* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Week 3 Figures ()
Fred Singer wikipedia , lookup
Citizens' Climate Lobby wikipedia , lookup
Climate sensitivity wikipedia , lookup
Scientific opinion on climate change wikipedia , lookup
Pleistocene Park wikipedia , lookup
Climate change and poverty wikipedia , lookup
Climate-friendly gardening wikipedia , lookup
Surveys of scientists' views on climate change wikipedia , lookup
Mitigation of global warming in Australia wikipedia , lookup
Snowball Earth wikipedia , lookup
Public opinion on global warming wikipedia , lookup
Effects of global warming on human health wikipedia , lookup
Iron fertilization wikipedia , lookup
Climate change, industry and society wikipedia , lookup
Hotspot Ecosystem Research and Man's Impact On European Seas wikipedia , lookup
Climate change in the Arctic wikipedia , lookup
Global Energy and Water Cycle Experiment wikipedia , lookup
Politics of global warming wikipedia , lookup
Attribution of recent climate change wikipedia , lookup
General circulation model wikipedia , lookup
Effects of global warming on Australia wikipedia , lookup
Solar radiation management wikipedia , lookup
Years of Living Dangerously wikipedia , lookup
Global warming hiatus wikipedia , lookup
Global warming wikipedia , lookup
Ocean acidification wikipedia , lookup
IPCC Fourth Assessment Report wikipedia , lookup
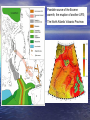
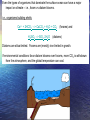
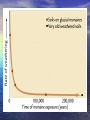
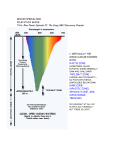
A Better Example of d18O data Compilation of paleotemperature estimates from oxygen stable isotope ratios for • surface-living planktonic foraminifera (red symbols), • thermocline-living planktonic foraminifera (blue symbols), and • benthic foraminifera (black symbols). All samples are from the North Atlantic. Norris, R.D., and P.A. Wilson, 1998, Low-latitude sea-surface temperatures for the mid-Cretaceous and the evolution of planktic foraminifera, Geol., 26, 823-826. Cooling down from the Cretaceous Greenhouse World. Prior to the boloid impact at 65 My, the Cretaceous world was already cooling. Mantle plumes were less-frequent, global temperatures were decreasing, Sea level was falling, Atmospheric CO2 levels were falling. Figure from textbook, showing global temperatures decreasing; from the end-Cretaceous estimate of about 28°C. (remember that present global temperature average is +15°C) This temperature decrease was not smooth, and there were still a few LIPS (mantle plumes) erupting. But in general, the Eocene (58 My to about 38 My) was warm compared to today. An example you have seen before. Remember: when d18O goes negative, that means that the seawater temperature is getting WARMER. The PETM – a massive release of Greenhouse Gases into the atmosphere – and the associated climate change Low-resolution marine stableisotope records of the PETM and the carbon isotope excursion, together with the seafloor sediment CaCO3 record. The carbon isotope (a) and oxygen isotope (b) records are based on benthic foraminiferal records and the from drill holes in the South Atlantic. The decrease in sedimentary CaCO3 reflects increased dissolution and indicates a severe decrease in seawater pH (that is, ocean acidification). From Zachos et al. Nature, 2008 There are two common stable isotopes of carbon: 12C and 13C. The ratio of these isotopes is expressed in relation to a standard (PeeDeeBelemnite) as d13C = [(Rsample/Rstandard) -1] x 1000 where R = (13C/12C). As d13C values increase, the abundance of the heavier isotope (13C) increases. Biological activity fractionates in favor of 12C High 12C input 12C enriched This enrichment of 12C within the biological reservoir, depletes the 12C in the exterior seawater, and the d13C ratio of the SEAWATER becomes HIGHER. PETM PETM Sea water High 12C input to biology: 12C enriched Seawater becomes depleted in 12C, Sea water d13C ratio becomes HIGH and POSITIVE High 12C output to seawater as methane (d13C = -60): Sediment 12C enriched X X Seawater becomes richer in 12C and depleted in 13C, d13C ratio is LOW and NEGATIVE. And this is the broad Eocene thermal maximum Low bioproductivity This is the PETM ‘spike’ Methane spike High bioproductivity Lots of 12C stored as ‘biology’ leaving 13C behind in the seawater The Paleocene-Eocene thermal maximum (PETM) (1) sea surface temperature rose by 5°C in the tropics; (2) by more than 7°C in the Antarctic and Arctic. (3) ocean acidification was strong (CCD was shallow). (4) with the extinction of 30 to 50% of deep-sea benthic formaminiferal species. A good ‘Rule of Thumb’ is that temperature changes in the polar regions are about TWICE those of the Global Average Temperature Change. That is => 7°C temperature increase in the Antarctic means about a 3.5°C increase in global temperatures. Or about the temperature increase expected over the next 100 years due to anthropogenic greenhouse gas emissions. The trigger for the initiation of the PETM was (probably) a period of intense flood basalt magmatism (surface and sub-surface volcanism) associated with the opening of the North Atlantic, by generating additional methane from sill intrusion into basin-filling carbon-rich sedimentary rocks. The theory. North Atlantic VP erupted in a warm climate with abundant methane hydrates stored in continental margin sediments. This increase in global temperatures destabilized the hydrates, releasing massive amounts of methane into the atmosphere. The additional warming due to the released methane/converted CO2 had a strong positive feedback, causing more hydrates to be destabilized over a 20,000 year period. The methane from the hydrates had a very negative d13C signature (-60 0/00), giving the observed correlation between global temperature spike and carbon isotope anomaly. Possible source of the Eocene warmth; the eruption of another LIPS; The North Atlantic Volcanic Province. The initiation of the PETM is marked by an abrupt decrease in the d13C proportion of marine and terrestrial sedimentary carbon, which is consistent with the rapid addition of >1200 gigatons of 13C depleted carbon, most likely in the form of methane, into the hydrosphere and atmosphere. The broad Eocene thermal maximum lasted only 210,000 to 220,000 years, with most of the decrease in d13C occurring over a 20,000year period (the PETM) at the beginning of the event. The PETM – a massive release of Greenhouse Gases into the atmosphere – and the associated climate change Low-resolution marine stableisotope records of the PETM and the carbon isotope excursion, together with the seafloor sediment CaCO3 record. The carbon isotope (a) and oxygen isotope (b) records are based on benthic foraminiferal records and the from drill holes in the South Atlantic. The decrease in sedimentary CaCO3 reflects increased dissolution and indicates a severe decrease in seawater pH (that is, ocean acidification). From Zachos et al. Nature, 2008 The initiation of the PETM is marked by an abrupt decrease in the d13C proportion of marine and terrestrial sedimentary carbon, which is consistent with the rapid addition of >1500 gigatons of 13C depleted carbon, most likely in the form of methane, into the hydrosphere and atmosphere. The Eocene Climate Optimum (warm period) lasted only 210,000 to 220,000 years, with most of the decrease in d13C occurring over a 20,000year period (the PETM). Methane is a powerful Greenhouse Gas: 24 times more effective at global warming than CO2. The Fate of methane gas in the atmosphere. It mixes quickly in the atmosphere, rises to the stratosphere where it is rapidly oxidized to CO2 – with an 8 to 10 years residence time CH4+O2= CO2+H20 And becomes another powerful greenhouse gas, CO2, with a much longer residence time. This is the window of long-wave radiation that keeps the earth habitable. Acid Oceans? During this massive methane release, the oxidation and ocean absorption of this carbon would have lowered deep-sea pH (increased ocean acidity dramatically). This low ocean pH would have led to rapid shoaling of the calcite compensation depth (CCD), followed by a gradual recovery. Evidence of a rapid acidification of the deep oceans would be evident in the abrupt transition from carbonate-rich sediment to clay, followed by a gradual recovery to carbonate. Samples of the ocean sediment from five South Atlantic deepsea sites, all within the geologic time frame of the PETM. During the Eocene, the CCD is inferred to have shoaled more than 2 km within a few thousand years. Graphs of the core samples show an abrupt transition from carbonate-rich sediment to clay, followed by a gradual recovery (100K years) to carbonate. Carbon dioxide levels and Cenozoic climate change. [note these are low resolution records] The pCO2 record is converted to radiative forcing (a measure of global warming). The benthic foraminifer d18O time series is a smoothed record of many observations. The trend towards more positive d18O results from a combination of deep-sea cooling and global ice volume increases. from Pearson and Palmer, Science, v406, 2000. Estimates of Cenozoic atmospheric pCO2 based on two independent proxies as measured in sub-tropical deep-sea sediment cores from the Pacific. The first curve is estimated from surface ocean pH as derived from the boron isotope ratios of planktonic foraminifers. I.e. 11B/10B isotope ratio depends on seawater pH. The second pCO2 curve is based on the d13C values of phytoplankton organic compounds known as alkenones. Both approaches assume chemical equilibrium between the ocean and atmosphere. Sea surface pH for the past 60 Myr. These results are inferred from calculated pCO2 data – obtained from boron isotope ratios (from foram shells). Vertical error bars result from analytical error in determining d11B ratio. During the Eocene, the oceans were TOO ACIDIC for corals with carbonate shells. Pearson and Palmer, Nature 406, 2000 SEQUENCE OF EVENTS AT PETM (Eocene climate maximum). 1. Hypothesis – initial cause? The eruption of the North Atlantic Igneous Province (the head of the Iceland mantle plume). 60 to 55 My. 2. Climate gets (generally) warm. 3. Ocean circulation changes – conveys surface warmth to deep ocean. Bottom water starts warming up. 4. Methane hydrates stored in the sediments now become unstable. Some continental margin slopes are de-stabilized, and slump, exposing more hydrates – which decompose to methane. Strong positive feed-back! 5. Large (1,500 gigatons) amounts of organic carbon are vented into the ocean and atmosphere. 6. The methane in the atmosphere and the oxidation of CH4 to CO2 (both greenhouse gases), cause a strong temperature spike (the PETM). 7. This increased global temperature causes more water evaporation, more coastal run-off, more nutrients into the ocean. 8. This increases biological productivity which removes the CO2 from the atmosphere (the biological pump), and temperatures ‘cool off’. 9. Heat spike (few 1000 years). Cooling off period – 70K to 100K years. Can it happen again? Is the PETM a good analog for future climate change? There are presently large quantities of methane stored as hydrates in the arctic permafrost and in the shallow continental margins. In the continental margin sediments, the low thermal conductivity of the sediments slows the transfer of a warming ocean to the hydrates (PETM took 20K years). But in the arctic, the melting permafrost exposes the hydrates to the atmosphere (or in shallow ponds and lakes) and the hydrates can be dissociated quickly. And they are. Using Eocene data to simulate future climate (Zachos et al, Nature, 2008) (a), ocean surface pH (b), ocean surface calcite saturation (c) and deep-ocean temperature changes (d) in response to the input of 5,000 Gt C of anthropogenic CO2 into the atmosphere, starting from preindustrial CO2 levels. Blue and green are w/wo a silicateweathering feedback. Projected changes in deep-ocean temperature in d assume a homogeneous warming following temperature sensitivities to a doubling of CO2 concentration: short-dashed line, 4.5 °C; solid line, 3.0 °C; long-dashed line, 1.5 °C. Present day carbon reservoirs More than half the present day (non-carbonate) carbon is locked up in Gas hydrates. What is methane hydrate – and how does it form? Methane hydrate is methane gas (CH4) ‘locked’ into a water-ice lattice. Places where you find large methane hydrate deposits. Shallow continental margins. (why?) Methane forms from the organic matter that rains down from the surface of the ocean – and is preserved in the sediments at the seafloor. Once this organic matter is buried, it does not oxidize. When the sediments are heated (by being buried) to 100°C, the organic matter is converted to methane gas, oil, and other hydrocarbons. During subduction on continental margins, the sediments are compressed and the methane escapes. Methane Hydrate CH4 + H2O = (at low temperature and high pressure) methane hydrate Looks like ice, but is unstable at atmospheric pressure and room temperature. Also called ‘clathrate’. Phase diagram showing the water depths (and pressures) and temperatures for gas hydrate (grey area) stability. Many sediments lie within the range denoted by the box. The line shows the temperature in the Earth as a function of depth (geotherm). At greater depths in the sediments, the geotherm crosses from the hydrate zone (purple region) to the gas zone. This means that gas hydrate in sediments usually overlies free gas. Because hydrate is a solid, it forms at the cold surface of the sediments, and traps the methane gas below (in the warmer sediments). But as seawater warms, the solid lid decomposes, and the methane escapes as bubbles. Which can reach the surface as methane gas. An acoustic profile over a methane vent, using a 200 kHz sonar. Taken from the Washington continental shelf, off Grays Harbor, last September. Rising methane bubble plume Probably fish Layers of high krill concentrations RED layer is seafloor Courtesy Julie Keister, UW/Oceanography Release of Methane hydrate Under stable ocean temperatures, methane hydrate can be released through slumping and permafrost warming due to rising arctic waters. Another way to release methane hydrate is through warming ocean temperatures. NOTE: While this figure is required by law to be shown in all paleoclimate courses, do not try this at home. It is not dangerous, just extremely disappointing… Notice that there are major climate shifts in the Cenozoic climate record that are not reflected solely in the atmospheric CO2 concentration. Other global disasters during the (troubled) Eocene. The Chesapeake Bay impact crater was formed by a bolide that impacted the eastern shore of North America about 35.5 million years ago, in the late Eocene. It is the second largest impact crater in the U.S. Continued slumping of sediments over the rubble of the crater have helped shape Chesapeake Bay. During the Eocene climate Maximum, there were no ice sheets, and the value of d18O for seawater was probably near zero (surface value today). The changes in d18O in deep seawater are shown on the curve on the right. Clearly it has been getting cooler (more positive d18O ratio) since the Eocene. But since 30 My ago, the d18O from the carbonate shells contains both ice volume and temperature information. Because substantial amounts of 16O is stored in ice sheets, we need to estimate HOW MUCH ice is present to use this for paleotemperatures. Benthic, deep-water forams Another possible mechanism for ‘cooling the earth’ - by removing CO2 from the atmosphere. The raising of continental plateaus. How would this decrease CO2 levels? Increased weathering of the rocks/mountains that are uplifted. Other geological processes related to seafloor spreading that can cause major climate change. The formation of high plateaus – and the increase in weathering (and the draw-down of atmospheric CO2 that results). Classic example – the Tibetan Plateau. The flux of sediments from the Tibetan plateau has increased dramatically over the last 40 My (by factor of 10). This both pulls CO2 directly from the atmosphere (as carbonic acid) but also fertilizes the upper ocean and increases the ‘biological pump’. This ‘fertilization’ is largely iron (which is a limiting nutrient for both forams and diatoms). But it can also provide dissolved silica, which promotes diatom blooms. And diatoms are more effective at pulling CO2 out of the atmosphere than forams. Some climate models have predicted that this difference (diatom blooms) is the amplifier of the orbital cycles that produces the glacial/interglacial cycles. Even the types of organisms that dominate the surface ocean can have a major impact on climate – i.e., foram vs diatom blooms. i.e., organisms building shells Ca++ + 2HCO3- --> CaCO3 + H2O + CO2 H4SiO4 --> SiO2·2H2O (forams) and (diatoms) Diatoms are silica-limited. Forams are (mostly) iron limited in growth. If environmental conditions favor diatom blooms over forams, more CO2 is withdrawn from the atmosphere, and the global temperature can cool. The uplifted Tibetan plateau also creates it’s ‘own weather’ – the Monsoons. The summer sun on the plateau causes an upwelling zone over the plateau, which draws (warm, very moist) air from the continental margins. This causes very heavy seasonal rains, which are effective in weathering of the rocks and flushing nutrients into the surrounding ocean. Possible problem with this hypothesis: negative feed-backs on weathering as a control on global climate. Increasing weathering in high plateaus can cause a DECREASE in weathering in areas that are NOT high plateaus. Other processes that control climate, on a shorter timescale – ‘Gateways’ (restrictive passages between continents) can open and close, and force changes in ocean circulation. Ocean circulation is the ‘pump’ that moves heat from the equator to the higher latitudes, and distributes heat over the earth. Opening of Drake passage at 20 My coincided with a new ice sheet forming at Antarctica Closing of the Isthmus of Panama between 10 and 4 My coincided with the formation of polar (and then midlatitude) ice sheets. Weathering can produce both positive and negative feedbacks. By producing glaciers that grind-up the rocks, and exposing more rock surface to weathering, the resulting drawdown in CO2 can make things cooler. A positive feedback. An example you have seen before. Remember: when d18O goes negative, that means that the seawater temperature is getting WARMER.