* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Gel electrophoresis of nucleic acids wikipedia , lookup
DNA sequencing wikipedia , lookup
SNP genotyping wikipedia , lookup
Genomic library wikipedia , lookup
Agarose gel electrophoresis wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Whole genome sequencing wikipedia , lookup
Gel electrophoresis wikipedia , lookup
Exome sequencing wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
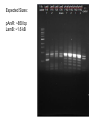
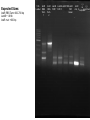
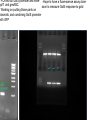
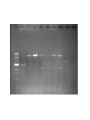
iGEM 8.3.10 - 8.6.10 Arsenic Bioremediation Possibly finished biobrick for ArsR by adding a RBS and terminator. Will send for sequencing today or Monday. Do we only send them the forward primer? Tried ArsB gradient with increased DMSO concentration, still no luck. Placed order for redesigned primers. We have all the genes we need on plasmids except for ArsB and possibly LamB, we're troubleshooting our verification PCR for contamination. Received questionable sequencing results from ArsR site directed mutagenesis. Most likely resulted from impure sample. Expected Sizes: pArsR: ~850 bp LamB: ~1.6 kB Expected Sizes: ArsR RBS Term: 600-700 bp LamB:~1.6 Kb ArsR mut: ~600 bp 1 Kb Ladder ArsR RBS Term. 1 ArsR LamB LamB LamB F2RLamB RBS F2R 1' F2R 2' F2R Term. Green 2 ArsR mut. 2 (-) (-) no no primers template Site Directed Mutagenesis: ArsR ArsR Coding Region: ATG TCA TTT CTG TTA CCC ATC CAA TTG TTC AAA ATT CTT GCT GAT GAA ACC CGT CTG GGC ATC GTT TTA CTG CTC AGC GAA CTG GGA GAG TTA TGC GTC TGC GAT CTC TCA CTG CTC TCG ACC AGT CGC AGC CCA AGA TCT CCC GCC ACC TGG CAT TGC TGC GTG AAA GCG GGC TAT TGC TGG ACC GCA AGC AAG GTA AGT GGG TTC ATT ACC GCT TAT CAC CGC ATA TTC CAG CAT GGG CGG CGA AAA TTA TTG ATG AGG CCT GGC GAT GTG AAC AGG AAA AGG TTC AGG CGA TTG TCC GCA ACC TGG CTC GAC AAA ACT GTT CC GGG GAC AGT AAG AAC ATT TGC AGT TAA The goal was to change the Xba cut site AGATCT to AGGTCT. This change coded for the same amino acid with a similar frequency to the AGA codon. Sequencing Results: ArsR Site Directed Mutagenesis Sample 2 Expected Sequence: tgccacctgacgtctaagaaaccattattatcatgacattaacctataaaaataggcgtatcacgaggcagaatttcagataa aaaaaatccttagctttcgctaaggatgatttctggaattcgcggccgcttctagagATGTCATTTCTGTTACCCATCCAATT GTTCAAAATTCTTGCTGATGAAACCCGTCTGGGCATCGTTTTACTGCTCAGCGAACTGGGAGAGTTATGC GTCTGCGATCTCT GCACTGCTCTCGACCAGTCGCAGCCCAAGGTCTCCCGCCACCTGGCATTGCTGCGTGAAAGCGGGCTA TTGCTGGACCGCAAG CAAGGTAAGTGGGTTCATTACCGCTTATCACCGCATATTCCAGCATGGGCGGCGAAAATTATTGATGAG GCCTGGCGATGTGA ACAGGAAAAGGTTCAGGCGATTGTCCGCAACCTGGCTCGACAAAACTGTTCCGGGGACAGTAAGAACAT TTGCAGTTAAtact agtagcggccgctgcaggcttcctcgctcactgactcgctgcgctcggtcgttcggctgcggcgagcggtatcagctcactca ArsR Site Directed Mutagenesis 2: aaggcggtaat CCTTGCGTCCGTCGAGACGATCGGAGCTAGTCGAGGAGCGAGTTTGCCAGCAACGGATGCTTCTCTT ATTAACTGGAATGT TTCTTAGAGTTCCCGGCCCCTTCTTGATGACATTTCTGGCAGACATCCGCCTGTTCCAAATTCCTGTTC AATTAACCCCGC CTGGTCATCAATTTTACTGCTCCCATGAACTGAATATAGTTATGATATCTGCGTATCTCTGCCATGTTAC TTTGCCATGTC GTCCACCAAGGTCTCCCGTTTCACTGGCATTGGTGCGGGGAAGCGGGACCTTGCTGGTGCCGACTG GTCGGTAGCTGTGTT CATTACCGCTTATCATAACTCATCCCATTCTGTGAGGCGTAAAATTAATGCCCAAAGCCTGTTTCATGT GAACGAAATATG GATCATGGGATGGTCCACAAACTGGATCTCCAAAACTGTTCCCGGAATCCAAAAATCATTTGTAGTTAA TGCTAATGATTT TTTCTATCTGATTCTCCGCTCCCTGACTCGCCTATTTTTATAGGTTAATGTGCATGATAGTAATGGTTTC TCACTACGTCG CGTGGCTCTTTTTATCCACATGTTCGCGGAATCACTATTGGTATATTTTTCTAACTACATGCCAATATGT AGCCGCTAATG AGACAATAACCCTGATAAATGCTTTTTAATATTGCTCCGCCCCCCAGACTAACTTCTCAACATTCCACT CTCACCCTTAAT CCCTTTAATCCCGGCATTGACTATTCCTGACCATGCTCACCCCCCTGCACTCGCCATCGTGCGCTATG CTGATCATACCCT Gold - Re-ligated golS onto plasmid since sequence homology last time was poor (79% homology) - Possibly have golB on a plasmid, but still need to do miniprep and digestion to verify (there were no bands from the first colony) Gel extraction: gesABC ges abc - PCR'ed out GolS promoter and more - Hope to have a fluorescence assay done golT and gesABC soon to measure GolS response to gold - Working on putting those parts on plasmids, and combining GolS promoter with GFP PCR from genome of ges works, but can't amplify ges or golT fragments from gel extraction - Haven't been able to PCR gesABC gel extraction template (lanes have a "smear" down them) RNA Decoder We have continued on our digestion/ligation/transformations for the project. We still need to PCR out EGFP and MicF, but otherwise we have all the parts we need. This morning we checked on transformants done yesterday and only had 2 of 5 plates growing colonies. When we ran a gel for our digestions of each part, every miniprep has some sequence that is ~6 kB in it. What could that be? Could it be affecting our transformations? Other Updates Dip'n Dots Fundraiser went well! Who should we plan on taking to the Jamboree - please fill out an intent to travel form. GAMES camp went well Next Friday will be our last meeting of the summer. During the semester, we'd like to have meetings one every 2-3 weeks. What days and times work well or don't work? We received donations from Midsci and S.J. Smith, and the tools team has been in contact with Tetravitae about potential sponsorship Sequencing Results: ArsR Site Directed Mutagenesis Sample 1