* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download ch3a FA11 - Cal State LA
Microbial metabolism wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Point mutation wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Metalloprotein wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Catalytic triad wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Biochemistry wikipedia , lookup
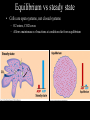
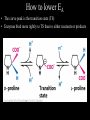
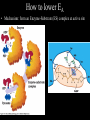
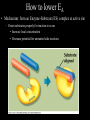
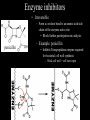
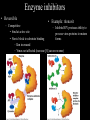
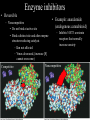
Bioenergetics • Study of energy transformations in living organisms • Thermodynamics – 1st Law: Conservation of E • Neither created nor destroyed • Can be transduced into different forms – 2nd Law: Events proceed from higher to lower E states • Entropy (disorder) always increases – Universe = system + surrounds Bioenergetics (E content of system) H = (useful free E) G + (E lost to disorder) TS • Gibbs Free Energy: G = H - TS – If G = negative, then rxn is exergonic, spontaneous – If G = positive, then rxn is endergonic, not spontaneous – Standard conditions (ΔG°’): 25oC, 1M each component, pH 7, H2O at 55.6M Bioenergetics A + B <--> C + D • Rate of reaction is directly proportional to concentration of reactants • At equilibrium, forward reaction = backward reaction k1[A][B] = k2[C][D] • Rearrange: k1/k2 = ([C][D])/([A][B]) = Keq • Relationship between ΔG°’ and K’eq is: G°’ = -2.303 * R * T * log K’eq If Keq >1, G°’ is negative, rxn will go forward If Keq <1, G°’ is positive, rxn will go backward • ΔG°’ is a fixed value at standard conditions • ΔG under actual cellular conditions can be different – e.g., for ATP hydrolysis inside a cell, can approach ΔG = -12 kcal/mol • We will work with ΔG°’ values Coupling endergonic and exergonic rxns Glutamic acid (Glu) + NH3 --> Glutamine (Gln) G°’=+3.4 kcal/mol ATP --> ADP + Pi G°’=-7.3 kcal/mol ---------------------------------------------------------------------------------------Glu + ATP + NH3 --> Gln + ADP + Pi G°’=-3.9 kcal/mol Glutamyl phosphate is the common intermediate ATP --> ADP + Pi ΔG°’= -7.3 kcal/mol ADP + Pi --> ATP ΔG°’= +7.3 kcal/mol C(diamond) + O2 --> CO2 ΔG°’= -94.8 kcal/mol PEP --> pyruvate + Pi ΔG°’= -14.8 kcal/mol C(graphite) + O2 --> CO2 ΔG°’= -94.1 kcal/mol P-creatine --> creatine + Pi ΔG°’= -11.0 kcal/mol G6-P --> glucose + Pi ΔG°’= -3.0 kcal/mol 1,3-BPG --> 3PG + Pi ΔG°’= -12.5 kcal/mol ---------------------------------------------------------------------------------------? What is ΔG°’ of: PEP + ADP --> pyruvate + ATP ΔG°’= -7.5 ---------------------------------------------------------------------------------------What is ΔG°’ of: G6-P + ADP --> glucose + ATP What is ΔG°’ of: P-creatine + ADP --> creatine + ATP What is ΔG°’ of: C(s, diamond) --> C(s, graphite) Equilibrium vs steady state • Cells are open systems, not closed systems – O2 enters, CO2 leaves – Allows maintenance of reactions at conditions far from equilibrium O2 Biological Catalysts 1) Req’d in small amounts 2) Not altered/consumed in rxn 3) No effect on thermodynamics of rxn a) Do not supply E b) Do not determine [product]/[reactant] ratio (Keq) c) Do accelerate rate of reaction (kinetics) 4) Highly specific for substrate/reactant 5) Very few side reactions (i.e. very “clean”) 6) Subject to regulation No relationship between G and rate of a reaction (kinetics) Why might a favorable rxn *not* occur rapidly? Overcoming the activation energy barrier (EA) • Bunsen burner: CH4 + 2O2 --> CO2 + 2H2O – The spark adds enough E to exceed EA (not a catalyst) • Metabolism ‘burning’ glucose – Enzyme lowers EA so that ambient fluctuations in E are sufficient Overcoming the activation energy barrier (EA) • Bunsen burner: CH4 + 2O2 --> CO2 + 2H2O – The spark adds enough E to exceed EA • Metabolism ‘burning’ glucose – Enzyme lowers EA so that ambient fluctuations in E are sufficient Catalyst shifts EA line to left <--- How to lower EA • The curve peak is the transition state (TS) • Enzymes bind more tightly to TS than to either reactants or products How to lower EA • Mechanism: form an Enzyme-Substrate (ES) complex at active site How to lower EA • Mechanism: form an Enzyme-Substrate (ES) complex at active site – Orient substrates properly for reaction to occur • Increase local concentration • Decrease potential for unwanted side reactions How to lower EA • Mechanism: form an Enzyme-Substrate (ES) complex at active site – Enhance substrate reactivity • Enhance polarity of bonds via interaction with amino acid functional groups • Possibly form covalent bonded intermediates with amino acid side chains How to lower EA • Possibly form covalent bonded intermediates with amino acid side chains – Serine protease mechanism: How to lower EA • Possibly form covalent bonded intermediates with amino acid side chains – Serine protease mechanism: How to lower EA • Mechanism: form an Enzyme-Substrate (ES) complex at active site – Induce bond strain • Alter bonding angles within substrate upon binding • Alter positions of atoms in enzyme too: Induced fit Induced fit Induced fit Enzyme kinetics: The Michaelis-Menten Equation S <--> P At low [S], rate/velocity is slow, idle time on the enzyme At very high [S], rate/velocity is maximum (Vmax), enzyme is saturated V = Vmax [S]/([S] + Km) Km = [S] at Vmax/2 A low Km indicates high enzyme affinity for S (0.1mM is typical) Enzyme kinetics: pH and temperature dependence Enzyme inhibitors • Irreversible – Form a covalent bond to an amino acid side chain of the enzyme active site • Block further participation in catalysis penicillin – Example: penicillin • Inhibits Transpeptidase enzyme required for bacterial cell wall synthesis – Weak cell wall = cell burst open Enzyme inhibitors • Reversible • Example: ritonavir – Competitive – Inhibits HIV protease ability to • bind at active site process virus proteins to mature • Steric block to substrate binding forms – Km increased – Vmax not affected (increase [S] can overcome) Enzyme inhibitors • Reversible – Noncompetitive • Do not bind at active site • Bind a distinct site and alter enzyme structure reducing catalysis – Km not affected – Vmax decreased, (increase [S] cannot overcome) Competitive • Example: anandamide (endogenous cannabinoid) – Inhibits 5-HT3 serotonin receptors that normally increase anxiety Noncompetitive Drug discovery • Average cost to market ~ $1B • Average time to market ~13 years • Size of market ~ $289B per year in US (2006) • S. aureus infections are a problem in hospital settings – Drug targets • Metabolic rxns specific to bacteria – Sulfa drugs (folic acid biosynthesis) • Cell wall synthesis – Penicillin, methicillin, vancomycin • DNA replication, transcription, translation – Ciprofloxacin (DNA gyrase) – Tetracyclins (ribosome) – Zyvox (ribosome) » Introduced in 2000, resistance observed within 1 year of use