equilibria C. Predicting Precipitates Predict whether Ag2SO4 will form as a precipitate if 500 mL of 0.010 M AgNO3 is added to 500 mL of 0.25 M K2SO4: 1Ag2SO4 (s) 2Ag+ (aq) + 1SO42- (aq) Qsp = [Ag+]2 [SO42-]1 [Ag+] = [SO42-] = Qsp Qsp 0.010 M 2 0.25 M = 0.0050 M = 0.012 M 2 = [0.0050]2 [0.012]1 = 3.0 x 10-7 < 1.2 x 10-5 = No precipitate should form Ksp Chemical Equilibrium V. Solubility Equilibria C. Predicting Precipitates Predict whether a precipitate will form if 200 mL of 0.20 M MgCl2 is added to 200 mL of 0.0025 M NaOH: 1Mg(OH)2 (s) 1Mg2+ (aq) + 2OH- (aq) Qsp = [Mg2+]1 [OH-]2 [Mg2+] = [OH-] = 0.20 M 2 0.0025 M = 0.10 M = 0.0012 M 2 Qsp = [0.10]1 [0.0012]2 Qsp = 1.4 x 10-7 > 5.6 x 10-12 = Ksp A precipitate of Mg(OH)2 should form Chemical Equilibrium V. Solubility Equilibria D. Common Ion Effect -the ________ solubility of _______ PbCrO4 in _____ water is ________ 4.8 x 10-7 mol/L, which means that you can ________ dissolve ________ 4.8 x 10-7 of PbCrO4 in ____ 1.00 L of _____ pure _____, water _______ 4.8 x 10-7 of _______ PbCrO4 will ____ not but _________ _______ dissolve in ____ 1.00 L of a ______ 0.10 M solution of _______, K2CrO4 because of the ________ common ___ ion ______ effect 1PbCrO4 (s) 1Pb2+ (aq) + 1CrO42- (aq) Ksp = [Pb2+]1 [CrO42-]1 = 2.3 x 10-13 product of the concentrations -since the _______ ____________ ions is _____ equal to a constant of both ____ _______, (the _________ solubility _______ product _______), constant if 2+] [CrO42-] goes up _______ __, [Pb _____ must go _____ down Chemical Equilibrium V. Solubility Equilibria D. Common Ion Effect -adding a _______ solution to an __________ equilibrium that contains a ________ common ___ ion _______ lowers the ________ solubility of a _________ substance ion or, according to containing that ___, Le Châtelier’s ________, Principle stresses _____________ the __________ equilibrium and causes the _______ system to _____ shift the __________ equilibrium in the _______ direction that _______ relieves the ______ stress 1PbCrO4 (s) 1Pb2+ (aq) + 1CrO42- (aq) Chemical Equilibrium V. Solubility Equilibria 1. Hypothesis: How Do Solubility Product Constants Compare? 2. Prediction: 3. Gather Data: A. Safety: Silver nitrate stains skin and clothing and is highly toxic, with an LD50 = 50mg/kg Avoid ingestion (don’t eat or drink it). Wash hands thoroughly with soap and water before leaving lab. Goggles mandatory. B. Procedure: 1. Using a pipette, place 20 drops of AgNO3 solution into test well A1 of a 20-well microplate. Place 20 more drops of the same solution in test well A2. Chemical Equilibrium V. Solubility Equilibria 3. Gather Data: B. Procedure: 2. Add 10 drops of NaCl solution to both test well A1 and test well A2. Record observations___________________ 3. To test well A2 only, add 10 drops of Na2S solution. Record observations______________________________ 4. Compare the contents of test wells A1 and A2. Record observations_____________________________________ Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: A. Write the complete thermochemical equation for the reaction that occurred in Step 2. B. Write the net ionic equation for the reaction in Step 2. C. Write the equation for the solubility equilibrium that was established in test wells A1 and A2 during Step 2. D. Write the solubility constant expression for the equilibrium established in test wells A1 and A2 during Step 2. E. Write the equation for the solubility equilibrium that was established in test well A2 during Step 4. Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: F. Match the chemical formula of each precipitate with its color. G. Compare the two Ksp values for the two precipitates. Infer which is the more soluble. H. Use Le Châtelier’s Principle to explain how the addition of Na2S in Step 4 affected the equilibrium in test well A2. Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: I. Calculate the molar solubilities of both precipitates in the experiment. Which of the precipitates is more soluble? Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: A. Write the complete thermochemical equation for the reaction that occurred in Step 2. AgNO3 (aq) + NaCl (aq) → AgCl (s) + NaNO3 (aq) B. Write the net ionic equation for the reaction in Step 2. Ag+ (aq) + Cl- (aq) AgCl (s) C. Write the equation for the solubility equilibrium that was established in test wells A1 and A2 during Step 2. 1AgCl (s) 1Ag+ (aq) + 1Cl- (aq) D. Write the solubility constant expression for the equilibrium established in test wells A1 and A2 during Step 2. Ksp = [Ag+]1 [Cl-]1 = 1.8 x 10-10 E. Write the equation for the solubility equilibrium that was established in test well A2 during Step 4. 1Ag2S (s) 2Ag+ (aq) + S2- (aq) Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: F. Match the chemical formula of each precipitate with its color. AgCl (s) is white; Ag2S (s) is black G. Compare the two Ksp values for the two precipitates. Infer which is the more soluble. Ksp for AgCl (s) = 1.8 x 10-10 Ksp for Ag2S (s) = 8 x 10-48; AgCl is more soluble H. Use Le Châtelier’s Principle to explain how the addition of Na2S in Step 4 affected the equilibrium in test well A2. 1AgCl (s) 1Ag+ (aq) + 1Cl- (aq) The addition of S2- to the equilibrium removes Ag+ from the equilibrium, causing the system to shift to the right in favor of the formation of Ag+, so at the same time the black precipitate Ag2S is forming, the white AgCl is dissolving to relieve the stress on the equilibrium. Chemical Equilibrium V. Solubility Equilibria 4. Analyze Data: I. Calculate the molar solubilities of both precipitates in the experiment. Which of the precipitates is more soluble? 1Ag+ (aq) + 1Cl- (aq) 1AgCl (s) 2Ag+ (aq) + 1S2- (aq) 1Ag2S (s) Ksp = [Ag+ ]1 [Cl-]1 Ksp = [Ag+ ]2 [S2-]1 1.8 x 10-10 = [Ag+ ]1 [Cl-]1 8 x 10-48 = [Ag+ ]2 [S2-]1 1.8 x 10-10 = s2 8 x 10-48 = = √ s2 √ 1.8 x 10-10 3 √ 2x 10-48 s = 1.3 x 10-5 M AgCl is more soluble = 4s3 3 √ s3 s = 1 x 10-16 M










































































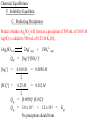





















![[A, 8-9]](http://s1.studyres.com/store/data/006655537_1-7e8069f13791f08c2f696cc5adb95462-150x150.png)
