* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download satellite cells
Extracellular matrix wikipedia , lookup
Cell culture wikipedia , lookup
List of types of proteins wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Tissue engineering wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cellular differentiation wikipedia , lookup
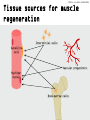
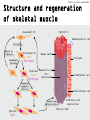
Manifestation of Novel Social Challenges of the European Union in the Teaching Material of Medical Biotechnology Master’s Programmes at the University of Pécs and at the University of Debrecen Identification number: TÁMOP-4.1.2-08/1/A-2009-0011 Manifestation of Novel Social Challenges of the European Union in the Teaching Material of Medical Biotechnology Master’s Programmes at the University of Pécs and at the University of Debrecen Identification number: TÁMOP-4.1.2-08/1/A-2009-0011 Dr. Péter Balogh and Dr. Péter Engelmann Transdifferentiation and regenerative medicine – Lecture 7 REGENERATION AND TRANSDIFFERENTIATIO N OF SKELETAL MUSCLE Conditions requiring skeletal muscle regeneration TÁMOP-4.1.2-08/1/A-2009-0011 Injury leading to extensive muscle damage Inherited diseases – Duchenne’s muscular dystrophy: • X-linked mutation of dystrophin gene • 1:3500 males affected • Dystrophin (2.4 Mb in size) is the largest known mammalian gene • Onset of the disease: DMD-afflicted patients are diagnosed in childhood. The progressive muscle-wasting disease affects striated muscle including limb muscles, diaphragm, and heart leading to cardiorespiratory failure, and death usually occurs in the Experimental models for studying muscle regeneration TÁMOP-4.1.2-08/1/A-2009-0011 • Mdx mice: spontaneous mutation of the distrophin gene (variable severities in different inbred mouse strains) • Distrophin/utrophin double mutant mouse • Canine X-linked muscular dystrophy (cxmd) is the best representation of DMD, but the phenotype is variable. TÁMOP-4.1.2-08/1/A-2009-0011 Embyonic development of skeletal muscle DT Limb DT NT MT Pax3/ Pax7 SC MyoD VLL NC Pax3 Nog Wnt11 Myf5 Bmp4 Nog Wnt1/3 MyoD Myf5 Myf6 MyoD Myogenesis Myogn Myf6, MyoD Pax3 Wnt7a Myf5 Shh SC TÁMOP-4.1.2-08/1/A-2009-0011 Transcriptional control of myogenic differentiation Injury Myofiber nuclei Fusion Differentiation Maturation Satellite cell Proliferation and RegeneratingSatellite cell (quiescent) self-renewal of satellite cells myofiber nuclei(quiescent) Quiescent Activation/Proliferation Myogenic stem cell Myogenic (MSC) progenitor cells (MPC) Differentiat ion Myoblast Myotube Transit Amplifying cells Cd34 Cdh15 Foxk1 Met Pax3 Pax7 Sox8 Sdc4 Sox15 Vcam1 Myf5 Myf6 MyoD Des Myog TÁMOP-4.1.2-08/1/A-2009-0011 Cellular sources for muscle regeneration • Satellite cells and their precursors • Endothelial cells associated with embryonic limb muscles • Mesangioblasts • Bone marow-derived stem cells • Pluripotent cells found within musclederived side population (SP) cells • Highly active Mdr-dependent expulsion of Hoechst 33342 dye TÁMOP-4.1.2-08/1/A-2009-0011 Tissue sources for muscle regeneration Interstitial cells Satellite cell Vascular progenitors Myofiber nuclei Bone marrow cells Muscle stem cells – satellite cells TÁMOP-4.1.2-08/1/A-2009-0011 • The satellite cells reside beneath the basal lamina of muscle, closely juxtaposed to muscle fibers • Approximately up 2–7% of the nuclei associated with a particular fiber • Heterogeneous composition: fusing/non-fusing subsets • Ontogeny: somite/perivascular cells expressing Pax3/Pax7 • Surface markers – Mouse: M-cadherin, CD34, VCAM, CD56, c-met (HGF-receptor) – Human: CD56 TÁMOP-4.1.2-08/1/A-2009-0011 Structure and regeneration of skeletal muscle Myofibril Quiescent SC Pax7+ Hematopoietic cel Activation Return to quiescence Asymmetric division Activated SC Pax7+ Myf5+MyoD+ Basal lamina Pericyte Myoblast Satellite cell (SC) Pax7Myonucleus Myf5+MyoD+ Endothelial cell Expansion (symmetric division) Interstitial cel Fusion and differentiation Myocyte MyoD+ Arteriole and capillaries Muscle fiber TÁMOP-4.1.2-08/1/A-2009-0011 Kinetics of muscle repair Maturation Differentiation Proliferation Activation 0 1 2 5 Days post injury 10 14 Problems with myoblast regeneration in Duchenne’s muscular distrophy TÁMOP-4.1.2-08/1/A-2009-0011 • Necessity for immunosuppression • Immunosuppressant drugs cause myoblast apoptosis • Short migratory distance following intramuscular injection – 100 injections/cm2 (totalling up to 4,000 injections in a single patient!) TÁMOP-4.1.2-08/1/A-2009-0011 Non-SCs contributing to muscle regeneration Vessels Blood Skeletal muscle Characterization MABs/pericytes Myoendothelial cells EPCs MSCs HSCs Side population CD133+ stem cells SCs and subpopulations MDSCs + CD133 stem cells Bone marrow HSCs Side population Mesenchymal stem cells MAPCs Other sources Adipose-derived stem cells MyoD-converted cells Dermis or other tissues Reprogramming iPS cells Mesenchymal differentation Expansion Autologous transplantation Commitment (if needed) Allogeneic transplantation (after genetic correction) TÁMOP-4.1.2-08/1/A-2009-0011 Summary • The prime candidates for skeletal muscle regeneration are the satellite cells, but cells from other sources (embryonic as well as non-embryonic) may also associate/promote the process. • Muscle regeneration is accomplished through (a) promoting vascular repair, (b) cellular differentiation from muscle stem cells and (c) possible transdifferentiation.