* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download particlephysics
Electric charge wikipedia , lookup
Weakly-interacting massive particles wikipedia , lookup
Quantum tunnelling wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Neutrino oscillation wikipedia , lookup
Super-Kamiokande wikipedia , lookup
Photon polarization wikipedia , lookup
Photoelectric effect wikipedia , lookup
ALICE experiment wikipedia , lookup
Quantum chromodynamics wikipedia , lookup
Double-slit experiment wikipedia , lookup
Identical particles wikipedia , lookup
Renormalization wikipedia , lookup
ATLAS experiment wikipedia , lookup
Nuclear structure wikipedia , lookup
Grand Unified Theory wikipedia , lookup
Monte Carlo methods for electron transport wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Strangeness production wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Future Circular Collider wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Mathematical formulation of the Standard Model wikipedia , lookup
Standard Model wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Atomic nucleus wikipedia , lookup
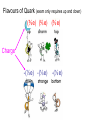
PARTICLE PHYSICS Summary Alpha Scattering & Electron Diffraction Rutherford’s picture of alpha scattering nucleus paths of scattered alpha particles The angle of deflection is determined by the K.E. of the alpha particle. The “no – go “ zone behind the nucleus gets smaller the larger the K.E. of the alpha particles Electron diffraction Utilising wave qualities of electrons i.e. diffraction sin =1.22λ d Diameter of nucleus λ = h (de Broglie) p Airy Disk Airy disk produced by electron diffraction around a spherical object (nucleus) Remember: is the angle from 0th order to 1st minimum No of electrons Rutherford scattering and electron diffraction 1/r curve predicted by Rutherford scattering Actual curve for electron scattering Angle Quantum feature of e- makes it Ch 17 Probing Deep into Matter 17.2 Scattering and scale Particle Zoo Categorisation of sub-atomic particles Boson Fermion • Half integer spin • Subject to exclusion principle • Whole integer spin • Not subject to exclusion principle • Transmit a force i.e. photons, gluons, Hadron Lepton • Made up of quarks i.e. protons, neutrons • Fundamental particle i.e. electrons, neutrinos Baryon Meson • 3 quarks i.e. proton, neutrons • Quark – antiquark i.e. 0 Flavours of Quark (exam only requires up and down) (⅔ e) (⅔ e) (⅔ e) Charge -(⅓ e) -(⅓ e) -(⅓ e) Quarks • are never seen on their own • have anti-versions of themselves Everything, other than mass, is opposite Hadron rules Quark combinations must have… • Net integer charge • Zero colour charge Ch 17 Probing Deep into Matter 17.1 Creation & annihilation Particle – Antiparticle Particles and antiparticles Same mass, everything else is opposite Electron Antielectron (Positron) Charge /C -e +e Spin -½ ½ 9.11 x 10-31 9.11 x 10-31 Mass /kg Annihilation & Creation What happens if particle-antiparticles meet +e -e Mass destroyed and turned into energy (& vice versa) All collisions must:• Conserve energy, linear momentum and charge • Conserve lepton and baryon number More of this later Example interactions e + + e + e- = electron e+ = positron - gamma ray (photon) (no charge) Mass converted into energy Example interactions In theory…(but very rarely happens) + e + + e Energy converted into mass More commonly… e + + e Photon interacts with a nearby nucleus producing mass The energy of a photon must be sufficient to create the mass of a particle & an antiparticle Erest = mc2 Provided by photon Electron produced E = hf So, if mass is to be created, photon energy must = 2mc2 (at least) 1 photon producing electron-positron pair It also works the other way around – knowing the mass of the particle-antiparticle pair that annihilate you calculate the energy of the photons produced. Theoretical requirement for electron neutrino Beta decay • decay occurs due to weak force • Random radioactive decay event due to exchange of Z0 or W± boson • Nuclei with excess n undergo - decay • Nuclei with excess p undergo + decay β Beta decay In the nucleus a neutron turns into a proton d u u u d d udd uud down quark → up quark Change in quark charge -⅓ e → ⅔ e = e+ e- must be emitted to conserve charge Beta decay 1 0 n 1 1 p+ e 0 -1 Interaction must also conserve baryon and lepton number e.g. n & p have baryon number = 1 n & p have baryon number = -1 e- has lepton number = 1 e+ ( e- ) has lepton number = -1 Beta decay 1 0 n 1 1 p+ e 0 -1 + e 0 0 anti electron neutrino Charge 0 1 + -1 Baryon 1 1 + 0 Lepton 0 0 + 1 + -1 Problem – doesn’t balance so create new particle + β Beta decay Work out the decay of a proton into a neutron 1 1 p 1 0 n+ + e + 0 1 e 0 0 electron neutrino Beta decay Another reason why neutrino’s needed to exist… Conservation of energy • Experimentally β particle energy varied – problem as decay releases fixed amount • (anti) neutrino has energy left over Pauli Exclusion Principle Within an atom, two identical particles cannot be in the same quantum state Applies only to Fermions – i.e. hadrons and leptons Explains why objects are solid, periodic table and electron shells Spin A quantum number that comes in lumps of ½ To be aware of… The closest to identical, two particles can be is to have all the same quantum numbers but opposite spin Think of an alpha particle… 2 protons, 2 neutrons Both protons/neutrons have identical quantum states but opposite spins This makes alpha particles very stable