* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Proteins of extracellular matrix
Point mutation wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biochemical cascade wikipedia , lookup
Lipid signaling wikipedia , lookup
Biosynthesis wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Proteolysis wikipedia , lookup
Paracrine signalling wikipedia , lookup
Proteases in angiogenesis wikipedia , lookup
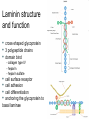
Extracellular matrix Jana Novotná Department of Med. Biochemistry 2nd Faculty of Medicine Charles University Composition of Extracellular Matrix (ECM) • Cells (mesenchymal origin) - fibroblasts - smooth muscle cells - chondroblasts - osteoblasts and epitelial cells • Organic fibrillar matrix • Organic nonfibrillar matrix • Water Function of ECM • Provides support and anchorage for cells. • Regulates and determine cells dynamic behaviour : - polarity of cells - cell differentiation - adhesion - migration • Provides mechanical support for tissues and organ architecture - growth - regenerative and healing processes - determination and maintenance of the structure • Place for active exchange of different metabolites, ions, water. Structure of ECM • collagen – the main ECM component, forms the main fibres • elastin • proteoglycans - heteropolysacharides • structural glycoproteins - fibronectin, laminin Collagen • The most abundant protein in the body, making 25%-35% of all the whole-body proteins. • Collagen contributes to the stability of tissues and organs. • It maintains their structural integrity. • It has great tensile strenght. • The main component of fascia, cartilage, ligaments, tendons, bone and skin. • Plays an important role in cell differentiation, polarity, movement. • Plays an important role in tissue and organ development. Collagen Structure Collagen is insoluble glycoprotein (protein + carbohydrate) Collagen polypeptide primary structure: - G –X–A–G –A–A–G –Y–A–G –A–A–G –X–A–G –A– –A–G –X–A–G–A–A–G –Y–A–G–A–A–G –X–A–G– –A–A–G –X–A–G –A–A–G –Y–A–G–A–A–G –X–A– G - glycine, X - proline or hydroxyproline, Y – lysin or hydroxylysine, A – amino acid Proline and hydroxyproline constitute about 1/6 of the total sequence, provide the stifness of the polypeptide chain. Carbohydrates : glucose, galactose • Three helical polypeptide units twist to form a triple-helical collagen molecule: a molecular "rope" which has some bending stiffness and does not undergo rotation. • The tropocollagen molecule has a length of approximately 300 nm and a diameter close to 1.5 nm. • In the typical fibrillar collagens, only short terminal portions of the polypeptides (the telopeptides) are not triple helical. Synthesis 1. Synthesis of a chains of preprocollagen on ribosomes. 2. Hydroxylation of lysine and proline in rER/Golgi by lysyl-5hydroxylase and prolyl-4hydroxylase. Reaction needs aketoglutarate, O2 and ascorbic acid is necessary to activate the hydroxylases and Fe2+ as a cofactor. 3. Glycosylation: addition of galactose and glucose to some hydroxylysine residues (galactosyl transferase and glycosyl transferase). 4. Assembly of a-chains to form procollagen. This involves the formation of disulphide bonds between parts of the polypeptide chains known as registration peptides, which occur at both ends of the preprocollagen. 5. Secretion of procollagen molecules by exocytosis into the extracellular space. 6. Cleavage of registration peptides is catalysed by procollagen peptidases. The resulting molecule is called tropocollagen. 7. Oxidation – deamination of the hydroxylysine, the removal of (NH2) group has a net oxidative effect and the formation of covalent crosslinks. Reaction is catalyzed by lysine oxidase (or catalase). 8. Self-assembly or polymerization of tropocollagen molecules form collagen fibrils. Cross-linkage between adjacent tropocollagen molecules stabilizes the fibrils. The typical staggered array of tropocollagen molecules in the collagen fibril. The telopeptides participate in covalent crosslinking. Collagen – Fiber Formation Collagen types I, II, III, V, IX, X Cross striated structure of collagen fiber reflect periodic comosition of individual tropocollagen molecules. Collagen fibrils of 1 mm diameter support the weight of 10 kg. Collagen Interactions Fiber forming collagen and nonfibrous collagen Tendon Cartilagous matrix Collagens Are Classified Into: 1. Fibril-forming collagens (I, II, III, V, X) 2. Fibril-associated collagens (FACIT) 3. Network-forming collagens 4. Anchoring fibrils collagens 5. Transmembrane collagens 6. Basement membrane collagens 7. Other collagens with unique function Major Collagen Types Fibril forming collagens (Most abundant collagen family - 90 % of the total collagens) Type Molecular composition Tissue distribution I [a1(I)2 2a(I)] Bone, dermis, tendon, ligaments cornea II [a1(II)]3 Cartilage, vitreous body, nucleus pulposus III [a1(III)]3 Skin, vessel wall, reticular fibres of most tissues (lung, liver, spleen, etc) V a1(V)a2(V)a3(V) Lung, cornea, bone, fetal membranes, together with type I collagen XI a1(XI)a2(XI)a3(XI) Cartilage, vitreous body Basement membrane collagens IV [a1(IV)2a2(IV)]; a1 – a6 Basement membrane Collagen IV has a short non-helical amino-terminal domain, a long Gly-X-Y repeat domain with numerous small interruptions and a highly conserved carboxylterminal globular NC1 domain. It polymerizes into a disulfide-bonded polygonal network via tetramerization between amino-terminal domains and dimerization between NC1 domains. Elastin • • • Elastin is a major protein component of tissues that require elasticity such as arteries, lungs, bladder, skin and elastic ligaments and cartilage. It is composed of soluble tropoelastin protein containing primarily, glycine and valine and modified alanine and proline residues. Tropoelastin is a ~65kDa protein that is highly crosslinked to form an insoluble complex. Elastin • The most common interchain cross-link in elastins is the result of the conversion of the amine groups of lysine to reactive aldehydes by lysyl oxidase. This results in the spontaneous formation of desmosine cross-links. Proteoglycans Proteoglycans represent a special class of glycoproteins that are heavily glycosylated (95%). They consisit of core protein with one or more attached glycosamino glycan chain(s). Glycosaminoglycans (GAG) Glycosaminoglycan (GAG) chains are long, linear carbohydrate polymers under physiological conditions they are negatively charged (due to the occurrence of sulfate and uronic acid groups). Disaccharide subunits are: 1. uronic acid D-glucuronic acid or L-iduronic acid 2. aminosugar N-acetyl glucosamin (GlcNAc) or N-acetyl galactosamin (GalNAc) • Amino sugars and uronic acids are the most common building blocks of the glycosaminoglycans. • In the amino sugars, the hydroxyl group at C-2 of the hexose is replaced by an amino group. This amino group is most often acetylated and sometimes sulfated. • In the uronic acids, C-6 of the hexose is oxidized to a carboxyl group. Linkage of GAGs to protein core by specific trisaccharide linker Hyaluronic Acid & Keratan Sulfate Chondroitin 6-sulfate & Heparan Supfate Dermatan Sulfate & Heparin Synthesis • The protein component is synthesized by ribosomes and transocated into the lumen of the RER. • Glycosylation of the proteoglycan occurs in the Golgi apparatus in multiple enzymatic steps. • First a special link tetrasaccharide is attached to a serine side chain on the core protein to serve as a primer for polysaccharide growth. Synthesis • Then sugars are added by glycosyltransferase. • Some glycosyltransferases catalyse sugar transfer to tyrosine, serine and threonine to give O-linked glycoproteins, • or to asparagine to give N-linked glycoproteins. • Mannosyl groups may be transferred to tryptophan to generate C-manosyl tryptophan • The completed proteoglycan is then exported in secretory vesicles to the extracellular matrix of the cell. Glycosaminoglycan Occurence • Proteoglycans can be categorised depending upon the nature of their glycosaminoglycan chains. • Hyaluronic acid (does not contain any sulfate) – non-covalent link complex with proteoglycans • Chondroitin sulfate • Dermatan sulfate • Heparan sulfate • Keratan sulfate – cartilage, bone – skin, blood vessels – basement membrane, component of cells surface – cornea, bone, cartilage, often aggregated with chondroitin sulfate Function of Proteoglycans • organize water molecules - resistent to compression - return to original shape - repel negative molecules • • occupy space between cells and collagen high viscosity - lubricating fluid in the joints • specific binding to other macromolecules • link to collagen fibers - form network - in bone combine with calcium salts (calcium carbonate, hydroxyapatite) • cell migration and adhesion - passageways between cells • anchoring cells to matrix fibers Structural Glycoproteins • Direct linkage to collagen or proteoglycans – anchoring collagen fibers to cell membrane – covalent attachment to membrane lipid • Major adhesive structural glycoproteins – fibronectin – laminin Fibronectin • Fibronectin is a high-molecular weight (~440kDa) glycoprotein • It binds to membrane-spanning receptor – integrin. • Fibronectin also binds extracellular matrix components such as collagen, fibrin and heparansulfate proteoglycans. • Cellular fibronectin is assembled into an insoluble fibrillar matrix. Fibronectin Structure • • • • Dimer connected at C-terminal by S-S linkage Rigid and flexible domains Cell binding domain RGDS (arg, gly, asp, ser) - binding receptor in cell membranes Domain is binding to - collagen type I, II and III - heparin sulfate - hyaluronic acid - fibrin Fibronectin Function • • • • Fibronectin is involved in cell adhesion, differentiation, growth, migration. Anchoring basal laminae to other ECM. Along with fibrin, plasma fibronectin is deposited at the site of injury, forming a blood cloth that stops bleeding and protects the underlying tissue. Fibronectin is necessary for embryogenesis (guiding cell attachment and migration during embryonic development) Laminin structure and function • • • • • • • cross-shaped glycoprotein 3 polypeptide chains domain bind - collagen type IV - heparin - heparin sulfate cell surface receptor cell adhesion cell differentiation anchoring the glycoprotein to basal laminae