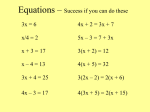* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 7 Lecture
Supramolecular catalysis wikipedia , lookup
Atomic theory wikipedia , lookup
Isotopic labeling wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Nuclear fusion wikipedia , lookup
History of chemistry wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Organic chemistry wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Water splitting wikipedia , lookup
Acid–base reaction wikipedia , lookup
Electrolysis of water wikipedia , lookup
Asymmetric induction wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Process chemistry wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Metalloprotein wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Rate equation wikipedia , lookup
Marcus theory wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
George S. Hammond wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Electrochemistry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Click chemistry wikipedia , lookup
Transition state theory wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Stoichiometry wikipedia , lookup
Chapter 7 Chemical Reactions O2 H2 H2 O 2006, Prentice Hall react to form? + heat CHAPTER OUTLINE Chemical Reactions Chemical Equation Balancing Equations Types of Chemical Reactions Activity Series of Metals Aqueous Reactions Precipitation Reactions Neutralization and Other Reactions Heat in Chemical Reactions 2 CHEMICAL REACTIONS A chemical reaction is a rearrangement of atoms in which some of the original bonds are broken and new bonds are formed to give different chemical structures. In a chemical reaction, atoms are neither created, nor destroyed. A chemical reaction, as described above, is supported by Dalton’s postulates. 3 CHEMICAL REACTIONS In a chemical reaction, atoms are neither 6 oxygen atoms = 6 oxygen atoms created, nor destroyed 4 CHEMICAL REACTIONS A chemical reaction can be detected by one of the following evidences: 1. Change of color 2. Formation of a solid 3. Formation of a gas 4. Exchange of heat with surroundings 5 Evidence of Chemical Change Emission of Lightof Heat Color Change Release or Absorption Formation of Solid Formation of aPrecipitate Gas Why use Chemical Equations? 1. Shorthand way of describing a reaction 2. Provides information about the reaction – Formulas of reactants and products – States of reactants and products – Relative numbers of reactant and product molecules that are required – Can be used to determine amounts of the reactants and products CHEMICAL EQUATIONS A chemical equation is a shorthand expression for a chemical reaction. Word equation: Aluminum combines with ferric oxide to form iron and aluminum oxide. Chemical equation: Al + Fe2O3 Fe + Al2O3 8 CHEMICAL EQUATIONS Reactants are separated from products by an arrow. Al + Fe2O3 Fe + Al2O3 Coefficients are placed in front of substances to balance the equation. 2 Al + Fe2O3 2 Fe + Al2O3 Subscripts 9 CHEMICAL EQUATIONS Reaction conditions are placed over the arrow. Al + Fe2O3 Fe + Al2O3 heat The physical state of the substances are indicated by the symbols (s), (l), (g), (aq). 2 Al (s) + Fe2O3 (s) 2 Fe (l) + Al2O3 (s) solid liquid 10 Symbols Used in Equations 1. energy symbols used above the arrow for decomposition reactions – = heat – hn = light – shock = mechanical – elec = electrical BALANCING EQUATIONS A balanced equation contains the same number of atoms on each side of the equation, and therefore obeys the law of conservation of mass. Many equations are balanced by trial and error; but it must be remembered that coefficients can be changed in order to balance an equation, but not subscripts of a correct formula. 12 BALANCING EQUATIONS The general procedure for balancing equations is: Write the unbalanced equation: CH4 + O2 CO2 + H2O Make sure the formula for each substance is correct 13 BALANCING EQUATIONS The general procedure for balancing equations is: Balance by inspection: CH4 + O2 CO2 + H2O Count and 1C compare each element on 4both H sides of the 2O equation = 1C 2H 3O 14 BALANCING EQUATIONS Balance elements that appear only in one substance first. Balance H 4 H present on each side CH4 + O2 CO2 + H2O 1 CH4 + O2 CO2 + 2 H2O 15 BALANCING EQUATIONS Balance O When finally done, check the 4 Ofor present smallest coefficientson possible each side 1 CH4 + O2 CO2 + 2 H2O 1 CH4 + 2 O2 CO2 + 2 H2O 16 Examples: 2 AgNO AgNO33 ++ H H22SS Ag Ag22SS ++ 2HNO HNO 3 3 2 Al(OH) Al(OH)3 3++3 H2SO4 Al2(SO4)3 + 6H2H O2O FeFe 4H H22 3Fe Fe ++ H42H O2O 3O 3O 4 4+ + 2 C4CH410 H10+ + 13 O O22 8CO CO 10 2 2+ + H 2OH2O 17 TYPES OF CHEMICAL REACTIONS Chemical reactions are can be classified into five types: Based on what the atoms do 1. Synthesis or combination 2. Decomposition 3. Single replacement 4. Double replacement 5. Combustion 18 SYNTHESIS or COMBINATION In these reactions, 2 elements or compounds combine to form another compound. A + B AB 19 DECOMPOSITION In these reactions, a compound breaks up to form 2 elements or simpler compound. AB A +B 20 SINGLE REPLACEMENT In these reactions, a more reactive element replaces a less reactive element in a compound. A + BC B + AC 21 DOUBLE REPLACEMENT The In these cation reactions, from onetwo compound compounds replaces combine the cation to in form another two new compound. compounds. + + AB + CD AD + CB 22 COMBUSTION A reaction that involves oxygen as a reactant and produces large amounts of heat is classified as a combustion reaction. CH4 (g) + 2 O2 (g) CO2 (g) + 2 H2O (g) 23 Examples: Classify each of the reactions below: 1. 2. 3. 4. Decomposition Mg + CuCl2 MgCl2 + Cu CaCO3 CaO + CO2 Synthesis Single replacement 2 HCl + Ca(OH)2 CaCl2 + 2 H2O reactive 4 FeMg + 3isOmore Fe2O3 than Cu 2 2 Double replacement 24 Single Displacement The Zinc Replaces the Copper Zn(s) CuCl 2 (aq) Cu(s) ZnCl 2 ACTIVITY SERIES OF METALS Activity series is a listing of metallic elements in descending order of reactivity. Hydrogen is also included in the series since it behaves similar to metals. Activity series tables are available in textbooks and other sources. 26 ACTIVITY SERIES OF METALS Elements listed higher will displace any elements listed below them. For example Na will displace any elements listed below it from one of its compounds. 2 Na (s) + MgCl2 (aq) 2 NaCl (aq) + Mg (s) 2 Na (s) + AgCl (aq) NaCl (aq) + Ag (s) 27 ACTIVITY SERIES OF METALS Elements listed lower will not displace any elements listed above them. For example Ag cannot displace any elements listed above it from one of its compounds. Ag (s) + CuCl2 (aq) No Reaction Ag (s) + HCl (aq) No Reaction 28 Example 1: Use activity series to complete each reaction below. If no reaction occurs, write “No Reaction”. Pb (s) + 2 HCl (aq) PbCl2 (aq) + H2 (g)Metals Pb is more reactive than H Fe Ni Sn Pb H Cu Ag 29 Example 2: Use activity series to complete each reaction below. If no reaction occurs, write “No Reaction”. Ni (s) + CuCl2 (aq) NiCl2 (aq) + Cu (s)Metals Ni is more reactive than Cu Fe Ni Sn Pb H Cu Ag 30 AQUEOUS REACTIONS These Many substances ionic solids are dissolve calledinelectrolytes. water to form ions. NaCl(s) Na+(aq) + Cl(aq) H2O K 2CrO4 (s) 2K H2 O + (aq) Ba(NO3 )2 (s) Ba H2O 24 (aq) + CrO 2+ (aq) 3 (aq) + 2 NO 31 AQUEOUS REACTIONS When ionic substances dissolve in water they separate into ions. K2CrO4 Ba(NO3)2 32 AQUEOUS REACTIONS electrolytes are substances whose water solution is a conductor of electricity electrolytes are ions dissolved in water (Na+ + Cl-) 33 Types of Electrolytes • salts = water soluble ionic compounds • acids = form H+1 ions in water solution – react and dissolve many metals – strong acid = strong electrolyte, weak acid = weak electrolyte • bases = water soluble metal hydroxides – increases the OH-1 concentration When will a Salt Dissolve? • a compound is soluble in a liquid if it dissolves in that liquid – NaCl is soluble in water • a compound is insoluble if a significant amount does not dissolve in that liquid – AgCl is insoluble in water • though there is a very small amount dissolved, but not enough to be significant AgCl remains solid = precipitate AQUEOUS REACTIONS Aqueous reactions occur only when one of the following conditions is present: 1. Formation of a solid: Precipitation 2. Formation of water: Neutralization 3. Formation of a gas: Unstable product 36 PRECIPITATION REACTIONS An aqueous chemical reaction that produces a solid as one of its products is called a precipitation reaction. The insoluble solid formed in these reactions is called a precipitate. K 2CrO4 (aq) + Ba(NO3 )2 (aq) BaCrO4 (s) + 2 KNO3 (aq) Precipitate 37 Example of a Precipitation Reaction Pb(NO3)2(aq) + 2 KI(aq) 2 KNO3(aq) + PbI2(s) Let’s Look Closer at PbI2 Formation Pb(NO3)2(aq) + 2 KI(aq) 2 KNO3(aq) + PbI2(s) SOLUBILITY RULES Chemists use a set of solubility rules to predict No exceptions S whetherNO 3 a product is soluble or insoluble. O Na+, K+ No exceptions L + NH4 U + Except those containing Ag B Cl, Br, I , Pb2+ L Except those containing 2 E SO4 Ba2+ , Pb2+, Ca2+ 40 SOLUBILITY RULES I N S O L S2, CO32 PO43 Except those containing Na+ , K+, NH4+ OH Except those containing Na+ , K+, Ca2+, NH4+ 41 Example 1: Write balanced equations for each reactions shown below. Indicate if no reaction occurs. NaCl (aq) + AgNO3 (aq) NaNO3 (?) + AgCl (?) NaCl (aq) + AgNO3 (aq) NaNO3 (aq) + AgCl (s) soluble precipitate 42 Example 2: Write balanced equations for each reactions shown below. Indicate if no reaction occurs. NH4Cl (aq) + KNO3 (aq) NH4NO3 (?) + KCl (?) No4NO Reaction NH4Cl (aq) + KNO3 (aq) NH 3 (aq) + KCl (aq) soluble 43 Example 3: Write balanced equations for each reactions shown below. Indicate if no reaction occurs. PbCl2 (aq) + 2 NaI (aq) PbI2 (?) + 2 NaCl (?) PbCl2 (aq) + 2 NaI (aq) PbI2 (s) + 2 NaCl (aq) precipitate 44 Molecular, Complete Ionic, and Net Ionic Equations A molecular equation is a chemical equation showing the complete, neutral formulas for every compound in a reaction. A complete ionic equation is a chemical equation showing all of the species as they are actually present in solution. A net ionic equation is an equation showing only the species that actually participate in the reaction. © 2012 Pearson Education, Inc. Writing Chemical Equations for Reactions in Solution: Molecular and Complete Ionic Equations • A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. • Complete ionic equations show aqueous ionic compounds that normally dissociate in solution as they are actually present in solution. • When writing complete ionic equations, separate only aqueous ionic compounds into their constituent ions. • Do NOT separate solid, liquid, or gaseous compounds. © 2012 Pearson Education, Inc. Writing Chemical Equations for Reactions in Solution: Net Ionic Equations • In the complete ionic equation, some of the ions in solution appear unchanged on both sides of the equation. • These ions are called spectator ions because they do not participate in the reaction. © 2012 Pearson Education, Inc. Writing Chemical Equations for Reactions in Solution: Proper Net Ionic Equations • To simplify the equation, and to more clearly show what is happening, spectator ions can be omitted. • Equations such as this one, which show only the species that actually participate in the reaction, are called net ionic equations. Ag+(aq) + Cl− (aq) AgCl(s) © 2012 Pearson Education, Inc. NEUTRALIZATION REACTIONS The Saltsmost are ionic important substances reaction withofthe acids cation and bases is called donated from the neutralization. base and the anion donated thereactions acid. from In these an acid combines with a base to form a salt and water. HCl (aq) + NaOH (aq) ¾ ¾ ® NaCl (aq) + H 2O (l) Acid Base Salt 49 Examples: Write balanced equations for each of the neutralization reactions shown below: 2 HNO3 + Ba(OH)2 Ba(NO3)2 + 2 H2O H2SO4 + 2 NaOH Na2SO4 + 2 H2O 50 GAS FORMING REACTIONS Some chemical reactions produce gas because one of the products formed in the reaction is unstable. Three such products are: Carbonic acid: H2CO3 (aq) CO2 (g) + H2O (l) Sulfurous acid: H2SO3 (aq) SO2 (g) + H2O (l) Ammonium: NH4OH (aq) NH3 (g) + H2O (l) 51 GAS FORMING REACTIONS When either of these products appears in a chemical reaction, they should be replaced with their decomposition products. 22HNO HCl + Na K22CO SO3 2 KNO NaCl 3++ HH2CO 3+ 2SO33 2 HNO 2 HCl3 + Na2SO CO33 22 KNO NaCl3 ++ CO SO2 (g) (g)++ H H22O O (l) (l) 52 Enthalpy: A Measure of the Heat Evolved or Absorbed in a Reaction • Chemical reactions can be exothermic (they emit thermal energy when they occur). • Chemical reactions can be endothermic (they absorb thermal energy when they occur). • The amount of thermal energy emitted or absorbed by a chemical reaction, under conditions of constant pressure (which are common for most everyday reactions), can be quantified with a function called enthalpy. © 2012 Pearson Education, Inc. Enthalpy: A Measure of the Heat Evolved or Absorbed in a Reaction • We define the enthalpy of reaction, ΔHrxn, as the amount of thermal energy (or heat) that flows when a reaction occurs at constant pressure. © 2012 Pearson Education, Inc. Sign of ΔHrxn • The sign of ΔHrxn (positive or negative) depends on the direction in which thermal energy flows when the reaction occurs. • Energy flowing out of the chemical system is like a withdrawal and carries a negative sign. • Energy flowing into the system is like a deposit and carries a positive sign. © 2012 Pearson Education, Inc. Exothermic and Endothermic reactions • (a) In an exothermic reaction, energy is released into the surroundings. (b) In an endothermic reaction, energy is absorbed from the surroundings. © 2012 Pearson Education, Inc. HEAT IN CHEMICAL REACTIONS Reactions In exothermic that reaction, release heat heatare is produced classified as and can exothermic. be written as a product. In Reactions endothermic that absorb reaction, heat heat areisclassified requiredas and can be written as a reactant. endothermic. Endothermic H2 (g) + Cl2 (g) 2 HCl (g) + 185 kJ N2 (g) +Exothermic O2 (g) + 181 kJ 2 NO (g) 57 Sign of ΔHrxn • When thermal energy flows out of the reaction and into the surroundings it is a ??? reaction and has a + or – enthalpy? • The enthalpy of reaction for the combustion of CH4, the main component in natural gas: • The magnitude of ΔHrxn tells us that 802.3 kJ of heat are emitted when 1 mol CH4 reacts with 2 mol O2. © 2012 Pearson Education, Inc. Stoichiometry of ΔHrxn • The amount of heat emitted or absorbed when a chemical reaction occurs depends on the amounts of reactants that actually react. • We usually specify ΔHrxn in combination with the balanced chemical equation for the reaction. • The magnitude of ΔHrxn is for the stoichiometric amounts of reactants and products for the reaction as written. © 2012 Pearson Education, Inc. Stoichiometry of ΔHrxn • The balanced equation and ΔHrxn for the combustion of propane is: • When 1 mole of C3H8 reacts with 5 moles of O2 to form 3 moles of CO2 and 4 moles of H2O, 2044 kJ of heat are emitted. © 2012 Pearson Education, Inc. Example 1: • An gas tank in a home barbecue contains 11.8 x 103 g of propane (C3H8). • Calculate the heat (in kJ) associated with the complete combustion of all of the propane in the tank. © 2012 Pearson Education, Inc. Example 1: © 2012 Pearson Education, Inc. Classifying Reactions • Also we can classify reactions by what happens: • Redox reactions are the exchange of e• Redox are all reactions except? Note OXIDATION-REDUCTION REACTIONS In an oxidation-reduction Reactions known as oxidation reaction, and reduction electrons are (redox) havefrom transferred many one important substance applications to another.in our everyday If one substance lives. loses electrons, another substance Rusting must gain of electrons. a nail or the reaction within your car batteries are two examples of redox reactions. 64 OXIDATION-REDUCTION REACTIONS Oxidation is defined as loss of electrons, and reduction is defined as gain of electrons. One way to remember these definitions is to use the following mnemonic: Oxidation Is Loss of electrons Reduction Is Gain of electrons OIL RIG Combination, decomposition, single replacement and combustion reactions are all examples of redox reactions. 65 OXIDATION-REDUCTION REACTIONS In Forgeneral, example, atoms in the offormation metals lose of electrons calcium to form cations, sulfide from calcium and areand therefore sulfur oxidized, while atomsCa of +non-metals gain electrons to S CaS form anions, and are therefore reduced. Ca2+ + 2 e S + 2 eS2- Ca Oxidation Reduction Therefore, the formation of calcium sulfide involves two half-reactions that occur simultaneously, one an oxidation and the other 66 a reduction. COMBUSTION A reaction that involves oxygen as a reactant and produces large amounts of heat is classified occurs in as a combustion reaction. the cylinders of the engine Combustion reactions are a subclass of Oxidation-Reduction reactions 2 C8H18(g) + 25 O2(g) 16 CO2(g) + 18 H2O(g) 67 Combustion Products • predicting the products of a combustion reaction; simply combine each element in the other reactant with oxygen Reactant Combustion Product contains C CO2(g) contains H H2O(g) contains S SO2(g) contains N NO(g) or NO2(g) contains metal M2On(s) Combustion Reactions • combustion reactions are always exothermic • in combustion reactions, O2 combines with the elements in another reactant to make the products 4 Fe(s) + 3 O2(g) → 2 Fe2O3(s) + energy CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g) + energy The flame on a gas stove results from the oxidation of carbon in natural gas. Reverse of Combustion Reactions • since combustion reactions are exothermic, their reverse reactions are endothermic • the reverse of a combustion reaction involves the production of O2 energy + 2 Fe2O3(s) → 4 Fe(s) + 3 O2(g) energy + CO2(g) + 2 H2O(g) → CH4(g) + 2 O2(g) • reactions in which O2 is gained or lost are redox reactions REDOX IN BIOLOGICAL SYSTEMS Many important biological reactions involve oxidation and reduction. In these reactions, oxidation involves addition of oxygen or loss of hydrogen, and reduction involves Oxidation loss of oxygen or gain of hydrogen. (loss of For example, poisonous methyl alcohol is hydrogen) metabolized by the body by the following reaction: CH3OH methyl alcohol H2CO + 2H• formaldehyde 71 REDOX IN BIOLOGICAL SYSTEMS The formaldehyde is further oxidized to formic acid and finally carbon dioxide and water by the Oxidation following reactions: (gain of 2 H2CO + O2 formaldehyde 2 H2CO2 + O2 2H2CO2 oxygen) formic acid CO2 + H2O formic acid 72 REDOX IN BIOLOGICAL SYSTEMS In Themany oxidation biochemical of a typical oxidation-reduction biochemical molecule reactions, can the transfer involve the transfer of hydrogen of two atoms hydrogen produces atoms energy to a in the cells. proton acceptor such as coenzyme FAD to produce its reduced form FADH2. 73 REDOX IN BIOLOGICAL SYSTEMS In summary, the particular definition of oxidationreduction depends on the process that occurs in the reaction. 74 THE END 75