* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Marvelous Metabolism
Survey
Document related concepts
Enzyme inhibitor wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Biochemistry wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Transcript
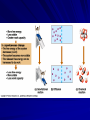
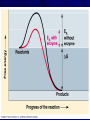
Marvelous Metabolism Chapter 8 I. Vivacious Vocabulary Metabolism - total of all an organisms chemical processes (all the reactions happening in an organism) Catabolic pathways - release energy by breaking down complex molecules into simpler ones ex. - respiration Anabolic pathways - consume energy to build complex molecules from simpler ones ex. - building proteins from amino acids Bioenergetics - study of how organisms II. Energy Capacity to do work A. Kinetic energy Energy of motion Ex. – Water through dam – Light powering photosynthesis – Heat is kinetic energy associated with RANDOM movement of atoms. B. Potential energy Stored energy due to location or structure Ex. – Water behind dam (before it’s released) – Chemical energy - stored in molecules due to arrangement of atoms Organisms are energy transformers!! III. Thermodynamics Study of energy transformations III. Thermodynamics A. First Law of Thermodynamics - energy can not be created or destroyed, but it can be transferred B. Second Law of Thermodynamics Every energy transfer increases the entropy (disorder/ randomness) of the universe – 1. In most energy transformations, part of the energy is lost as heat (heat is energy in it’s most random state) – 2. Entropy of a system can decrease, as long as the entropy of the universe (system+ surroundings) increases IV. Free Energy Portion of a system’s energy that can perform work when temperature is uniform throughout the system DG = DH - T DS DG = change in free energy DH = change in system total energy T = absolute temp (in Kelvin oC + 273) DS = change in entropy IV. Free Energy In any spontaneous process, the free energy of a system decreases (DG must be negative) For a reaction at equilibrium, DG = 0 A. Exergonic reaction Energy Out - occur with a release of free energy -DG = spontaneous reaction ex. - Cellular respiration C6H12O6 + 6O2 ---->6CO2 + 6H2O DG = -686 kcal/mole B. Endergonic reaction Energy In - absorbs free energy from it’s surroundings + DG = nonspontaneous reaction ex - Photosynthesis DG = +686 kcal/mole This reaction is powered by light energy from the sun C. Metabolic Disequilibrium If a cell were allowed to reach chemical equilibrium, DG = 0, the cell would be dead. To keep this from happening, the product of one reaction becomes a reactant for another reaction. This keeps products from building up and prevents the cell from reaching equilibrium. V. Awesome ATP A. Types of Work - cells do three types of work 1. Mechanical - muscle contraction, movement of chromosomes 2. Transport - pumping across membranes 3. Chemical - pushing endergonic reactions that would not occur spontaneously V. Awesome ATP B. ATP - source of energy that powers cellular work Adenosine triphosphate (adenine + ribose + 3 phosphate) 1. Bonds between phosphate groups can be broken by hydrolysis. This releases energy DG = -7.3 kcal/mol ATP + H2O ----> ADP + Pi V. Awesome ATP 2. When ATP is hydrolyzed (broken down), it can be used to drive endergonic reactions by transferring the Pi (phosphate) group to some other molecule. This is called Phosphorylation. V. Awesome ATP 3. ATP can also be regenerated by adding a Pi to ADP. This is an endergonic reaction. DG = +7.3 kcal/mol This reaction happens continuously in a cell and is driven by breaking down glucose in respiration. < < 8/33: 06-08-ATPStructure-L <> Exciting and Energetic Enzymes!!! I. What are Enzymes? Enzymes are protein catalysts!! Catalysts speed up the rate of a reaction without being consumed! II. Remember Free Energy? Activation Energy EA – Free Energy needed to start a reaction – Required to break bonds of reactants – Heat works! It speeds up the molecules, which collide more and stronger III. How do Enzymes Work? Heat doesn’t work for cells because it denatures proteins...so...Go Enzymes! Enzymes lower EA barrier so transition state can be reached at room temperature Only can speed up reactions that would occur anyway Enzymes work on a specific substrate or reactant III. How do Enzymes Work? Enzyme Substrate ---------------- > Product(s) Sucrase Sucrose + H20 ------------- > Glucose + Fructose Enzymes are so specific, can distinguish between isomers Enzymes usually end in -ase Figure 6.11 Example of an enzyme-catalyzed reaction: Hydrolysis of sucrose IV. The Detail and the Visual Enzymes have active sites where the substrate fits! When substrate enters the active site, it induces the enzyme to change shape slightly so it’s nice and snug. This is an induced fit. (Big H-bond Hug!) About 1000 substrate molecules/second or more Figure 6.14 The induced fit between an enzyme and its substrate V. How do enzymes do these cool things? Can stress or twist bonds to induce a break! (less heat will be required) Active site may give a micro environment – Lower pH – Brief covalent bonds with side chains of the enzyme The more substrate, the faster the reaction occurs until the saturation point where enough to fill each enzyme (vmax) Sometimes Cofactors (coenzymes) required -non-protein helpers, vitamins, minerals VI. Environment can change A LOT! Enzymes have an optimal temperature and pH Temperature increases the speed of reactions with enzymes....the molecules move faster Too high a temp and the protein enzymes denature and don’t work VII. Evil Enzyme Inhibitors Competitive Inhibitors: – Mimic the normal substrate and compete for admission into active site. Non-competitive Inhibitor – Bind to enzyme somewhere other than active site, change shape of enzyme so substrate doesn’t fit. Bad inhibitors include...DDT (pesticides) and Antibiotics (inhibit bacteria) VIII. Regulators Enzymes often have regulator molecules! These bond to an allosteric site (away from active site) Enzyme has two shapes...1 active and 1 inactive Activators bind to allosteric site and stabilize the active shape Inhibitors bind to allosteric site and causes enzymes inactive shape VIII. Regulators Feedback Inhibition – Metabolic path is shut off because the end-product acts as an inhibitor by allosterically binding – This prevents wasting resources when there is enough product IX. Order! Order! It’s not just random soup out there in cell land! Enzyme teams hang out together in a multienzyme complex Some enzymes are in fixed locations in the cell as fixtures in membranes Some are in solution inside organelles – Respiration enzymes are in Mighty Mitochondria Emergent Properties Rule!!