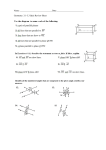
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download emboj2009118-sup
Molecular mimicry wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Adaptive immune system wikipedia , lookup
Innate immune system wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Supplementary information for Kim & Hempstead Supplementary information NRH2 is a trafficking switch to regulate sortilin localization and permit proneurotrophin-induced cell death Taeho Kim and Barbara L. Hempstead This file includes: Supplementary Figures S1 to S7 Supplementary materials and methods 1 Supplementary information for Kim & Hempstead Supplementary figures Supplementary Figure S1 Co-expression of NRH2 with sortilin or p75NTR in a subpopulation of spinal motor neurons. Spinal cord sections from C57/BL6 mice (P1) were double-immunostained using anti-NRH2 (1074), anti-sortilin (BAF2934), or anti-p75NTR antibodies (BAF1157). Scale bars, 20 m. 2 Supplementary information for Kim & Hempstead Supplementary figure S2 Co-expression of sortilin and NRH2 in the ganglion cell layer (GCL) of developing mouse retina. (A) Frozen sections of E15.5 mouse retina were subjected to doubleimmunofluorescence microscopy following immunostaining with control IgGs (a, b). Hematoxylin and eosin (H&E) staining documents retinal morphologies (c). ONF, optic nerve fibers; GCL, ganglion cell layer; IPL, inner plexiform layer; NBL, neuroblastic layer. (B) Both sortilin and NRH2 are highly expressed at the GCL of E15.5 mouse retina. Insets, higher magnification showing that a subset of ganglion cells co-express NRH2 and sortilin (arrowheads). (C) Sortilin immunoreactivity is reduced and intracellularly-restricted in the GCL of P1 retina with decreased NRH2 expression (arrowheads). Scale bar, 50 m. 3 Supplementary information for Kim & Hempstead Supplementary figure S3 NRH2 interacts with p75NTR and sortilin in vivo. Endogenous interaction of NRH2 with sortilin or p75NTR was assessed by immunoprecipitation of rat brain lysates from P1 animals (A), or DRG or spinal cord lysates from E17 rat embryos (B) with the indicated antibody, followed by probing with anti-NRH2 (1074 or JM), anti-sortilin or antip75NTR antibody as indicated. Arrowhead indicates the association of p75NTR and sortilin detected in DRG lysates in vivo. Lysate, 40 g. 4 Supplementary information for Kim & Hempstead Supplementary figure S4 The juxtamembrane domain of NRH2 promotes sortilin binding. Fulllength or truncated NRH2 constructs were co-expressed with sortilin in 293 cells, and their associations were assessed by co-immunoprecipitation with sortilin. Asterisks indicate incompletely N-glycosylated NRH2 intermediates, verified by N-glycanase reaction (data not shown). 5 Supplementary information for Kim & Hempstead Supplementary figure S5 NRH2 increases cell surface expression of sortilin but not p75NTR. HT-1080P/S cells were transfected with FLAG-NRH2, and sortilin (A) or p75NTR expression (B) on the cell surface expression was monitored by immunofluorescence detection in membrane non-permeabilized conditions using a 20X objective. Scale bar, 50m. 6 Supplementary information for Kim & Hempstead Supplementary figure S6 Expression levels of sortilin, p75NTR and NRH2 in primary neuronal cultures and cell kines. Cellular lysates obtained from cultured rat cortical neurons (Ctx), hippocampal neurons (Hip), DRG neurons (DRG), PC12 cells and HEK 293 cells transfected with sortilin and FLAG-NRH2 (293-SN) were treated with N-glycanase or not (37°C, 6 hours) following the manufacturer’s instruction (ProZyme, San Leandro, CA), and subjected to Western blot analysis. Arrowheads indicate that NRH2 is heavily N-glycosylated, but the extent of Nglycosylation slightly varies among cell types. DRG neurons express high levels of sortilin, p75NTR and NRH2, whereas cortical neurons express low levels of sortilin and NRH2 with no detectable p75NTR expression. In a longer exposure, both p75NTR and NRH2 were weakly detected in hippocampal lysates (data not shown). Separate immunoblotting results indicate that SCG neurons express similar or higher levels of p75NTR as well as NRH2 and sortilin as compared to DRG neuron (data not shown). 7 Supplementary information for Kim & Hempstead Supplementary figure S7 Effects of NRH2 expression on Trk signaling, and effects of Trk activation upon NRH2 levels. (A) NRH2 knockdown does not alter Trk-mediated pro-survival signals. DRG neurons-infected with CTL or shNRH2-expressing lentivirus were NGF-starved, and then treated with NGF (50 ng/ml) for 0, 15, and 60 min at DIV8. Harvested cell lysates were subjected to immunoblot analysis. No obvious alterations in the kinetics of Trk, PI3K (AKT phosphorylation) or MAPK activation (ERK1/2 phosphorylation) by NRH2 knockdown were detected, while sortilin expression levels were decreased in NRH2-silenced neurons. (B) NRH2 expression is not changed following TrkA activation. DRG cultures at DIV5 were NGF-starved for 20 hours in 25mM KCl containing media and then treated with NGF (50 ng/ml) or not for 12 hours in 25mM KCl supplemented media. The harvested lysates were subjected to Western blot analysis, and probed with the indicated antisera. 8 Supplementary information for Kim & Hempstead Supplementary materials and methods Cell cultures HEK 293 cells and human fibrosarcoma HT-1080 cells (ATCC# CCL-121) were maintained in DMEM containing 10% fetal bovine serum, supplemented with 100 U/ml penicillin, 100 U/ml streptomycin, and 2 mM glutamine. To generate HT-1080 cells stably expressing sortilin (HT1080S), or both p75NTR and sortilin (HT-1080P/S), HT-1080 cells were transfected with wild-type human p75NTR plasmid by electroporation (Amaxa, Cologne, Germany), and selected using 500 g/ml of G418 (Invitrogen, Carlsbad, CA). A p75NTR-positive clone or parental HT-1080 cells were further transfected with wild-type human sortilin, selected with 70 g/ml hygromycin (Invitrogen), and maintained in HT-1080 media supplemented with 40g/ml hygromycin and/or 250 g/ml G418. Lipofectamine (Invitrogen) was used to transfect DNA into 293 cells, and the amount of DNA for the same set of transfection was adjusted with corresponding control vector. Primary neuronal cultures Dissociated primary cultures of DRG neurons from embryonic day 15 (E15) were prepared from timed-pregnant Sprague Dawley rats. Neurons were plated on poly-L-lysine and collagen-coated plates in a Neurobasal media (Invitrogen) containing B27 supplement (Invitrogen), 2 mM glutamine, 1 mM sodium pyruvate, and 11 mM glucose. Cultures were maintained with 50 ng/ml NGF and 2.5 µg/ml fluorodeoxyuridine (5-FUDR) for 3 to 4 days. Primary cortical cultures were prepared from E18 rat embryos and maintained in Neurobasal media containing B27 and Lglutamine (0.5 mM) on poly-L-lysine coated plates in the presence or absence of 5-FUDR. In all cases, the percentage of glia in cultures was less than 5 % at DIV7. Immunoprecipitation and immunoblotting Co-immunoprecipitation experiments were performed by collecting cell lysates in TNE buffer (10 mM Tris, pH 7.5, 150 mM NaCl, 1 mM EDTA, and 1% Nonidet P40) with protease inhibitors (Sigma) after 40 hours of transfection, and immunoprecipitation was performed using anti-HA affinity matrix (Roche Applied Science, Indianapolis, IN) or anti-FLAG affinity gel (Sigma). The immune complexes were then separated by SDS-PAGE, transferred to a 9 Supplementary information for Kim & Hempstead polyvinylidene difluoride (PVDF) membrane (Milipore, Billerica, MA), and immunoblotted with appropriate antibodies. The immunoreactive protein bands were detected by enhanced chemiluminescence (GE Healthcare, Buckinghamshire, UK). For densitometric analysis, immunoreactive bands were scanned and intensity was quantitated using Image J software (NIH, Bethesda, MD). To detect endogenous interactions among NRH2, sortilin, and p75NTR, whole brains were dissected from embryonic day 17 (E17) rats, homogenized, and lysed in TNE buffer with protease and phosphatase inhibitors (Sigma). After centrifugation and preclearing with immobilized streptavidin (Pierce Biotechnology, Rockford, IL), lysates (3 mg / IP) were incubated with biotinylated anti-p75NTR (BAF1157), anti-sortilin (BAF2934), or normal goat IgG (BAF108). The immunocomplexes were precipitated using immobilized streptavidin and analyzed by Western blotting with anti-NRH2 (1074), anti-sortilin (monoclonal), and antip75NTR (9992) antibodies. Immunofluorescence microscopy HT-1080P/S cells or DRG neurons were fixed in 3% paraformaldehyde and permeabilized in blocking buffer containing 0.1% Triton X-100 for 30 min. Specimens were incubated with primary antibodies in blocking buffer followed by incubation with subtype-specific fluorescenated secondary antibodies. Stained cells were imaged using a Retiga EXi digital camera (QImaging, BC, Canada) mounted on Olympus (Center Valley, PA) BX61 microscope. For immunofluorescence analysis of tissues, paraffin embedded DRG or spinal cord sections (10 m thickness) from P1 mice were incubated with anti-NRH2, anti-p75NTR and anti-sortilin antibodies followed by incubation with secondary antibodies. For controls, the same amount of normal rabbit IgG or biotinylated goat IgG were applied. To selectively label sortilin, NRH2 or p75NTR present on the plasma membrane, HT1080P/S cells or DRG cultures were incubated with pre-cooled blocking buffer and incubated with primary antibodies detecting ECDs of target proteins followed by incubation with secondary antibodies at 4°C. Cells were then fixed with 3% paraformaldehyde followed by DAPI staining. HT-1080P/S cells were examined by epifluorescence microscopy and staining intensity of each flour in individual cells was integrated using Image J software (NIH, Bethesda, 10 Supplementary information for Kim & Hempstead MD). Ten to 20 cells / constructs / conditions were analyzed at random locations in each experiment, and normalized with NRH2 or GFP non-expressing adjacent cells. Confocal fluorescence microscopy was performed on DRG neuron specimens by using an LSM510 microscope (Carl Zeiss, Oberkochen, Germany) fitted with a Zeiss 63x, 1.4 numerical aperture objective with standard filter sets and standard (1 Airy disk) pinhole. For quantitative analysis of sortilin expression in CTL or NRH2-depleted DRG neurons, confocal and conventional fluorescence microscopy were performed in a membrane permeabilized (n=4) or nonpermeabilized condition (n=5). Twenty to 40 lentivirus-infected cells (GFP-positive) were examined randomly in each experiment and staining intensity was analyzed using Image J software. Generation of lentiviruses To generate a lentiviral construct encoding short-hairpin RNA (shRNA) for rat NRH2 (shNRH2), targeting forward and reverse hairpin oligos corresponding to position 340-358 of rat NRH2 (GenBank Accession number: NM_139259) (5’-TGCAACATCATTCCTGTCTATTC AAGAGATAGACAGGAATGATGTTGCTTTTTTC-3’ and 5’- ACGTTGTAGTAAGGACAG ATAAGTTCTCTATCTGTCCTTACTACAACGAAAAAAGAGCT-3’) were annealed and ligated into the HpaI and XhoI sites of the pLentiLox3.7 (pLL3.7) vector (Addgene, Cambridge, MA). The resulting dual cassette vector (pLL3.7-shNRH2), therefore, co-expresses enhanced green fluorescent protein (EGFP) as a reporter gene and rat NRH2-targeting shRNA (shNRH2) under the control of CMV and U6 promoter, respectively. To restore NRH2 expression in pLL3.7-shNRH2, three silent mutations in the shNRH2 target sequence (C344T, C347T and T356G; GenBank# NM_026012) were introduced to the FLAG-NRH2 plasmid by site-directed mutagenesis (Stratagene, La Jolla, CA), and the resulting shNRH2-resistant FLAG-NRH2 (FLAG-NRH2R) was replaced with EGFP in pLL3.7-shNRH2 construct using NheI and BsrGI sites (pLL3.7-shNRH2/FLAG-NRH2R). To overexpress NRH2, EGFP in pLL3.7 plasmid was replaced with FLAG-NRH2 (pLL3.7-FLAG-NRH2). All plasmid constructions were verified by sequencing. To prepare lentiviral stocks, 293FT cells (Invitrogen) were transfected with a shuttle plasmid (control pLL3.7, pLL3.7-shNRH2, pLL3.7-shNRH2/FLAG-NRH2R, or pLL3.7-FLAGNRH2) and the packaging plasmids pLP1, pLP2, and pLP/VSVG (Invitrogen). Virus-containing 11 Supplementary information for Kim & Hempstead medium was collected 48 hours after transfection, cleared of debris by low-speed centrifugation, filtered through 0.45 m filters, and frozen at -80°C. Viral titers, as determined by scoring GFP or FLAG-positive cells using fluorescent microscope, were up to 1X109 transducing unit/ml. Production of proNGF conditioned media HEK 293 cells were transfected with the control vector or HA-tagged proNGF, and culture medium was changed into serum-free media after 16 hours of transfection. Forty hours later, conditioned media were harvested, filtered (0.45 m filter) and analyzed by Western blotting using anti-HA (HA.11) or anti-proNGF (413) antibodies. The concentration of proNGF in HAproNGF-condition media was estimated as 10 ng/ml in comparison with band intensity from purified proNGF. 12