* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 20 - Simpson County Schools
Asymmetric induction wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Hydroformylation wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Homoaromaticity wikipedia , lookup
Aromaticity wikipedia , lookup
CHAPTER 20
Unsaturated Hydrocarbons
20.1 BONDING IN UNSATURATED
HYDROCARBONS
Alkene – general formula {CnH2n}
Unsaturated
hydrocarbon; suffix is -ene
(C=C)
must contain at least one carbon=carbon (double bond)
Diene: a compound that contains 2 C=C bonds.
Useful as starting components for polymers
20.2 NOMENCLATURE OF ALKENES
20.3 GEOMETRIC ISOMERISM IN ALKENES
20.4 CYCLOALKENES
Number the carbon atoms of the double bond as “1” and
“2” in the direction that gives the substituent
encountered first the smaller number.
Continue numbering around the ring.
Identify, name and number any substituents as is done
for (cyclo)alkanes.
20.5 PREPARATION AND PHYSICAL
PROPERTIES OF ALKENES
slightly lower boiling/melting pt than alkanes
due to lower molar mass
insoluble in water (nonpolar)
zero-4 carbons a gas at room temp, 5-17 C liquid,
above 17 solid
Produced through elimination reactions such as
cracking of petroleum & dehydration of alcohols.
Cracking or pyrolysis is the process of heating
saturated hydrocarbons to very high temperatures in
the presence of a catalyst.
Dehydration results in the elimination of water
from a reactant molecule.
20.6 CHEMICAL PROPERTIES OF ALKENES
Addition at the C=C double bond is the most
common reaction
Another common reaction of alkenes is oxidation
at the double bond
Ethene is called ethylene gas (the gas that is
emitted from ripening fruit and is a plant growth
hormone)
20.7 ALKYNES: NOMENCLATURE AND
PREPARATION
Alkyne – general formula {CnH2n-2}
Unsaturated hydrocarbon
suffix is -yne
(C≡C)
20.8 PHYSICAL AND CHEMICAL
PROPERTIES OF ALKYNES
must contain at least one carbon≡carbon (triple
bond)
gas at room temperature
slightly soluble in water but still considered nonpolar
Ethyne is commonly called acetylene; used in
oxyacetylene cutting/welding torches and as an
intermediate in the manufacture of many other
substances.
HC≡CH
very reactive due to triple bond; undergoes
addition reactions very readily.
Produce alkenes or alkanes upon addition
reactions. Some of which are the starting points
for further reactions – such as polyvinyl chloride
(PVC).
Acetylene (ethyne) is prepared inexpensively
from calcium carbide and water (the old minor’s
lamp reaction) or through cracking methane
20.9 AROMATIC HYDROCARBONS: STRUCTURE
general formula {C6H6} Unsaturated hydrocarbon;
ending may be “-benzene”
benzene ring; single/double bond – 6 C
liquids or solids at room temperature
nonpolar; therefore, insoluble in water
do not tend to undergo addition or elimination
reactions, but do undergo substitution reactions –
where one or more of the H atoms is substituted.
many are carcinogenic (cancer producing)
found in coal tar & tar from cigarette smoke
Some of these monosubstituted benzenes have
special names that become parent names.
20.10 NAMING AROMATIC COMPOUNDS
The C6H5 group is known as the phenyl group
and the name phenyl is sometimes used to name
compounds.
What is the “phenyl” part?
Polysubstituted benzenes – more than one substituent on
a benzene ring.
Naming: Carbon atoms are numbered starting at one of
the substituted groups. Numbering may be either
clockwise or counterclockwise but must be done in the
direction that gives the lowest possible numbers to the
substituent groups. When the compound is named as a
derivative of one of the special parent compounds (found
on previous page), the substituent of the parent compound
is considered to be on carbon 1 of the ring.
Draw the following:
2, 6-diiodophenol and toluene
Name:
20.11 POLYCYCLIC AROMATIC COMPOUNDS
Polycyclic aromatic compounds – other aromatic
ring systems besides benzene
Structures consist of 2 or more rings in which 2
carbon atoms are common to 2 rings.
20.12 SOURCES AND PHYSICAL PROPERTIES
OF AROMATIC HYDROCARBONS
Halogenation – when benzene reacts with chlorine or
bromine in the presence of a catalyst. Replaces H.
Nitration – when benzene reacts with a mixture of
concentrated nitric acid and conc. sulfuric acid at about
50°C, nitrobenzene is produced. Nitro (-NO2) is
substituted for H.
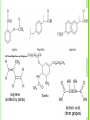
Draw the structure of 1,3,5-trinitrobenzene and
the explosive TNT, 2,4,6-trinitrotoluene.
Alkylation – an alkyl halide is reacted with
benzene in the presence of AlCl3 catalyst and the
alkyl group substitutes for an H atom.
REVIEW FUNCTIONAL GROUPS
HOMEWORK:
Required:
2-56 (even)