* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download march_22_lecture
DNA vaccination wikipedia , lookup
Immune system wikipedia , lookup
Major histocompatibility complex wikipedia , lookup
Complement system wikipedia , lookup
Adaptive immune system wikipedia , lookup
Innate immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Molecular mimicry wikipedia , lookup
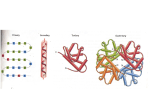
What makes a microorganism or virus a significant human pathogen? • Ability to effectively cause infection i.e. genes expressed which facilitate entry into body • Ability to effectively replicate in the body and evade immune response • Ability to effectively exit body in a form which can be transmitted directly or indirectly to a new human host • Ability to produce gene products which cause pathological effects such as toxins and/or superantigens Pathogenic Organisms are Cleared by the Immune System in Progressively More Specific Ways as Infection Progresses QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see thi s picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. CL0NAL SELECTION 1. REMOVE ANTIBODIES AGAINST SELF 2. SELECT B CELLS WHICH HAVE INITIAL AFFINITY FOR ANTIGEN 3. SELECT B CELLS WITH HIGHER AND HIGHER AFFINITY FOR ANTIGEN 4. SELECT B CELLS WHICH PRODUCE SPECIFIC TYPE OF ANTIBODY QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. Expression of the Antibody Molecule as a Surface Antigen Allows Clonal Selection to Occur QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. VDJ rearrangement is the first step in generating antibody which has affinity for antigen QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. VDJ recombination mediated by RAG recombinase brings together V,D and J segments to form a functional coding region while generating significant diversity QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Mutation in 3 hypervariable regions of variable chains allows antibody with higher affinity to be produced Clonal selection leads to mutliplication of high affinity clones Single base changes occur at a rate of 10-3 to 10-5 per base pair per cell generation in B cells undergoing somatic hypermutation AID=activation induced cytidine deaminase UNG=Uracil N glycosylase (removes uracil from DNA) QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. Specific regions of variable regions are targets of somatic mutation Somatic mutation continues until high affinity antibodies are selected Constant region of antibody molecules controls function of antibody DNA recombination used to switch antibody type RECOMBINATION IN HEAVY CHAIN GENES USED FOR CLASS SWITCHING INITIAL ANTIB0DYCu IgM ANTIBODY SECRETED and Cd IgD ANTIBODY ON CELL SURFACE PRODUCED BY ALTERNATIVE SPLICING LATER ANTIBODY Cg--IgG or Ca IgA or Ce IgE -SECRETED IgG ANTIBODIES CAUSE EFFICIENT DESTRUCTION OF BACTERIA BECAUSE THEY BIND TO Fc RECEPTOR AND PROMOTE OPSONIZATION AND PHAGOCYTOSIS The Cells of the Immune System Derive from a Common Stem Cell: they Cooperate to Carry Out the Immune Response QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. T-LYMPHOCYTES: Can play a role in stimulating B cells to produce antibody or activating macrophages (Helper T cells--CD4+) OR Can act directly to kill a cell expressing a foreign antigen (Killer T cells--CD8+) QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Killer T cells (CD8+) React to a Viral Antigen Presented by Class I MHC and Causes Virus Infected Cells to be Killed QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. A T cell recognizes target antigens through the T cell receptor (TCR) The T cell receptor resembles a membrane bound immunoglobulin molecule QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Structure of the T-cell receptor The T-cell receptor heterodimer is composed of two transmembrane glycoprotein chains, a and b. The extracellular portion of each chain consists of two domains, resembling immunoglobulin V and C domains, respectively. Both chains have carbohydrate side chains attached to each domain. A short segment, analogous to an immunoglobulin hinge region, connects the immunoglobulin-like domains to the membrane and contains the cysteine residue that forms the interchain disulfide bond. T-cell receptor a and b chain gene rearrangement and expression The TCR a and b chain genes are composed of discrete segments that are joined by somatic recombination during development of the T cell. For the a chain a Va gene segment rearranges to a Ja gene segment to create a functional V-region exon. Transcription and splicing of the VJa exon to Ca generates the mRNA that is translated to yield the T-cell receptor a chain protein. For the b chain the variable domain is encoded in three gene segments, Vb, Db, and Jb. Rearrangement of these gene segments generates a functional VDJb V-region exon that is transcribed and spliced to join to Cb; the resulting mRNA is translated to yield the T-cell receptor b chain. The a and b chains pair soon after their biosynthesis to yield the a : b T-cell receptor heterodimer. A killer T-cell must receive its initial encounter with antigen accompanied by a costimulating signal from a professional antigen presenting cell (APC) which is stimulated by the innate immune system to present a second stimulatory signal to the T cell Once the killer T cell is educated by the APC, it is capable of killing cells which present the foreign antigen without a second signal QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Class I MHC Peptide-Binding Site-Peptide and Class I MHC are the target of TCR in CD8 (Killer) T cells Class I MHC Peptide-Binding Site-Peptide and Class I MHC are the target of TCR in CD8 (Killer) T cells QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The a1and a2 domains come together to form a groove in which peptides are displayed. The two views shown reveal that the peptide is surrounded on three sides by a b sheet and two a helices, but it is accessible from the top of the structure QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Peptides are bound to MHC class I molecules by their ends. MHC class I molecules interact with the back-bone of a bound peptide through a series of hydrogen bonds and ionic interactions at each end of the peptide. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Peptides bind to MHC molecules through structurally related anchor residues. Peptides eluted from two different MHC class I molecules are shown. The anchor residues (green) differ for peptides that bind different alleles of MHC class I molecules but are similar for all peptides that bind to the same MHC molecule. The upper and lower panels show peptides that bind to two different alleles of MHC class I molecules. The anchor residues that bind a particular MHC molecule need not be identical, but are always related (for example, phenylalanine (F) and tyrosine (Y) are both aromatic amino acids, whereas valine (V), leucine (L), and isoleucine (I) are all large hydro-phobic amino acids). Peptides also bind to MHC class I molecules through their amino (blue) and carboxy (red) termini. The Recognition of an Antigen by CD8+ T cells is Influenced by the Allele at the MHC Locus which Presents the Antigen QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. C4+Helper T Cells Stimulate B Cells to Produce Antibodies TH1 and helper T cells recognize antigen presented by MHC class II molecules QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. On recognition of their specific antigen on infected macrophages, TH1 cells activate the macrophage, leading to the destruction of the intracellular bacteria. When helper T cells recognize antigen on B cells, they activate these cells to proliferate and differentiate into antibody-producing plasma cells Antigen that binds to the Bcell antigen receptor signals B cells and is, at the same time, internalized and processed into peptides that activate armed helper T cells. Signals from the bound antigen and from the helper T cell induce the B cell to proliferate and differentiate into a plasma cell secreting specific antibody QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Armed helper T cells stimulate the proliferation and then the differentiation of antigen-binding B cells QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The specific interaction of an antigen-binding B cell with an armed helper T cell leads to the expression of the B-cell stimulatory molecule CD40 ligand (CD40L) on the helper T-cell surface and to the secretion of the B-cell stimulatory cytokines IL-4, IL-5, and IL-6, which drive the proliferation and differentiation of the B cell into antibody-secreting plasma cells. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Peptides that bind MHC class II molecules are variable in length and their anchor residues lie at various distances from the ends of the peptide. The sequences of a set of peptides that bind to the mouse MHC class II Ak allele are shown in the upper panel. All contain the same core sequence but differ in length. In the lower panel, different peptides binding to the human MHC class II allele HLA-DR3 are shown. The lengths of these peptides can vary, and so by convention the first anchor residue is denoted as residue 1. Note that all of the peptides share a negatively charged residue (aspartic acid (D) or glutamic acid (E)) in the P4 position (blue) and tend to have a hydrophobic residue (for example, tyrosine (Y), leucine (L), proline (P), phenylalanine (F)) in the P9 position (green). Constant region of antibody molecules control function of antibody DNA recombination used to switch antibody type IgG ANTIBODIES CAUSE EFFICIENT DESTRUCTION OF BACTERIA BECAUSE THEY BIND TO Fc RECEPTOR AND PROMOTE OPSONIZATION AND PHAGOCYTOSIS Bacterial Capsules Can Protect Against Macrophage and Complement Mediated Lysis How Some Pathogens Evade Macrophage Phagocytosis • QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Some pathogens such as Pseudomonas, enteropathogenic E. coli (EPEC), and Yersinia can block phagocytic uptake into macrophages. By translocating specialized type III effector proteins into the macrophage cell, these pathogens interrupt the signal transduction cascades necessary for actin rearrangement and thereby prevent phagocytosis. Other pathogens that use a type IV secretion system (such as Brucella and Legionella), or type III secretion systems (such as Salmonella, and probably Chlamydia), can induce their own uptake into macrophages by stimulating actin rearrangements proximal to attached bacteria, and then secrete additional effectors into the host cell to modify the trafficking of the invasion vacuole in order to prevent fusion with lysosomes. Vacuoles containing Legionella and Brucella, for example, then acquire markers of the endoplasmic reticulum, thereby creating a protective niche for replication within cells. COMPLEMENT SYSTEM PLAYS AN IMPORTANT ROLE IN RESPONSE TO PATHOGENS QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The first protein in the classical pathway of complement activation is C1, which is a complex of C1q, C1r, and C1s. C1q is composed of six identical subunits with globular heads and long collagen-like tails. The tails combine to bind to two molecules each of C1r and C1s, forming the C1 complex C1q:C1r2:C1s2. The heads can bind to the constant regions of immunoglobulin molecules or directly to the pathogen surface, causing a conformational change in C1r, which then cleaves and activates the C1s zymogen. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see thi s picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The constant regions of IgM and some IgGs contain a binding site for C1q. Binding of C1q activates C1s and C1r. Activated C1s (a serine protease) cleaves two serum proteins C4 is cleaved into a large fragment C4b, which binds covalently to sugar residues on cell-surface glycoproteins, and a smaller, inactive, fragment C4a which diffuses away. C2 is cleaved into C2b, which binds noncovalently to a site on C4b, leaving a smaller, inactive, fragment of C2a which diffuses away. The complex of C4b•2b catalyzes the cleavage of C3. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. C3 is the most abundant protein of the complement system (~1.2 mg/ml). Because of its abundance and ability to activate itself it greatly magnifies the response. C4b•2b cuts C3 into two major fragments--C3b and C3a. C3b, binds covalently to glycoproteins scattered across the bacterial cell surface. Macrophages and neutrophils have receptors for C3b and can bind the C3bcoated cell or particle preparatory to phagocytosis. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. C3a, a small fragment of C3, is released into the surrounding fluids. It can bind to receptors on basophils and mast cells triggering them to release histamine and other contents of their vacuoles. When C3a is released locally at the site of infection it causes an inflammatory response attracting more immune system cells to the site of the infection. However, C3a, if released throughout the body (systemic release) can cause anaphylaxis. Complement component C5 is cleaved when captured by a C3b molecule that is part of a C5 convertase complex C5 convertases are formed when C3b binds either C4b,2b to form C4b,2b,3b, or C3b,Bb to form C3b2,Bb. C5 binds to the C3b in these complexes. C5 is cleaved by the active enzyme C2b or Bb to form C5b and the inflammatory mediator C5a which works like C3a. The production of C5b initiates the membrane attack complex. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The Membrane Attack Complex: Lysis Bacteria By Creating Pores QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. C5b triggers the assembly of a complex of one molecule each of C6, C7, and C8, in that order. C7 and C8 undergo conformational changes that expose hydrophobic domains that insert into the membrane. This complex causes moderate membrane damage in its own right, and also serves to induce the polymerization of C9, again with the exposure of a hydrophobic site. Up to 16 molecules of C9 are then added to the assembly to generate a channel of 100 Å diameter in the membrane. This channel disrupts the bacterial cell membrane, killing the bacterium. The electron micrographs show erythrocyte membranes with membrane-attack complexes in two orientations, end on and side on. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The Iditarod: Commemorating the 1925 Emergency Delivery of Diphtheria Serum to Nome, Alaska The First Signs of an Epidemic Nome, Alaska in 1925 Doctor Curtis Welch Qui ckTime™ and a TIFF (U ncompr essed) decompressor are needed to see thi s pi cture. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. It was a normal mid-January afternoon in Nome. Doctor Curtis Welch, physician and director of the US Public Health Service, was doing paperwork in his office at the Merchants and Miners Bank of Alaska building. An Innuit man came into the office asking the doctor to come quickly, his two children were very sick. Dr. Welch raced to the Sand Spit Innuit settlement, west of the Snake River on the fringes of Nome. The children's temperatures were dangerously high, and their breathing was labored and shallow. Dr. Welch asked the mother how the children had become ill and what their symptoms had been. She replied that they had been sick for about three days. She thought it was a bad cold because their throats had become red and sore. Dr. Welch tried to examine their throats, but they could not open their mouths far enough for him to do so. He tried to comfort the mother and then returned to his office.Dr. Welch had wished many times that he had access to a good laboratory where he could send specimens for analysis. It was very strange. Children don't die of sore throats, but the two Innuit children were dying. At one point he considered diphtheria but it was highly unlikely. He hadn't seen a case in northern Alaska in twenty years. Despite the doctor's efforts the Innuit children died the following day. http://www.lucidca fe.com/library/idita rod.html Recognizing diptheria and the need for antitoxin QuickTi me™ a nd a TIFF (Uncompre ssed ) decomp resso r are need ed to se e th is p icture. A few days later, on January 21, Dr. Welch was called to the home of a family in Nome to examine their six-year-old son. The child had been sick for two days with a sore throat. Dr. Welch examined the boy's throat and recognized immediately the dirty white patches of the diphtheria membrane. The doctor realized the terrible implications of this diagnosis. Diphtheria, left unchecked, would spread with devastating speed. Dr. Welch met at once with Nome's mayor and city council. He told them of the imminent epidemic and stressed that some way must be found to get the diphtheria antitoxin to Nome within 2 weeks. The serum would check the spread of the disease and would help those already infected. His main concern was with the native population that had little or no immunity to diseases from the outside. A flu epidemic in 1919 had wiped out entire Innuit villages. http://www.lucidcafe.co m/library/iditarod.html QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. • Diptheria antitoxin reaches Nome by Dogsled in Five Days There was widespread relief when it was discovered that the Alaska Railroad Hospital in Anchorage had 300,000 units of the life saving serum. It was not much, but it would be enough to stem the tide of the epidemic. It was decided to transport the serum by train from Anchorage to Nenana, a town on the Tanana River 220 miles north of Anchorage, and then by a relay of dog teams over the 674 miles between Nenana and Nome. The last leg reached Nome on Monday, February 2 at 5:30 in the morning. Dr. Welch was awakened by a persistent knocking on his front door. When he opened it he found an exhausted Gunnar Kaasen, the musher of the final leg of the relay. Kaasen handed him a twenty pound, fur-andcanvas-covered package containing the 300,000 units of serum. In the street were his 13 dogs harnessed to a sled, their heads and bushy tails hanging almost to the ground. They had covered the last fifty-three miles of the epic relay in seven and a half hours. These dogs, and the teams that preceded them, had traversed 674 ice-and-snow covered miles in less than five days. They delivered to Dr. Welch the life-saving serum that within a week would break the back of the diphtheria epidemic. QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Diphtheria • Diphtheria is still common in many other parts of the world, including the Caribbean and Latin America. During the last few years, large epidemics of diphtheria have occurred in the former Soviet republics. Outbreaks have also been reported in Algeria, China, and Ecuador. The majority of cases in many of these epidemics have been in adults and adolescents. http://www.astdhpphe.org/infect/dip.html Quic kTime™ and a TIFF (Unc ompres sed) decompress or are needed to see this picture. Corynebacterium diphtheriae Diphtheria QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. http://gsbs.utmb.edu/microbook/ch032.htm Diphtheria QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. http://gsbs.utmb.edu/microbook/ch032.htm BACTERIAL EXOTOXINS CAN INCLUDE A POLYPEPTIDE (B) DESIGNED FOR CELL ENTRY AND A SECOND POLYPEPTIDE (A) WHICH CAUSES TOXICITY TO HUMAN CELL TOXIN CAN ENTER CELL THROUGH ENDOCYTOSIS OR DIRECTLY DIPHTHERIA TOXIN: TARGET RIBOSOMAL PROTEIN OF HUMAN CELL--MOST SERIOUS TOXIC EFFECT, CARDIOTOXICITY DIPHTHERIA TOXIN INACTIVATES PROTEIN SYNTHESIS BY ADP RIBOSYLATION OF TRANSLATION FACTOR EF-2 AT A SPECIFIC MODIFIED HISTIDINE RESIDUE Bacterial Toxins • Endotoxins-bacterial lipopolysaccharides • Exotoxins--specific polypeptides produced by bacteria which cause toxic effects BACTERIAL EXOTOXIN GENES ARE OFTEN ACQUIRED BY GENE TRANSFER PATHOGENIC E. COLI • ENTERTOXIGENIC (ETEC) (secretory diarrhea) • ENTEROPATHOGENIC (Malabsorptive diarrhea) (EPEC) • ENTEROHEMORAGGHIC (Malabsorptive diarrhea and dysentery) (EHEC) (E. coli 0157) All three types are major causes of infant deaths worldwide MECHANISM OF ACTION ST AND LT TOXINS IN ETEC INTERFERENCE WITH cAMP or cGMP METABOLISM LEADS TO LOSS OF CONTROL OVER WATER FLOW AND INTESTINAL WATER LOSS QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. Cholera QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. • • • • • • • QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. • The profuse diarrhea and subsequent fluid and electrolyte loss associated with infection with enterotoxigenic Escherichia coli and Vibrio cholerae arise following binding of the luminal epithelium of the intestine by the toxins Etx and Ctx, respectively (a) Binding to the cell is mediated by the ability of the B subunit of the toxin to interact and crosslink with ganglioside GM1 receptors. (b) Crosslinking triggers vesicular uptake and (c) Trafficking, a process that is also influenced by the presence of an RDEL (or KDEL in the case of Ctx) sequence on the A2 fragment (d) Enzymatic cleavage allows the A1 fragment to enter the cytosol, where (e) it catalyses adenosine diphosphate (ADP) ribosylation of GSa, a membrane G protein that regulates the activation of adenylate cyclase. (f) Once activated, adenylate cyclase accelerates the conversion of adenosine triphosphate (ATP) to cyclic adenosine monophosphate (cAMP), which subsequently induces phosphorylation of protein kinase A (PKA) and leads to the opening of the cystic fibrosis transmembrane regulator (CFTR) chloride ion (Cl–) channel. (g) Efflux of Cl– from the cell results in a simultaneous osmotic shift of water from the cell into the intestinal lumen, accompanied by cell death. This results in the watery diarrhea characteristic of infection by V. cholerae and E. coli DELIVERY OF EPEC TOXIC PROTEIN TO HUMAN INTESTINAL CELLS BY A TYPE III SECRETION SYSTEM EPEC PILI BIND TO INTESTINAL EPITHELAL CELLS; CHANNEL IS FORMED MAKING DIRECT CONNECTION BETWEEN EPEC CYTOPLASM AND INTESTINAL CELL CYTOPLASM TOXIC PROTEIN INJECTED WITHOUT EXPOSURE TO HUMAN IMMUNE SYSTEM Effects of EPEC infection on host intestinal epithelial cells • • • QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. • • • • EPEC initially adheres to the host cell by its bundleforming pili, which also mediate bacterial aggregation. Following initial attachment, EPEC secretes several virulence factors by a type III-secretion system. Signal transduction events occur within the host, including activation of phospholipase C (PLC) and protein kinase C (PKC), inositol triphosphate (IP3) fluxes, and Ca2+ release from internal stores. The bacterium intimately adheres to the cell by secreting its own receptor, Tir, into the host and binding to it with its outer membrane ligand, intimin. Intimin can also bind beta1-integrins. Several cytoskeletal proteins are recruited to the site of EPEC attachment, including actin, alpha-actinin, talin, and ezrin. Cytoskeletal rearrangements occur following Tirintimin binding, resulting in the formation of a pedestal-like structure upon which the pathogen resides. EHEC • Shiga like toxin circulates through bloodstream, enters kidney and causes kidney damage Pedestal Formation by EPEC and EHEC occurs by different mechanisms • • • • • • EPEC Tir becomes tyrosine-phosphorylated in the host-cell plasma membrane and binds the adaptor protein Nck. Nck recruits N-WASP or a WIPミN-WASP complex to trigger activation of the Arp2/3 complex, which leads to actin assembly. EHEC Tir localizes to the plasma membrane, but is not tyrosine phosphorylated. Other EHEC proteins (X) in addition to Tir are translocated into host cells. This combination of Tir and other factors promotes recruitment and activation of N-WASP by an unidentified mechanism (designated with a question mark). N-WASP then stimulates Arp2/3-based actin QuickTime™ and a TIFF (Uncompressed) decompressor are needed to see this picture. The Immune System is Active in the Intestine QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. • M cells and dendritic cells are specialized cells which collect antigen • Some pathogens have systems which allow them to invade M cells and pass through into the body PATHOGENICITY OF SALMONELLA Salmonella invasion into host epithelial cells QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. • Salmonella secrete virulence proteins, including SopE and SptP, into host cells by the type IIIsecretion system. • SopE functions as a guanidine exchange factor for small GTP-binding proteins, probably mediating the exchange of GDP for GTP on a Rho subfamily member, CDC42. • SptP is a tyrosine phosphatase required for invasion, probably by disrupting the cytoskeleton. • Invasion also stimulates phospholipase C (PLC) activity, leading to inositol triphosphate (IP3) and Ca2+ fluxes, which in turn may be involved in cytoskeletal rearrangements leading to membrane ruffling and Salmonella internalization. Shigella-mediated cytoskeletal rearrangements • • • • QuickTime™ and a TIFF (LZW) decompressor are needed to see this picture. • The outer membrane protein, IcsA, is sufficient to drive actin-based motility of Shigella. IcsA directly binds two proteins, vinculin and neural-Wiskott-Aldrich Syndrome protein (N-WASP). Vinculin undergoes proteolysis within the host cell upon Shigella infection, producing a 90-kDa fragment that can bind to IcsA and to the vasodilatorstimulated phosphoprotein (VASP). VASP in turn can recruit profilin to the bacterial surface, which can provide actin for tail construction. N-WASP binding of IcsA can also recruit profilin to the bacterial surface and may be another means of obtaining monomeric actin for tail formation and subsequent bacterial motility. Viral Gastroenteritis • It is thought that viruses are responsible for up to 3/4 of all infective diarrheas. • Viral gastroenteritis is the second most common viral illness after upper respiratory tract infection. • In developing countries, viral gastroenteritis is a major killer of infants who are undernourished. Rotaviruses are responsible for half a million deaths a year. • Many different types of viruses are found in the gut but only some are associated with gastroenteritis. Rotavirus Particle (Courtesy of Linda Stannard, University of Cape Town, S.A.) Rotaviruses • Naked double stranded RNA viruses, 80 nm in diameter. • Also found in other mammals and birds, causing diarrhea. • Causes disease in all age groups but most severe symptoms neonates and young children. in • Asymptomatic infections common in adults and older children. Symptomatic infections again common in people over 60. Rotaviruses • Accounts for 50-80% of all cases of viral gastroenteritis. • Usually endemic, but responsible for occasional outbreaks. • 80% of the US population have antibody against rotavirus by age of 3. • 2.5 million cases in children under 5 per year in US; 70,000 hospitalizations and 20-70 deaths per year in US • Up to 30% mortality rate in malnourished children • Responsible for up to half a million deaths per year, worldwide Rotaviruses • More frequent during the winter. • Fecal-oral spread. • 24-48 hr incubation period followed by an abrupt onset of vomiting and diarrhea, a low grade fever may be present. • Diagnosed by electron microscopy or by the detection of rotavirus antigens in feces by ELISA or other assays. Rotaviruses • 1998--RotaShield vaccine introduced • Increase in frequency of intussusception, a blockage or twisting of the intestine • 20 deaths attributed to vaccine in first 1,000,000 children vaccinated • Rotashield withdrawn • 2006-RotaTeq vaccine approved by FDA for US use after large scale testing--no increase in intussusception reported Adenovirus Particle (Courtesy of Linda Stannard, University of Cape Town, S.A.) Enteric Adenoviruses • Naked DNA viruses, 75 nm in diameter. • Fastidious enteric adenovirus types 40 and 41 are gastroenteritis. • Associated with cases of endemic gastroenteritis, usually in young children and neonates. Can cause occasional outbreaks. • Possibly the second most common viral cause of gastroenteritis (7-15% of all endemic cases). • Similar disease to rotaviruses • Most people have antibodies against enteric adenoviruses by the age of three. • Diagnosed by electron microscopy or by the detection of adenovirus antigens in faeces by ELISA or other assays. associated with Astrovirus Particles (Source: ICTV database) Astroviruses • • Small RNA viruses, named because of star-shaped surface morphology, 28 nm in diameter. Associated with cases of endemic gastroenteritis, usually in young children and neonates. Can cause occasional outbreaks. • Responsible for up to 10% of cases of gastroenteritis. • Similar disease to rota and adenoviruses. • Most people have antibodies by the age of three. • Diagnosed by electron microscopy only, often very difficult because of small size. Calicivirus Particles (Source: ICTV database) Caliciviruses • • • • • Small RNA viruses, characteristic surface morphology consisting of hollows. particles 35 nm in diameter. Associated mainly with epidemic outbreaks of gastroenteritis, although occasionally responsible for endemic cases. Like Norwalk type viruses, vomiting is the prominent feature of disease. Majority of children have antibodies against caliciviruses by the age of three. Diagnosed by electron microscopy only, often difficult to diagnose because of small size. Norwalk-like Virus Particles (Source: ICTV database) Norwalk-like Viruses • Small RNA viruses, with ragged surface, 35 nm in diameter, now classified as caliciviruses. • Always associated with epidemic outbreaks of gastroenteritis, adults more commonly affected than children. • Associated with consumption of shellfish and other contaminated foods. Aerosol spread possible as well as faecal-oral spread. • Also named "winter vomiting disease", with vomiting being prominent symptom, diarrhea usually mild. • Antibodies acquired later in life, in the US, only 50% of adults are seropositive by the age of 50. • Diagnosis is made by electron microscopy and by PCR. the Control of Infectious Disease • Sanitation and other environmental controls • Innate defenses of the body • Vaccination • Anti-infective therapy Sanitation as a Defense Against Infectious Disease • Once germ theory of disease was understood --sanitary engineering became a key line of defense against infectious disease Goals of Sanitary Engineering • Remove wastes safely from environment • Eliminate possible sources of contamination by infectious agents • Provide safe water, food and air