* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cellular Respiration
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Electron transport chain wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Citric acid cycle wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Microbial metabolism wikipedia , lookup
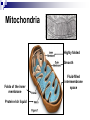
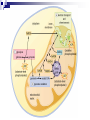
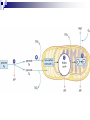
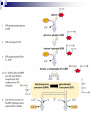
Cellular Respiration How do Organisms Get Energy? Photoautotrophs (e.g. plants) Chemoautotrophs (e.g. archaebacteria) Through the sun Make organic compounds without sun’s energy Heterotrophs (e.g. animal, fungi, most protists/bacteria) Through plants Glucose is the ultimate source of energy glucose stores energy in its bonds and cellular respiration is a way to acquire this energy Exergonic Redox Reaction C6H12O6 + 6O2 6H2O + 6CO2 + ENERGY as the bonds WITHIN the glucose molecule are broken down, energy is released To summarize even more, glucose stores energy in its bonds and cellular respiration is a way to acquire this energy! These bonds are broken through EXERGONIC OXIDATION-REDUCTION reaction C6H12O6 oxidized to 6CO2 , and 6O2 reduced to 6H2O each carbon in C6H12O6 is converted to CO2 each hydrogen in C6H12O6 is converted to ½H2O Combustion of Glucose In test tube In cellular respiration In cellular respiration energy is released from combustion of glucose. 34% of this energy goes to ATP; 66% goes to heat Aerobic vs Anaerobic Respiration C6H12O6 + 6O2 6H2O + 6CO2 + ENERGY In the above equation oxygen is the electron acceptor in the oxidation of glucose Most organisms are obligate aerobes – they require oxygen and cannot survive without it Obligate anaerobes (some species of bacteria) use other molecules as the final electron acceptor and must live in environments that has no oxygen Facultative anaerobes can tolerate aerobic and anaerobic conditions Mitochondria Highly folded Smooth Folds of the inner membrane Protein-rich liquid Fluid-filled intermembrane space Energy Transfer Terminology Substrate-level Phosphorylation: ATP forms directly in an enzyme-catalyzed reaction. Oxidative Phosphorylation: ATP forms indirectly through a series of enzymecatalyzed redox reactions involving oxygen as the final electron acceptor. Done mainly through compounds called energy carriers These reactions harness energy, which is eventually transferred to ATP Energy Carriers NAD+ and FAD+ are low energy, oxidized coenzymes that act as electron acceptors. When an electron(s) are added to these molecules, they become reduced to NADH and FADH2. In this case, reducing a molecule gives it more energy. Aerobic Respiration: Overview Occurs in Four Distinct Stages: 1. 2. 3. 4. Glycolysis: 10-step process in the cytoplasm. Pyruvate Oxidation: 1-step process in the mitochondrial matrix. Krebs Cycle: 8-step cyclical process in the mitochondrial matrix. Electron Transport Chain : Multi-step process in the inner mitochondrial membrane. Glycolysis - Overview Takes Place in the Cytoplasm Enzymes break down glucose (6 carbons) into two smaller molecules of pyruvate (3 carbons), releasing ATP Does not require oxygen Glycolysis – Key parts of process 2 ATPs are used in steps 1 & 3 to prime glucose for splitting. F 1,6-BP splits into DHAP and G3P. DHAP converts to G3P. 2 NADH are formed in step 6. 2 ATP are formed by substrate-level phosphorylation in both steps 7 and 10. 2 pyruvates are produced in step 10. Glycolysis Energy Yield & Products: 4 ATP produced – 2 ATP used = 2 net ATP 2 NADH 2 pyruvates Further processing in aerobic cellular respiration (if oxygen is available) Glycolysis – Energy Created Creates 2 molecules of ATP (2 x 31 kJ/mol) Yields 62 kJ of energy, from a possible 2870 kJ/glucose (only a 2.2% energy conversion) Most energy is still trapped in pyruvate and the 2 NADH molecules, but some lost as heat Earliest cells in Earth’s history thought to have used this method of energy metabolism since oxygen is not required and enough energy is produced to sustain life in unicellular organisms simple organisms today use glycolysis for energy In multicellular organisms glycolysis takes place first in the cytoplasm, and then more processes in the mitochondria to yield more energy Pyruvate Oxidation (if oxygen is present…) The following occurs for each pyruvate: 1. 2. 3. CO2 removed. NAD+ reduced to NADH and the 2-carbon compound becomes acetic acid. Coenzyme A (CoA) attaches to acetic acid to form acetyl-CoA. Pyruvate Oxidation Pyruvate Oxidation Energy Yield & Products: 2 NADH 2 acetyl-CoA 2 CO2 (released as waste) Acetyl-CoA CoA comes from vitamin B5 Proteins, lipids, and carbohydrates are catabolized to ‘acetyl-CoA’ It can be used to make fat or ATP [ATP] determines what pathway this molecule takes If O2 is present, ‘acetyl CoA’ moves to the Kreb’s Cycle (aerobic respiration) If O2 is NOT present, ‘acetyl CoA’ becomes ‘lactate’ (anaerobic respiration / fermentation)