* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download nitrogen_cycle
Soil compaction (agriculture) wikipedia , lookup
Crop rotation wikipedia , lookup
Soil respiration wikipedia , lookup
Soil contamination wikipedia , lookup
Soil food web wikipedia , lookup
Human impact on the nitrogen cycle wikipedia , lookup
Plant nutrition wikipedia , lookup
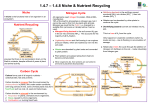
The nitrogen cycle Learning objectives: • To be able to describe how the element nitrogen cycles through the environment • To be able to explain the central importance of soil nitrates in the process and the importance of microbes and bacteria in the process. • To be able to identify the form in which nitrogen is found at different stages. The Nitrogen Cycle Why is nitrogen important? • It is part of proteins, DNA, RNA and chlorophyll. It is necessary for life. Where is most nitrogen contained? • Most of the nitrogen in the world is nitrogen gas (N2) 78% of the air • It is inert (unreactive) and most organisms cannot use it. • Organisms use nitrogen when it has been fixed (made into a more reactive, compound) The Nitrogen Cycle Nitrogen-fixation Biological • Mutualistic nitrogen fixing bacteria in root nodules produce ammonia • Free-living nitrogen-fixing bacteria in soil Industrial • the Haber process produces ammonium ions (NH4+) in fertiliser Natural Lightening bolts produce enough energy to react nitrogen and oxygen to form nitrates • NITROGEN-FIXING BACTERIA are found in the soil. They can convert nitrogen from the air into nitrates. • Plants are able to take up the nitrates with their roots. • These bacteria are commonly found in the roots of legume plants like peas, beans and clover. • They make lumps on the roots called root nodules. They change the nitrogen into nitrates that the plant can use. • N2 + 6H 2NH3 using the enzyme nitrogenase Nitrogen gas is changed into nitrates in the following ways • LIGHTENING • The energy in a lightning bolt can split the nitrogen molecule in the air allowing each nitrogen atom to react with oxygen to form nitrogen oxides. • These oxides are washed to the ground by the rain where they form nitrates. • DECOMPOSERS • Decay bacteria break down the dead remains and animal wastes releasing ammonia compounds into the soil • Ammonification. • Microbial saprophytes break down proteins in detritus to form ammonia in two stages: • first they digest proteins to amino acids using extracellular protease enzymes, • then they remove the amino groups from amino acids using deaminase enzymes. The Nitrogen Cycle NITRIFICATION • Nitrifying bacteria are autotrophic organisms that obtain their energy from ammonium or nitrite ions. • They oxidise ammonia to nitrate and use the energy released to live instead of respiration • These bacteria are useful to humans because they increase the fertility of the soil. The Nitrogen Cycle • Green plants absorb nitrogen in the form of ions: • ammonium (NH4+) or nitrate (NO3-) ions via the roots. • Green plants use these ions to make amino acids and proteins, which are passed to consumers by feeding. The Nitrogen Cycle • Nitrogen leaves organisms by excretion (urine), death and decomposition. • Saprophytes breakdown dead organic matter ( detritus) and release ammonium ions. The Nitrogen Cycle • DENITRIFICATION • In anaerobic soils denitrifying bacteria convert nitrate ions (NO3-) into nitrogen gas (N2) • This reduces soil fertility. • These only thrive in waterlogged soils (e.g. bogs). This is why carnivorous plants live there (no nitrates available to them in the soil). The Nitrogen Cycle The Nitrogen Cycle • MAN'S EFFECT ON THE NITROGEN CYCLE • Growing crops reduces the nitrogen content of soil • Adding nitrate fertilisers increases plant productivity • Crops with root nodules do not need fertilisers and can increase soil fertility if they are ploughed back into the ground • Excess nitrates are washed into rivers and lakes where they can cause EUTROPHICATION