* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Alcohols
Discodermolide wikipedia , lookup
Asymmetric induction wikipedia , lookup
George S. Hammond wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Kinetic resolution wikipedia , lookup
Ene reaction wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Elias James Corey wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
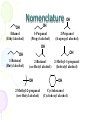
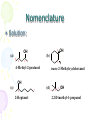
Chapter 14 Alcohols, Ethers, and Thiols Alcohols • Alcohol: an -OH (hydroxyl) group bonded to a tetrahedral carbon • Nomenclature 1.longest chain with the -OH group numbered lowest possible (rings OH=1) 2.change the ending of the parent alkane from e to -ol 3.name and number substituents and list them in alphabetical order Nomenclature OH Ethan ol (Ethyl alcohol) OH OH 1-Propan ol (Prop yl alcohol) 2-Prop anol (Is op ropyl alcohol) OH OH 1-Butan ol (Bu tyl alcohol) OH 2-Bu tanol (sec-Bu tyl alcohol) OH 2-Methyl-2-p ropan ol (t ert -Butyl alcoh ol) 2-Meth yl-1-propanol (Is ob utyl alcoh ol) OH Cycloh exanol (Cycloh exyl alcoh ol) Nomenclature • Problem: write the IUPAC name for each alcohol OH (a) OH (b) OH (c) (d) OH Nomenclature • Solution: (a) 5 4 OH 1 OH (b ) 2 2 4-Methyl-2-pentan ol t rans-2-Meth ylcycloh exanol OH (c) 2 2-Heptanol 7 (d) 3 2 1 OH 2,2-D imeth yl-1-p ropanol Nomenclature – in the IUPAC system, a compound containing two hydroxyl groups is named as a diol, one containing three hydroxyl groups as a triol, and so on – IUPAC names for diols, triols, and so on retain the final "-e" in the name of the parent alkane – we commonly refer to compounds containing two hydroxyl groups on adjacent carbons as glycols CH2 CH2 OH OH CH3 CHCH2 HO OH 1,2-Eth anediol (Eth ylen e glycol) 1,2-Propan ediol (Propylene glycol) CH2 CHCH2 OH OHOH 1,2,3-Propan etriol (Glycerol, Glycerin) Physical Properties • Alcohols are polar molecules – the C-O and O-H bonds are both polar covalent Physical Properties – in the liquid state, alcohols associate by hydrogen bonding Physical Properties – bp increases as MW increases – solubility in water decreases as MW increases Molecular Weigh t bp (°C) Solubility in Water Structu ral Formula N ame CH3 OH CH3 CH3 methanol ethan e 32 30 65 -89 infinite ins olub le CH3 CH2 OH ethan ol propane 46 infinite 44 78 -42 1-propanol bu tane 60 58 97 0 1-bu tanol pen tane 74 117 72 36 CH3 CH2 CH3 CH3 CH2 CH2 OH CH3 CH2 CH2 CH3 CH3 CH2 CH2 CH2 OH CH3 CH2 CH2 CH2 CH3 ins olub le infinite ins olub le 8 g/100 g ins olub le Acidity of Alcohols • Alcohols have about the same pKa values as water; aqueous solutions of alcohols have the same pH as that of pure water • phenols contain an OH group that is more acidic – phenols are weak acids and react with NaOH and other strong bases to form water-soluble salts OH + NaOH Phenol H2 O O- Na+ + H2 O S od ium phenoxide (a w ater-soluble salt) Dehydration • Dehydration: elimination of a molecule of water from adjacent carbon atoms gives an alkene – heating an alcohol with either 85% H3PO4 or concentrated H2SO4 – 1° alcohols are the most difficult to dehydrate – 2° alcohols undergo reaction at lower temperatures – 3° alcohols easily undergo acid-catalyzed dehydration at temperatures only slightly above room temperature Dehydration CH3 CH2 OH Ethan ol OH Cycloh exanol H2 SO4 180°C H2 SO4 140°C CH2 =CH2 + H2 O Ethylene + H2 O Cyclohexen e CH3 CH3 H2 SO4 CH3 CCH3 CH3 C=CH2 + H2 O 50°C OH 2-Meth yl-2-p ropanol 2-Methylpropene (Isobutylene) (t ert -Butyl alcoh ol) Your Lab Work BP = 160 oC BP = 89 oC Dehydration – when isomeric alkenes can be obtained, the alkene having the greater number of alkyl groups on the double bond predominates – examples: OH CH3 CH2 CHCH3 2-Butanol H3 PO4 -H2 O CH3 CH=CHCH3 + CH3 CH2 CH=CH2 2-Bu tene 1-Butene (80%) (20%) CH3 CH3 CH3 H2 SO4 CH3 CHCHCH3 CH3 C=CHCH3 + CH3 CHCH=CH2 -H2 O OH 3-Meth yl-2-b utanol 2-Methyl-2-b utene 3-Methyl-1-bu tene (major prod uct) Dehydration-Hydration • competing reactions – the following equilibrium exists C C + H2 O dehydration h yd ration An alk ene C C H OH An alcoh ol – in accordance with Le Chatelier's principle, excess water favor alcohol formation, whereas removal of water from the equilibrium mixture favors alkene formation Addition or Elimination Here A is acting negative and Q is acting positive. If you can remove A or Q from the reaction what happens? If you can remove the alkene from the reaction what happens? If you can precipitate the product from this reaction what happens? Eliminations This reaction is run is a solvent which the product salt is insoluble. How would you make the chlorocyclohexane from the cyclohexane? 3 More electrophylic Additions Oxidation • Oxidation of a 1° alcohol gives an aldehyde or a carboxylic acid – 1° alcohol to acid is carried out using K2Cr2O7 in aqueous sulfuric acid CH3 ( CH2 ) 6 CH2 OH 1-Octanol K2 Cr2 O7 H2 SO4 O K Cr O 2 2 7 CH3 ( CH2 ) 6 CH H2 SO4 Octanal O CH3 (CH2 ) 6 COH Octanoic acid – it may be possible to stop the oxidation at the aldehyde stage by distilling the mixture; the aldehyde usually has a lower boiling point than either the 1° alcohol or the carboxylic acid Oxidation – oxidation of a 2° alcohol gives a ketone K2 Cr2 O7 OH H2 SO4 2-Is opropyl-5-methylcyclohexanol (Menthol) O 2-Isopropyl-5-methylcyclohexan on e (Menth on e) – tertiary alcohols are resistant to oxidation CH3 OH 1-Methylcyclopentanol K2 Cr2 O7 H2 SO4 (no oxidation) Ethers • The functional group of an ether is an oxygen atom bonded to two carbon atoms – the simplest ether is dimethyl ether – the most common ether is diethyl ether CH3 -O-CH3 CH3 CH2 -O-CH2 CH3 D imethyl ether Dieth yl eth er Nomenclature • Although ethers can be named according to the IUPAC system, chemists almost invariably use common names for low-molecular-weight ethers – common names are derived by listing the alkyl groups bonded to oxygen in alphabetical order and adding the word "ether” – alternatively, name one of the groups on oxygen as an alkoxy group CH3 CH2 OCH2 CH3 D iethyl eth er OCH3 Cyclohexyl meth yl eth er (Methoxycycloh exane) Nomenclature • Cyclic ether: an ether in which one of the atoms in a ring is oxygen – cyclic ethers are also known by their common names – ethylene oxide is an important building block for the organic chemical industry; it is also used as a fumigant in foodstuffs and textiles, and in hospitals to sterilize surgical instruments – tetrahydrofuran is a useful laboratory and industrial solvent O Eth ylene oxide O Tetrahydrofu ran (THF) Physical Properties • Ethers are polar compounds in which oxygen bears a partial negative charge and each carbon bonded to it bears a partial positive charge – however, only weak forces of attraction exist between ether molecules in the pure liquid – consequently, boiling points of ethers are close to those of hydrocarbons of similar molecular weight – ethers have lower boiling points than alcohols of the same molecular formula CH3 CH2 OH Ethanol bp 78°C CH3 OCH3 D imethyl ether b p -24°C Making an Ether • Williamson Ether Synthesis Symmetrical Ethers Reactions of Ethers • Ethers resemble hydrocarbons in their resistance to chemical reaction – they do not react with oxidizing agents such as potassium dichromate – they do not react with reducing agents such as H2 in the presence of a transition metal catalyst – they are not affected by most acids or bases at moderate temperatures (but can eliminate) • Because of their general inertness and good solvent properties, ethers such as diethyl ether and THF are excellent solvents in which to carry out organic reactions Thiols • Thiol: a compound containing an -SH (sulfhydryl) group – the most noteworthy physical property of low-molecularweight thiols is their stench – they are responsible for scents such as those from rotten eggs and sewage – Skunk scent is due to a molecule which has two thiols CH3 CH=CHCH2 SH 2-Butene-1-thiol CH3 CH3 CHCH2 CH2 SH 3-Methyl-1-butanethiol Thiols - Nomenclature • IUPAC names are derived in the same manner as are the names of alcohols – to show that the compound is a thiol, the final -e of the parent alkane is retained and the suffix -thiol added • Common names for simple thiols are derived by naming the alkyl group bonded to -SH and adding the word "mercaptan" CH3 CH3 CH2 SH Ethaneth iol (Ethyl mercap tan) CH3 CHCH2 SH 2-Methyl-1-propan ethiol (Isobu tyl mercaptan) Physical Properties • Because of the small difference in electronegativity between sulfur and hydrogen (2.5 - 2.1 = 0.4), an S-H bond is nonpolar covalent – thiols show little association by hydrogen bonding – thiols have lower boiling points and are less soluble in water and other polar solvents than alcohols of similar molecular weight Thiol methanethiol ethan ethiol 1-bu tanethiol bp (°C) 6 35 98 Alcoh ol methanol ethan ol 1-bu tanol bp (°C) 65 78 117 Reactions of Thiols • Thiols are weak acids (pKa 10), and are comparable in strength to phenols – thiols react with strong bases such as NaOH to form water-soluble thiolate salts CH3 CH2 SH + NaOH Ethanethiol (pKa 10) H2 O CH3 CH2 S-Na+ + H2 O Sodium ethanethiolate Reactions of Thiols • The most common reaction of thiols in biological systems is their oxidation to disulfides, the functional group of which is a disulfide (-S-S-) bond – thiols are readily oxidized to disulfides by O2 – they are so susceptible to oxidation that they must be protected from contact with air during storage – disulfides, in turn, are easily reduced to thiols by several reducing agents. 2HOCH2 CH2 SH A thiol oxidation reduction HOCH2 CH2 S-SCH2 CH2 OH A disulfide Important Alcohols Coal or meth ane 2 H2 CO Carbon monoxide CO catalys t CH3 OH Meth anol H2 O, H2 SO4 CH2 =CH2 Ethylen e O2 catalyst CH3 CH2 OH Ethanol CH3 COOH Acetic acid O2 CH2 O oxidation Formaldehyde H2 SO4 180°C O H2 O, H2 SO4 H2 C CH2 Ethylene oxide CH3 CH2 OCH2 CH3 Dieth yl eth er HOCH2 CH2 OH Ethylene glycol Important Alcohols H2 O, H2 SO4 CH3 CH=CH2 Propene several s teps OH CH3 CHCH3 2-Propan ol OH HOCH2 CHCH2 OH Glycerin, glycerol Acid Base Equilibrium and Basic conditions What? • Substitution or Elimination – Leaving group must be weak base. – Attaching group is stronger base which is negatively charged or neutral with lone pair electrons. Acidic Conditions What? • Substitution or Elimination – Acid activates the leaving group so that it is a weak base. – Attaching group is a neutral lone pair atom. Alcohols, Ethers, and Thiols End Chapter 14 Review • Alkanes – Reactions of only – Combustion – Radical Halogenation Review • Aromatic – Reactions of only – Substitution (Electrophilic Aromatic Substitution) Review • Alkenes – Reactions of – Electrophilic Addition Review • Alkenes – Synthesis – Elimination • Basic • Acidic Review • Alkynes – Just do everything twice • 2 eliminations • Or • Two additons Review • Alcohols – Synthesis Review • Alcohols – Reactions of Review • Thiols – Synthesis Review • Thiols – Reactions Review • Alkylhalides – Synthesis Review • Alkylhalides – Reactions of