* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Zhang Yufeng - USD Biology
Evolution of metal ions in biological systems wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Citric acid cycle wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biochemistry wikipedia , lookup
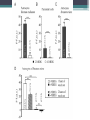
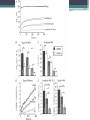
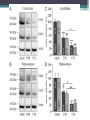
Energy Metabolism in Astrocytes and Neurons Yufeng Zhang Fuel selection • The energy requirements of the brain are very high • Lipids contain more energy compare to other substrate • Other organs use lipids as fuel • Fatty acid metabolism has a role in neurodevelopment, neurotransmission, and repair processes Reasons for low FFA oxidation • (i) a slow passage of fatty acids across the blood–brain barrier (BBB) • (ii) a low enzymatic capacity for the fatty acid degradation • (iii) side effects of long-chain fatty acids in the mitochondrial ATP synthesis BBB Permeability • In situ perfusion technique demonstrating that long-chain NEFA are rapidly transported through the BBB to a large portion and with similar rates ▫ passive transport or alternatively ▫ protein-mediated diffusion FFA in mitochondria • Eightfold lower than that of heart mitochondria under comparable conditions • High oxygen consumption with pyruvate or glutamate as hydrogen donors, as compared with heart and skeletal muscle mitochondria • Low-level oxidation of long-chain fatty attributed to ▫ low translocation rate ▫ low enzymatic capacity of the β-oxidation pathway Harmful side effect Other reasons • Oxidation of Fatty Acids Increases the Tendency of Neural Tissue to Become Hypoxic • Increases the Risk of enhanced Oxidative Stress • Fatty Acid Oxidation is too Slow for Matching the ATP Requirements Astrocyte-Neuron Metabolic Cooperation • Blood delivery increases with metabolic demand, • CBF and CMRglc increase more than oxygen utilization, and • Both oxidative and nonoxidative processes are involved to meet the increased metabolic requirements Cytoarchitectural organization of brain • Astrocytes have emerged as active players in brain energy delivery, production, utilization, and storage • Astrocytes possess unique cytoarchitectural and phenotypic features that ideally position them to sense their surroundings and dynamically respond to changes in their microenvironment Regulation of Cerebral Blood Flow • Astrocytes play a key role in neurovascular coupling ▫ Coupling between neuronal activity and increased CBF (AKA hyperemia) ▫ Essential contributors to both vasoconstriction and vasodilation. 1) Increased astrocytic Ca++ concentrations—activation of glycolysis--elevation of extracellular lactate --- dilation. 2) More oxygen ---astrocytic Ca2+ signals would induce a constricting tone ---keeping CBF at an appropriate lower level Metabolic Specialization of Neurons and Glia • Brain dependent upon the uninterrupted supply of energy substrates from the circulation. • In addition to glucose, including lactate, pyruvate, glutamate, and glutamine can also be energy substrate • Lactate used to be considered a metabolic dead end Metabolic Profile • Neurons sustain a high rate of oxidative metabolism compared to glial cells. • 6-phosphofructose-2-kinase/fructose-2,6bisphosphatase-3 (Pfkfb3) is virtually absent in neurons ▫ a potent activator of the glycolytic enzyme phosphofructokinase-1 (PFK) • The use of lactate – high ATP, thereby sparing glucose for the PPP. • Astrocytes take up glucose and characteristically present a high glycolytic rate, end with lactate Neurotransmitter Recycling and Anaplerosis • Astrocytes can rapidly remove neurotransmitters released into the synaptic cleft ▫ Glutamate: overstimulation of glutamate receptors is highly toxic to neurons • Astrocytes also play an important role in transferring this neurotransmitter back to neurons Astrocyte-Neuron Lactate Shuttle • Amount of glucose that astrocytes take up is higher to their energy requirements • (1) neuronal activity increases extracellular glutamate which is avidly taken up via a Na+dependent mechanism • (2) increase in Na+ activates the Na+/K+ ATPase thereby increasing ATP consumption • (3) leads to a large increase in the production of lactate in the extracellular space • (4) lactate can be used as an energy substrate for neurons Astrocytic Glycogen Metabolism • Glycogen is the largest energy reserve of the brain • Glycogen has been found to be almost exclusively localized in astrocytes Prior to inhibitory avoidance training, rats received intrahippocampal injections of (DAB, a potent inhibitor of glycogen phosphorylase), lactate, or antisense oligodeoxynucleotides, as indicated. Short-term memory (STM) was assessed 1 hr later (experiment 1), while long-term memory (LTM) formation was assessed after 1 and 7 days (experiments 2–7). Astrocytic glycogen is more than a simple emergency reserve, and plays an important and active role in complex brain physiological functions, in particular through an astrocyteto-neuron transfer of energy metabolites in the form of lactate. Defense against Oxidative Stress • The brain is particularly susceptible to oxidative damage ▫ high rate of oxidative energy metabolism ▫ high unsaturated fatty acids content ▫ Relatively low intrinsic antioxidant capacity • Astrocytes have higher levels of various antioxidant molecules • Neurons are dependent upon the high antioxidant potential of astrocytes GSH—the most abundant antioxidant molecule in the brain neurons are highly dependent on astrocytes for the supply of the precursor amino acids necessary for their own GSH synthesis Metabolic Plasticity in Astrocytes • Astrocytes have a greater metabolic plasticity than neurons • Any significant alteration of astrocytic pathways caused by pathological stimuli could potentially contribute to neuronal dysfunction • Enhancing astrocytic functions may represent therapeutic avenues for neurodegenerative disease Neuronal Excitability • Glucose-sensing neurons increase their firing rate in response to elevation of central glucose levels, ultimately resulting in decreased blood glucose and insulin levels and the suppression of hepatic gluconeogenesis • Orexin neurons also function as lactate sensors • Salt-intake behavior is also influenced by extracellular astrocyte-derived lactate What extent neurons are fed directly by glucose or by lactate? • Method: ▫ ▫ ▫ ▫ Mixed culture of neuronal and glial cells Probes 2-NBDG and 6-NBDG Optical measurements Protein extraction and Immunoblot Conclusion • Neural cells don’t use Fatty Acid as fuel, which helps to protect neural cells against enhanced oxidative stress • Neuroenergetics has evolved from a neurocentric view into a more integrated one in which complementarities and cooperativities between astrocytes and neurons. ▫ lactate shuttling from astrocytes to neurons Thank you