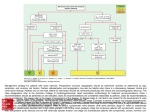
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download of kazakh state medical academy
Survey
Document related concepts
Heart failure wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Pericardial heart valves wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Infective endocarditis wikipedia , lookup
Coronary artery disease wikipedia , lookup
Myocardial infarction wikipedia , lookup
Cardiac surgery wikipedia , lookup
Artificial heart valve wikipedia , lookup
Jatene procedure wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Aortic stenosis wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Transcript
MINISTRY OF PUBLIC HEALTH OF THE REPUBLIC OF KAZAKHSTAN EDUCATIONAL-METHODICAL SECTION OF KAZAKH STATE MEDICAL ACADEMY ON SPECIALITIES OF HIGHER AND POST GRADUATE EDUCATION KARAGANDA STATE MEDICAL ACADEMY Taijanova D. J., Toleuova A.S., Kotlyarova O.A., Yevseenko L.V. SPECIAL LECTURES OF RHEUMATHOLOGY Educational manual Karaganda 2007 UDK 616.002.77 BBK 54.12я7 S 78 The educational methodical manual “Special lectures of reumathology" of Taijanova D.J.,Toleuova A.S.,Kotlyarova O.A.,Yevseenko L.V.. Karaganda, 2007 – 91p. Reviewers: Umbetalina N.S. – d.m.s., professor, the head of department of internal diseases EIF and SPE. Molotov – Luchanskiy V. B. - c.m.s., assistant professor, the head of department for the assistant professors’ course of nursing management at KSMA. Seisenbekov T.Z.- professor of the internal disease department of FPDME of Kazak Medical Academy,professor In this educational methodical manual, are presented the contemporary concepts about etiology, classification and pathogenesis of frequently meeting diseases of rheumathology, the contemporary diagnostics methods, the criteria of diagnostics and differential diagnostics of diseases, contemporary approaches to the treatments. Educational methodical manual is intended for the students of High Medical School - Institute of Higher Education and interns. Confirmed and recommended for edition of Academic Council of KSMA Protocol № 9____ of ___29.03. 2007г 2 List of abbreviation AV – atrio- ventricular ARF - Acute rheumatic fever ASL-O - antistreptolysin O DNA-ase – desoxyribonucleinase CHF- chronic heart failure ECG - the electro-cardiography EchoCG- echo-echocardiography LV- left ventricle RV – right ventricle ACE – angiotensine converting enzyme INR - international normalized ratio HIV - Human immunodeficiency virus SLE - systemic lupus erythematosus RNA – ribonuclein acide PCR - polymerase chain reaction ICD-10 - international classification of diseases ESR - Erythrocyte sedimentation rate ANA - antinuclear antibody MRI – magnetic resonance investigation NSAIDs – non-steroid anti-inflammatory drugs 3 1. ACUTE RHEUMATIC FEVER 1.1. Definition Acute rheumatic fever (ARF) is systematic inflammatory disease of the connective tissues with the main localization of the process in cardiovascular system, developed due to acute infection (Group A beta hemolytic streptococcus of) in predisposed individuals, mostly in children up to the age of 7-15 years. The clinical manifestations include polyarthritis, caditis, subcutaneous nodules, erythema marginatum, and chorea in varying combinations. 1.2. Epidemiology ARF occurs in all parts of the world: there is no known racial predisposition. In temperate climates, ARF peaks to the cooler months of the year, in winter and early spring or shortly after schools open in the fall. Data of the last 10 years researches show that it is more common in developing countries where 80% children of the world live. In underdeveloped countries it remains a major problem, with annual incidence rates of 100 per 100 000 in the childhood age group, compared with fewer than 5 per 100 000 in most Western countries. In the 1980s an unexplained increased incidence was noted in a number of regions in the United States, but levels there are still well below those in underdeveloped regions. Outbreaks of the disease were reported in Salt Lake City, Utah, Columbus and Akron, Ohio, Pirrsburgh, Pennsylvania, Nashville and Memphis. The lagest outbreak was in Salt Lake City and its environs, where approximately 200 cases occurred between 1985 and 1989. Equally surprising was the fact that in many of these outbreaks m the victims were predominantly white, middle-class children dwelling in the suburbs. According to generalized data of J.Padmavati (1982), rheumatic fever among schoolboy in developing country is 6-22 on 1000 children populations, at the some time it is the progressive course of the disease with valvular heart diseases formation, pulmonary hypertension and early death-rate because of the heart failure. Social problems and poor diet and medical status of the country have also its own importance in the development of rheumatic fever. In the countries of Former Soviet Union for the last 25 years mortality and morbidity of rheumatic fever decreases 73 times more. For instance, 10-years longrange study of primary rheumatic fever morbidity has shown its reduction with 0,54 of 1000 the child populations in 1970, to 0,18 - in 1979 (Annekova Z.A., 1981.) In some communities there is a high incidence of rheumatic fever in certain racial groups. In Britain, rheumatic fever is much more common in Asian immigrants, and in New Zealand it is eight times more common in the Polynesian than in the Caucasian population. Nevertheless, specific racial predilection has never been substantiated. Crowding, poor housing, poor hygiene, and inadequate medical care all contribute, overcrowding being the predominant factor. Increased rheumatogenicity of streptococci has frequently been demonstrated during epidemics. Possibly heightened rheumatogenicity occurs with repeated upper 4 respiratory infections in crowded communities where there is an undue acceptance of symptoms of infection. In some country with high economics and culture Sweden, Denmark, Switzerland and others the problem of the rheumatic fever has forfeited the past importance that has served the cause to significant reduction of attention to it both scientist, and health authorities. This reduction is conditioned by combination of the several main factors: 1) improvement of living conditions of the population in economic developed country, increasing level of natural immunity, and the decrease of streptococcal environment and danger of streptococcal infections; 2) detection and active treatment of the carriers of streptococcal infection, antibacterial and surgical treatment acute chronic tonsillitis, maxillitis; 3) improvement of the formal forms of the fight with rheumatic fever and increasing to qualifications by a therapeutics and pediatrician in the field of theumatology. However, it is impossible completely to exclude and possible role of evolutionary changing responsiveness of population with progressing increasing level of natural immunity, as well as changing virulence of etiological factor of streptococcus 1.3. Etiology An association between septic throats and rheumatic fever was noted in the nineteenth century, and in the 1930s there were many reports of outbreaks of rheumatic fever following tonsillitis or scarlet fever in closed communities. The development of serological tests for streptococcal infections established the role of the Group A b-haemolytic streptococcus. This was confirmed when penicillin treatment sharply reduced the incidence of rheumatic fever in epidemics of streptococcal infections, and effective prophylaxis came close to abolishing recurrences. The way in which the streptococcus causes rheumatic fever is still only partly understood. Characteristics of the streptococcus are critical. The organism must be able to attach firmly to pharyngeal cells and produce a brisk antigenic response. Impetigo strains do not cause rheumatic fever. Steins representing a number of the more than 80 M-protein serotypes of group A streptococci can cause ARF. “There is a substantial body of the evidence to indicate, however, that group A streptococci vary in their rheumatogenic potential. Stains clusters or epidemic usually belong to a limited number of serotypes- 3:5:18:24 and others and are often heavily encapsulated, as the evidence by their growth as muco8id colonies on blood agar plates. Streptococci do not persist in cardiac tissues in rheumatic patients. Both the peptidoglycan moiety of the cell wall and streptolysins O and S have produced cellular damage in experimental situations, but no animal model of rheumatic fever has been developed. The host reaction The latent period, the generally higher antistreptococcal titres in patients who develop rheumatic fever compared with those who do not, and the transient 5 appearance of circulating immune complexes, point to an abnormal immune response to streptococcal antigens cross-reactive to mammalian tissues. There is now a strong evidence for this, both on a humoral and cellular level, implying a genetically programmed predisposition. A monoclonal antibody to B cell surface antigens, called D8/17, has been found in over 90 per cent of patients with rheumatic fever but in only 10 per cent of healthy controls. Much needs to be done before these pieces of the jigsaw can be put together. 1.4. Pathogenesis Pathogenesis is unknown. These possible mechanisms are (1) a hyperimmune response, either autoimmune or allergic in nature, particularly streptolysins S and O, both of which can initiate tissue injury; (2) inflammation mediated by antigeantibody complexes, perhaps localizes to sites of tissue inhury; and (3) “qoutoimmuni” phenomena induced by the similarity of certain streptococcal and human tissue antigens. An immunologic explanation is considered most plausible, although the latter mechanism cannot be entirely ruled out. At autoimmine reaction to a streptococcal infection would hypothetically produce tissue damage or manifestations of rheumatic disease, as follows: 1) group A streptococcus would produce pharyngeal infection; 2) streptococcal intigen would result in antibody production in a hyperimmune host; 3) antibodies would react with the streptococcal antigen and with host tissues that are antigenically similar to streptococcus ( i.e., antibodies are unable to distinguish streptococcal antigen form cardiac tissue antigen); 4) autoantibody reacting with host tissues would produce tissue damage. It leads to one of the cardinal sign is Ashoff- Talalaev’s body. 1.5. Pathomorphology Ashoff-Talalaev’s body (rheumatic body) is formed by 3 irregular basophilic cells, sometimes multinucleated, gigantic multinucleated cells of myogeic origin with eosinopylic cytoplasm, cardiohystiocytes (Anischkow’s myocute), lymphoid and plasmatic cells, labrocytes and leucocytes. Aschoff- Talalaev’nodules (granulomatous nodular lesion with central necrotic area) is often located in perivascular connective tissues or in the myocadial interstitial (often of the left ventricle), papillary muscles, septum, and also in endocardium in the vascular walls in rheumatic vasculitis. At present in path anatomical investigation bodies are found less than before, which is related with the change of morphological and clinical picture of the rheumatic fever. Other morphologic substrate of the heart lesion is non- specific inflammatory reaction of serous membrane like in joints. It is formed of the edema of the intramuscular connective tissues by saturation of fibrin, infiltration by cellular elements mainly by polymorhous leucocytes and lymphocytes. In rheumatic fever the inflammation of the muscular fibers also occurs like hypertrophy or atrophy to the complete lysis of myocytes with furthers their regeneration when the inflammatory process decreases. 6 1.6. Pathology So, the classic histological feature of rheumatic fever is the Aschoff nodule, a perivascular lesion with a central core of necrotic material surrounded by large cells with polymorphous nuclei and basophilic cytoplasm, and an outer layer of lymphocytes. Nodules have a widespread distribution in connective tissues, including those of joints, tendons, and blood vessels. In the heart they are found in myocardial tissue, most valvular lesions consisting of less organized collections of chronic inflammatory cells. The nodules heal by fibrosis, sometimes leading to extensive interstitial myocardial fibrosis. The mitral valve leaflets become thickened, with impairment of closure exacerbated by ring dilation. Progressive distortion of the leaflets and subvalvular apparatus may lead to severe regurgitation. Leaflet and chordal fusion may lead to mitral stenosis, the structure of the valve becoming severely distorted by progressive fibrosis and eventually calcification. This process usually progresses slowly over many years, but proceeds rapidly in some children in developing countries. Similar but less severe changes occur in the tricuspid valve in up to 10 per cent of cases. In the aortic valve thickening of the leaflets is also seen, leaflet edges developing a rolled appearance. The dominant lesion is usually aortic regurgitation. 1.7. Classification A new classification of rheumatic fever has been adopted by the Plenary Session of the Association of Rheumatologists of Russia in May 2003. This classification envisages the abandonment of the term "rheumatism" in its former interpretation. The term "acute rheumatic fever" (ARF) appears to be more justified since it makes a physician elucidate its association with Group A streptococcal infection of the throat and prescribe antibiotics for eradication of this infection in its acute period (primary prophylaxis) and for the prevention of repeated attacks (secondary prophylaxis). The current interpretation of the clinical syndromes/symptoms and laboratory parameters that entered into diagnostic criteria for ARF is presented. There are 2 types of outcome of the disease. In case of recovery we are dealing with the complete regression of the clinical symptomatology of ARF, normalized laboratory parameters and no residual changes. Chronic rheumatic diseases of the heart are considered to mean a disease characterized by a cardiac valvular lesion as postinflammatory marginal fibrosis of valvular leaflets or cardiac abnormality (failure and/or stenosis) developed after prior ARF. Emphasis is laid on the significance of evidence of Group A streptococcal infection that precedes the development of the disease. Examples of clinical diagnosis in accordance with the terminology of the International Classification of Diseases, the 10th edition, are given in the paper (table 1). 7 Table 1- Classification of rheumatic fever Clinical variant Clinical sings outcome Main Additional Acute rheumatic Carditis Fever convalescence fever Polyarthritis Stage of heart failure CSV NYNA 0 I IIA IIB fever III 0 I II III IV Chronic Repeated Carditis, Arthralgia, rheumatic rheumatic fever polyarhtritis abdominal of the heart Chorea, syndrome, - without of Erythema serositis valvular marinatum, disease subcutaneous - with nodules valvular disease 1.8. Acute rheumatic fever Postinfectious complicatin of А- streptococcal tonsillitis or pharyngitis like systemic inflammatory disease of connective tissue with the primary location in cardiovascular system (cariditis) joints, (migratory polyarthritis), the brain (chorea), and the skin (erythema marginatum, subcutaneous nodules), in predisposed individuals, mostly in children up to the age of 7-15 years, due to autoimmune reaction of the body in streptococcal antigen and cross reactivity with similar auto gens affected human tissues (molecular mimcria phenomen). 1.9. Repeated rheumatic fever - new episode of ARF (but nor recurrent of the first), that is developed as carditis, not often carditis and polyarthritis, seldom chorea. 1.10. Clinical feature Rheumatic fever is rare under 4 years of age, most cases occurring in the 6- to 15year age group. Some two-thirds of patients give a history of prior sore throat, usually 1 to 5 weeks before the development of rheumatic symptoms. Although there is considerable variation in the clinical presentation of rheumatic fever, most cases feature migratory polyarthritis, carditis, or both. Presentation may be abrupt, with fever and joint pains, or more gradual with a subacute course. In some cases no acute bout is recognized at all, this patient presented with established rheumatic heart disease. So, there are three stages of rheumatic fever: 1) relation with acute streptococcal infection; 2) presence of absolute signs of rheumatism (Duckett- Jones criteria); 3) tendency to formation of valve defects. There are 3 periods of the progression of rheumatism: 1 st period prolongs from 2-4 weeks after streptococcal infection, progresses without any symptoms or with signs characterized for stage of convalescence. 2nd period is characterized by the 8 development of polyarthritis, carditis and other signs characterized for primary rheumatic fever. In 3rd period observed multiple signs of the recurrent rheumatic fever with progressive cardiac defects and development of hemodynamic disorders are observed. Such characteristics show development of rheumatic fever from the onset to end of disease with functional failure of the most affected organ- the heart. Rheumatic polyarthritis Rheumatic polyarthritis is present in as many as 75% of cases and it is the cardinal diagnostic criteria of primary rheumatic fever, Arthritis affects larger joints, especially wrists, elbows, knees, and ankles. Objective signs are usually limited to minor warmth and swelling, but pain may be excruciating, especially with pressure or movement. Characteristically one joint will be affected for 2 to 3days and then the inflammatory process moves to another region, but two or more joints may be affected simultaneously to some degree (migratory arthritis). The arthritis of rheumatic etiology is extremely painful and lasts for 1-5 weeks and subsides without residual deformity. Prompt response of therapeutic dosage of salisylates or nonsteroid-antiinflammatory drugs is characteristic. Arthralgia without objective signs may occur in other joints or may be the only feature, symptoms varying from minor discomfort to severe pain. Untreated joint pains are usually settle over 1 to 4 weeks. Rheumatic carditis Rheumatic fever involves the endocarditis, myocaridium, and pericardium, and thus the disease is capable to include a true pancarditis. Carditis is the most important manifestation of AFR because it is the only one that can cause significant permanent organ damage or death. The diagnosis of carditis requires the presence of one of manifestations: - sinus tachycardia; - pericarditis but it does not lead into constrictive pericarditis; - cardiomegaly detected by physical signs, X rays and echocardiography; - congestive heart failure; - organic cardiac murmurs not previously present; - functional murmurs- apical systolic, apical mid-diastolic (Cary-Coombs murmur), basal diastolic; - prolongation of PR interval; - ECG changes of pericarditits (raised ST segment) or myocarditis (inverted or flattened T wave) or 1st degree heart block. Severe course of carditis is more typical for children and adolescent than for adult patient. The onset of the disease is marked by significant dyspnea on physical loading and rest, constant pains in the heart, heartbeat. In the early stages of congestive heart failure, rapid distention of the hepatic capsule may lead to the right upper quadrant aching and tenderness over the liver. Recurrent carditis has the features of all the firths rheumatic attack, but the process is heaver and result in combined and multifunction heart valvular diseases, gaining chronic long-term or latent current with unstable effect of antirheumatic therapy. 9 The Rheumatic pleurisy is often manifestation of rheumatic polyserositis; it is accompanied by a pain in breathing, pleural friction rub in the zone of the effusion and increasing of the body temperature. Rheumatic chorea (Sydenham’s chore, Chorea minir, “St Vitus’ Dance” It develops in less than 10% of cases; however it is the most diagnostic feature of rheumatic fever. Girls are more frequently affected and occurrence in adults is rare. Chorea is characterized by personality changes, emotional liability, and rapid, purposelss, involuntary movements, most all other evidence of extremities and face. The arms and legs flail about in erratic, jerky, uncoordinated movement that may sometimes be unilateral (hemichorea). Facial tics, grimaces, grins, and contortions are evident. The speech is usually slurred or jerky, the tongue, when protruded, retracts involuntarily, while asynchronous contractions of lingual muscles produce a “bag of worms”. The involuntary motions disappear during sleep and may partially be suppressed by rest, sedation and volition. Patients with chorea display generalized muscle weakness and an inability to maintain a titanic muscle contraction. Thus, when the patient is asked ot squeeze the examiner’s fingers, a squeezing and relaxing motion occurs that has been described as “milkmaid’s grip”, “flabby shoulders”- the patient’s head dives into shoulders when the patient is raised by armpits (axillary space), “Filatov’s eyes a and tongue”are present: the patient can not to close the eyes and show the tongue and other symptoms are marked. Erythema Marginatum Rheumatic fever affects skin- erythema marginatum is an erytheamtous macule or papule and then extends outward while the skin in the centre returns to normal Adjacent lesions coalesce, forming circinate or serpiginous patterns. The lesions may be raised or flat, are vary greatly in size and mostly on the trunk and proximal extremities, sparing the face. The lesions are evanescent, migrating from place to place, at times changing before the observer’s eyes, and leaving no residual scarring. Process may go on intermittently from weeks to months uninfluenced by anti- inflammatory therapy. In the great majority if a case, erythema marginatum is accompanied by cariditis; it also tends to be associated with subcutaneous nodules. Subcutaneous nodules These are firm, painless subcutaneous lesion that varies in size from a few millimeters to approximately 2 cm. The skin overlying them is freely movable and is not inflated. Sites of predilection include the extensor surfaces of the elbows, knees, and wrists, the occiput, and spinous processes of the thoracic and lumbar vertebrae. Nodules ordinarily do not appear until at least 3 weeks after the onset of an attack, usually lasting from 1 to 2 weeks and disappear rapid without scarring. These are rare major manifestations occurring in less than 10% of cases. 1.11. Other associated features Other features are not specific enough to be diagnostic. Epistaxis is quite common, as there is abdominal pain, particularly at the onset of an attack. Pleural effusions may occur, especially in association with pericardial effusions. 10 1.12. Diagnosis Although diagnosis is easy in many cases, a number of factors may cause problems. 1. There is no single diagnostic test. The diagnosis is made on the basis of the clinical pattern with support from laboratory tests. 2. Many illnesses may mimic the clinical presentation. These include infective endocarditis and viral and other causes of polyarthritis, especially early rheumatoid arthritis. Monarthritides, including those of septic and traumatic origin, can cause initial confusion. Acute streptococcal infections may be followed by a period of mild fever, lethargy, malaise, and elevated erythrocyte sedimentation rate, sometimes lasting 2 to 3 weeks. These reactions may be difficult to distinguish from rheumatic fever but they do not show major rheumatic manifestations and generally have a briefe course. Rheumatic fever rarely settles in less than 2 months. It may continue for several months, particularly in patients with carditis where 2 to 4 per cent are still active for 6 months. 3. The murmur of a minor congenital cardiac lesion may first be noted during a viral infection. Even in expert hands the distinction of mild congenital aortic stenosis or prolapse of the mitral valve from a rheumatic lesion may be impossible. Changing murmurs and the evidence of involvement of both aortic and mitral valves favour rheumatic fever. 4. The features of rheumatic fever may vary according to severity and the time of presentation. With early presentation diagnosis may only be possible after a period of observation of clinical progress and laboratory tests. With late presentation some clinical and laboratory abnormalities may have subsided and the clinician may have to be content with a diagnosis of “probable” rheumatic fever. With a view to providing a basis for diagnosis and minimizing overdiagnosis, Duckett Jones published diagnostic criteria, subsequently modified by the American Heart Association with additional comments made by the World Health Organization working party. 1.13. Guidelines for the diagnosis o f initial attack of theumatic fever, (Jones – Kisel, updated 1992) Diagnostic criteria were at first formulated by A.A. Kisel in 1940, who differentiates 5 absolute criteria of rheumatic fever: subcutaneous nodules, erythema marginatum, chorea, polyarthritis and carditis. Later these signs were included to basic criteria of diagnosis by T. Duckett Jones in 1944 and subsequently modified by committee of the Heart Association (1992). Following are the diagnostic criteria of rheumatic fever with additions by A.I. Nesterov 1973: I. Major Manifestations: 1. Carditis- characterized by relative cardiac dullness increasing, systolic diastolic murmur in the point of apex beat, typical congiguration of the heart pericardium friction rub, prolonged PR interval on ECG, sings of heart failure in children or adolescents till 25 years old, without any reasons. 2. Polyarthiritis- arthralgia, limitation of movement of affected joins, defiguration (swelling, warmth, erythema). 11 3. Cchorea minor- is rapid purposeless, involuntary movement, most noticeable in extremities and face. 4. Subcutaneous nodules are firm, painless subcutaneous lesions that vary in size form a few millimeters to approximately 2 cm. Sites of predilection include the extensor surfaces of the large joints. 5. Erythema marginatum is the rash as an erythematous macule or papule which then extends outward in the skin in the center to normal. They appear mostly on the trunk and proximal extremities, sparing the face. 6. Rheumatic anamnesis is associated disease with the previous streptococcal infection, and acute and exacerbation of chronic tonsillitis and the presence of somebody with rheumatic fever in a family 7. Effective therapy by antirheumatic drugs lasts during 3-5 days of treatment. II. Minor criteria: - Fever - Arthralgia - Previous rheumatic fever - Raised ESR and C- reactive protein - Leucocytosis - Prolonged PR interval on ECG III. Specific laboratory sings: - Leucocytes, - ESR, seromucoid proteins, fibrinogen, a1 and a2- globulins, - C- reactive protein and. - Circulatory antibodies and their high titer to streptolysin, streptokinase, and streptohyaluronidase - High titer of circulatory anti streptococcal antibodies. - Presence of hyper immune reaction may be detected by the presence of ASL-O, DNA-ase B and other enzymes. Two major manifestations, or one major and two minor criteria, make rheumatic fever very likely if supported by the evidence of the preceding streptococcal infection. The absence of a streptococcal infection make the diagnosis doubtful, except two situations in which rheumatic fever is first discovered after a long latent period from the antecedent infection (1) Sydenham’s; (2) prolonged insidious carditis. For the diagnosis of persistent, latent and atopic progression of rheumatism used next syndromes (by A.I. Nesterov 1976). First clinic- epidemiological syndrome is the presence of relation with streptococcal infection. Second, clinic- immunologic syndrome is presence of onset, palpitation, arthralgia, and sub febrile temperature after throat infection and high titer of streptococcal antibodies, change in humeral immunity system, symptoms of auto immunization, and biochemical signs of inflammation and so on. 12 Third, syndrome is cardiovascular which may be examined with the help of clinical, laboratory and instrumental methods of investigations, for the presence of carditis and extra cardiac locations of rheumatic fever. 1.14. Laboratory tests A throat swab should be taken although group A streptococci are isolated in only 15 to 20 per cent of cases. Blood cultures and other appropriate investigations are required to exclude infective endocarditis. Mild anemia and leucocytosis are common. 1.15. Acute phase reactants The two most useful acute-phase reactants are the erythrocyte sedimentation rate and the C-reactive protein levels, which should be measured weekly. When the patient presents congestive heart failure the initial erythrocyte sedimentation rate may be high to normal, rising over the following 2 weeks as symptoms improve. C-reactive protein levels are not affected in this way. Later in the illness mild elevation of the erythrocyte sedimentation rate may persist after the C-reactive protein level has returned to normal. It may then be reasonably assumed that residual rheumatic activity is of low grade, although the inflammatory process may not have completely subsided. Streptococcal serology Streptococcal titres should be measured in three occasions with 2-weeks intervals. Multiple tests are desirable, usually the streptococcal antistreptolysin O, antiDNAse, and antihyaluronidase. A two-fold rise in titre may be considered diagnostic. Because of the late presentation, titres are usually convalescent and are considered positive when they exceed a given level - 250 for antistreptolysin O, 320 for anti-DNAse, and 300 for antihyaluronidase. Using these criteria, positive tests are obtained in over 90 per cent of patients with acute rheumatic fever. The inexpensive streptozyme test is often favoured but shows poor reproducibility and specificity. 1.16. Course The average duration of an untreated attack of ARF is about 3 months. The duration tends to be longer- up to 6 months in patients with severe carditis. Fewer than 5% of patients have continuing thematic activity for longer than 6 months. In a few of these the disease is limited by chorea and is otherwise benign. Other patients demonstrate the evidence of persistent inflammatory activity, including arthritis, cardits, and subcutaneous nodules. “Chronic rheumatic disease” occurs more frequently in patients who had one or more previous involvement in chronic rheumatic fever tends. 1.17. Management Admission to hospital is highly desirable for diagnosis and management, and for the education of a patient and family. Following initial blood cultures, a (1.2 mega units) course of penicillin is given, either intramuscular benzathine penicillin or 10 days treatment with oral penicillin. Long-term prophylaxis should then be continued. 13 Rest Rest remains the cornerstone of treatment. This recommendation has never been subjected to a controlled trial but is based on clinical observation when symptoms persist and carditis progresses in many patients who are not rested. Patients should remain on bed-chair rest until symptoms subside and acute-phase reactants have been normal for two successive weeks. It is likely that earlier mobilization will prove less important in patients who have had arthritis alone and, provided symptoms are settled and the C-reactive protein level is normal, gradual mobilization may be considered in those with a prolonged course even when the erythrocyte sedimentation rate remains mildly elevated. In patients with carditis, however, the period of rest should be strictly maintained. On the acute phase reactants have settled, modest restriction of exercise activity is recommended for 2 weeks only. Measurement of phase reactants should be, however, continued with 2-weeks intervals for 2 months. With this regimen a rebound is unlikely in patients with arthritis alone, but may occur in patients with carditis, rarely requiring a further period of rest. Anti-inflammatory drugs Treatment with anti-inflammatory drugs has naturally received intensive study. Joint pains and fever usually settle rapidly with aspirin or steroids. It is questionable, however, whether either drug has any effect on the duration of the illness or the extent of valve damage after the illness. One must conclude that, if there is any long-term benefit from the use of aspirin or corticosteroids, it is so small as to be unmeasurable. Despite this, steroid administration is often given to patients with moderate carditis, covering the withdrawal with aspirin because of the rather common rebound in symptoms. Such policies seem to reflect the desire of doctors to administer drugs rather than to allow nature to effect a cure. Aspirin should be given to control pain, the dosage tailored according to need. Prednisone may also be given for this purpose but can usually be reserved for the situation where aggressive carditis appears lifethreatening. Steroids sometimes appear to help to arrest progressive heart failure. The required dose of prednisone is usually not more than 40 to 60mg daily, and gradual reduction in dosage can usually start within 1 week. Three points should be noted: 1) treatment should not be started until the diagnosis is secure; 2) when there is evidence of deteriorating cardiac output, dilation of the heart, and congestive heart failure, a severe valve lesion is usually present and valve replacement may give the only chance of survival; 3) aspirin and prednisone depress acute phase reactants. The bout cannot be considered settled until the erythrocyte sedimentation rate and C-reactive protein levels have been normal in successive weeks, the first measurement being taken 2 weeks after stopping suppressive treatment. 1 Antibiotic therapy: benzathine penicillin (Penidure LA) 1.2 megaunits IM for the eradication of infection, then bicilline 1 500 000 and every 20- 21st day during 14 hospitalization. Erythromycin (or benzathine penicillin G) 250mg 6 hourly can be used in penicillin allergic patients. 2 Anti-inflamatooty therapy: a) corticosteroids (pridnisolone 20-30mg/d -10-14 days; then decrease dose of 2,5 mg/d every 3-4 days to 10 mg/d , then the decrease to 2,5 mg every week; b) salicylates (aspirin) markedly reduce fever and relieve joints pain and swelling. Adults may require 0.6-0.9g (2-3tabs of aspirin) 4 hourly. It can be increased to 2g 4 hourly. Usually 4-6 weeks treatment is required, then it should be tapered and should not be stopped abruptly otherwise rebound can occur. Chorea Treatment of chorea, other than with penicillin prophylaxis, is rarely required and does not influence on the duration of symptoms. Bed rest is required only when chorea appears early and phase reactants are still positive, or in the rare case of extreme motor disturbance. For troublesome symptoms phenobarbitone, diazepam, or haloperidol are useful. 1.18. Recurrence of rheumatic fever and prophylaxis Recurrence of rheumatic fever In preantibiotic days recurrences of rheumatic fever were recorded up to 70 per cent of patients. It was subsequently shown that a further streptococcal infection, often with a different strain, preceded each recurrence. The “rheumatic” patient retains the tendency to develop rheumatic fever with further group A streptococcal infections. The risk of recurrence is the highest in the first 3 years after the first attack, in young patients, and in patients with rheumatic heart disease. Carditis with a recurrence is more common in those patients in whom it was present in the first attack but it may occur in any patient. Recurrent attacks frequently lead to progressive deterioration in valvular and myocardial function. The need for meticulous prophylaxis in all patients is clear. The base of treatment is systems of staging therapy: Hospital- outpatient department- health resort. I stage: In hospital realize the active anti-inflammatory therapy, spend out the penicillin prophylaxis and rehabilitation, checking physical capacity to work of the patient are realized. II stage: Direction in specialized rheumatic resort - a continuation of the treatment by anti-inflammatory drugs (aspirin), binicillin 5, continuation of rehabilitation. III stage: regular medical check-up. 1. Realization of medical actions, directed on final liquidation of the active rheumatic process. 2. Undertaking symptomatic therapy circulation disorder in patients with valvular heart disease; decision the questions of surgical treatment of heart valvular disease. 3. Decision question of rehabilitations, ability to work, job placement. 4. Realization of the primary prevention of the rheumatic fever and secondary prevention relapse of diseases. 15 Prevention Fortunately the group A streptococcus remains uniquely sensitive to long-term, low-dose penicillin, which may be administered orally or parenterally. Regrettably, non-compliance can reach astonishing levels with oral prophylaxis. In the 1960s Gordis showed that 90 per cent of patients became non-compliant if they had four or more of the following characteristics: female, adolescent, large sibship, not admitted to the hospital with acute attack, without activity restriction, or unaccompanied to clinic by parents. With rare exceptions treatment with benzathine penicillin is recommended. The injection is sometimes painful for 1 to 2 days; but, this may be ceased somewhat by incorporating a low dose of local anaesthetic or procaine penicillin with the injection. The minimal treatment period for patients without carditis is often given as 5 years, or to 18 years of age, but recurrences beyond 5 years are by no means uncommon. Annual recurrence rates above 5 per cent were recorded in years 10 to 15 in the prepenicillin era. A minimal treatment period of 10 years is desirable. For patients with established heart disease more prolonged prophylaxis is mandatory and treatment is recommended at least to the age of 45 years. Pressure may rise change by oral prophylaxis after some years but this is recommended only in particularly reliable patient. Primary prevention Organization of the complex individual, public and national measures, directed on liquidation of the primary disease by rheumatic fever: - Advocacy of hardening; - development of the physical culture and sport; - fight with congestion in house, school, kindergartens, public institutions; - undertaking broad individual, public sanitary-hygenic action, reducing possibility streptococcal of infection of populations, at first place children; Primary prevention of ARF consists of accurate diagnosis and appropriate treatment of streptococcal sore throat. Secondary prevention This strategy focuses on the group of persons who have already suffered a rheumatic attack and who are inordinately susceptible to a following recurrence and innunologically significant streptococcal upper respiratory infection. - Benzathine penicillin (Penidure LA) 1.2 megaunits IM every 4 weeks until the age of 25 or 5 years after the last attack. - Sulphonamide or erythromycin: if patient is allergic to penicillin, sulphadiazine 1g daily or erythromycin 250mg orally twice daily may be substituted. 1.19. Prognosis: - Initial episodes of rheumatic fever may last months in children and weeks in adults. - The immediate mortality rate is 1-2%. Persistent rheumatic carditis with cardiomegaly, heart failure and pericarditis imply a poor prognosis. 16 - After 10 years, 2/3 of patients will have detectable valve disease. Mitral regurgitation is more common in adults and aortic regurgitation in children. 1.20. REFERENCES 1. Bisno A.L. The resurgence of acute rheumatic fever in the United States. //Annual Review of Medicine. – 1990. - № 41. – Р.319-29. 2. Combined Rheumatic Fever Study Group. A comparison of short-term, intensive prednisone and acetylsalicylic acid therapy in the treatment of acute rheumatic fever. // New England Journal of Medicine. – 1965. – Vol. 272. – Р.63-70. Feinstein A.R., Spagnuolo M., Gill, F.A. Yale Journal of Biological Medicine. – 1961. - № 33. – Р.259-278. 3. The genetics of rheumatic fever: relationship to streptococcal infection and autoimmune disease. / Gibofsky A., Khanna A., Suh E., Zabriskie J.B. // Journal of Rheumatology. – 1991. August supplement. – Р.1-5. 4. Gordis L., Markowitz M., Lilienfeld A.M. Why patients don't follow medical advice: a study of children on long-term antistreptococcal prophylaxis.// Journal of Pediatrics. – 1969. - № 75. – Р.957-958. 5. Markowitz M., Gordis L. Rheumatic fever, (2nd edn). - Saunders, Philadelphia, 1972. 6. UK and US Joint Report. The evolution of rheumatic heart disease in children: five year report of a cooperative clinical trial of ACTH, cortisone and aspirin. // Circulation. – 1960. - № 22. – Р.503-515. 7. Resurgence of acute rheumatic fever in the intermountain area of the United States. / Veasy L.G., Wiedmeier S.E., Orsmond G.S., Ruttenbert H.D., Boucek M.M., Roth S.J., Tait V.F., Thompson J.A., Daly J.A., Kaplan E.L., Hill H.R. // New England Journal of Medicine. – 1987. - Vol.316. – Р.421-427. 8. Nasonova V. A., Kuzmina N. N., Belov B. S. Medicine. – 2004. - №8. – Р. 44. 2. VALVULAR HEART DISEASES 2.1. MITRAL VALVE DISEASE 2.1.1. Introduction The recent year valvular heart diseases were one of the often reason for invalidization and mortality of young patients so recognizing a heart murmur on physical examination is the usual means of initially diagnosing of valvular heart disease. Thus, the clinical examination remains important for detecting valvular heat disease, recognizing cardiac deterioration, and assessing follow-up status. The patient with valvular heat disease often recalls a history of a heat murmur, and therefore the first recognition of the murmur may be helpful in establishing the etiology. The hear murmur detected in early adulthood often suggests a 17 conferential or rheumatic basis, whereas a murmur developed in later years in often due to the degenerative changes in valvular structure. The physician should carefully assess the patient’s physical activities and note the initial onset of noninvasive and invasive cardiac studies as well as the decision for surgical correction. The noninvasive technologies of electrocardiography, chest radiography, echocardiography, radionuclide angiography, and stress testing play an important role in assessing the impact of valvular heart disease on cardiac function and determining the timing of operative intervention. In recent years advances in echocardiography have resulted in more accurate assessment of valvular orifice size, and catheterization in reserved to confirm impressions and identify underlying coronary artery disease. But the using these methods can be limited by condition of the patient, as well as that complex modern equipment are equipped only large cardiologic hospitals. In this connection, the careful physical examination of the patients in combination with use the simple instrumental methods (in the first place x-ray and electrocardiography) must give the maximum of information. The often reasons of the wrong diagnostics of valvular heart disease are following: 1) lack of knowledge about frequency something or other valvular heart disease; 2) belief that the clinical picture valvular heart disease is stable, meantime as it is greatly changed on different stages of the disease; 3) defect of the objective patients’ examination when important diagnostic signs are not revealed; 4) wrong interpretation of revealed signs; 5) prevalence in clinical picture of complications of valvular heart disease, which are estimated as symptoms of other diseases; 6) combination of the valvular heart disease with the other diseases, leaving on the first plan in the general clinical picture. For avoid these mistakes, its necessary two conditions. First, perfect knowledge of the semiotics of valvular heart disease and secondly, clear pathophysiological analysis of revealed symptoms and correct their assessment. 2.1.2. Definition Valve disease causes abnormalities in blood flow across the cardiac valves. Normal valves demonstrate two critical flow characteristics: unidirectional flow and unimpeded flow A diseased valve can produce two types of functional derangements: 1) valve regurgitation – the valve leaflets fail to close securely, permitting backward flow (valvular insufficiency and valvular incompetence are synonymous term); 2) valve stenosis- the valve orifice becomes restricted, impeding forward. Regurgitation and stenosis can occur together in the same valve as a “mixed lesion” or either one can occur alone as a “pure lesion”. 2.1.3. Normal mitral valve Anatomy 18 The normal mitral valve is a complex structure, consisting of leaflets, annulus, chordae tendineae, and papillary muscles. Its anatomy as studied at autopsy shows an unusual degree of variation between normal subjects. Of the two leaflets, the anterior one is the larger, both from base to margin, and also in its perimeter. It is attached to the root of the aorta and the membranous septum at the base of the heart, and is continuous with the chordae peripherally. It thus passes across the centre of the left ventricular cavity, dividing the inlet from the outlet portion. The posterior cusp is attached to the mitral ring and to the anterior cusp at both commissures. It is continuous with the posterior wall of the left atrium, and is divided into three portions by two scallops. The chordae arise from the ventricular margins of both cusps, and are inserted into the heads of the papillary muscles. There are multiple subdivisions in the chordae as they pass from papillary muscles to the cusps, which form an effective secondary pathway, additional to the main one between the cusps, for blood to enter the ventricle. There are two papillary muscles, one anteromedial and the other posterolateral. In general, the former is larger, and has a more uniform structure than the latter which may be double. Both may have up to six heads, giving rise to chordae. The papillary muscles are continuous with the trabecular and subendocardial layer of the ventricular wall. Both are supplied by a single end-artery. The mitral ring is an insubstantial structure whose function is to support valve cusps only. It is incomplete in the region of aortic root and the membranous septum. However, it is surrounded by a well developed circumferential ring of myocardium which supports it, and whose contraction during systole has the effect of significantly reducing the diameter of the valve orifice. Histologically, the normal valve cusp has a dense collagenous core, continuous with the valve ring, the valve fibrosa. This is covered on atrial and ventricular surfaces by a thin layer of loose connective tissue, and finally by endocardium. Physiology The normal mitral valve has a cross-sectional area of approximately 5 cm. This allows ventricular filling to occur with a peak rate of 500 to 1000ml/ s, with only a very small pressure drop across it. Left ventricular filling occurs mainly during early diastole, the rapid filling phase, and left atrial systole. During middiastole (diastasis), ventricular volume remains virtually constant, and the mitral valve itself almost closes. As heart rate increases during exercise, diastasis becomes shorter, while the duration of the rapid filling period remains virtually unchanged. At rest, approximately two-thirds of the stroke volume enters during early diastole and the remaining one-third during left atrial systole. With rapid hearts rates, occurring at peak exercise, left ventricular filling time in normal subjects may fall to less than 100ms. If stroke volume is taken as 100 ml, mean filling rate is of the order of 1l/s, which is achieved with only a very small pressure difference between left atrium and left ventricle. This gives some indication of the effectiveness of normal mechanisms underlying left ventricular filling. 19 2.1.4. Mitral valve disease Mitral valve heart diseases are mitral regurgitation (insufficiency) and mitral stenosis. Mitral regurgitation is a result of incomplete valve closure, and mitral stenosis is the decreased orifice of mitral valve. As a result hemodynamic abnormalities and decreased myocardium function and leads to congestive heart failure. 2.1.5. Epidemiology The mitral valvular disease met the most often (50-70% according to different authors) an aortic valvular disease is less the8-27%. The tricuspid valvular disease meet not more often than in 1% cases, however the combinations with other valvular defeat is noted approximately about in a half of the patients. 2.1.6. Classification of valve heart disease, (MUHARLAMOV 1977) Mitral valve disease 1. Mitral regurgitation 2. Mitral stenosis 3. Mitral valvular heart disease (compound) with prevalence of mitral regurgitation 4. Mitral valvular heart disease (compound) with prevalence of mitral stenosis 5. Mitral valvular heart disease (compound) without accurate prevalence of mitral regurgitation or mitral stenosis Compound valvular disease is a mixed lesion consisting of stenosis and regurgitation in the same valve, it occurs frequently. This is to be expected because a stenotic, immobile valve is often unable to close completely. Combined valvular disease, or multivalvular disease, is often seen because the rheumatic heart diseases typically affect different heart valves. Classification of mitral valve disease (Bakylev 1955) Depending on disorders of pulmonary and systemic circulation 5 stages distinguished: Stage I – there is no dyspnea neither at a rest nor on exertion; Stage II- abnormalities of pulmonary circulation on exertion (dyspnea on exertion); Stage III-significant pulmonary circulation disorders (hypertension) and initial systemic circulation disorder (ankle edema); Stage IV- Significant pulmonary and systemic circulation disorders (dyspnea on rest, significant edema to anasarca); Stage V- cardiac dystrophy. 2.2. Mitral stenosis 2.2.1. Etiology Chronic rheumatic heart disease is by far the most common cause of mitral stenosis, though a number of other well defined pathological processes exist. Mitral stenosis may be congenital, when it is frequently associated with other lesions causing obstruction of left ventricular outflow, including aortic or subaortic stenosis or coarctation of the aorta. In such cases, the chordae are usually short, and the spaces between them obliterated. The leaflets are thick, with rolled edges. The insertion of the papillary muscles may be abnormal, being either directly from 20 the free wall of the ventricle or from the septum. In parachute mitral valve, there is only a single papillary muscle. Congenital mitral stenosis may be associated with hypoplasia of the left ventricular cavity and the aorta, and also with endocardial fibroelastosis. Other causes besides rheumatic acquired mitral stenosis are rare. It has been reported in occasional patients with calcified mitral valve ring, in infective endocarditis, when bulky vegetations may cause obstruction to flow or when granulomatous infiltration has occurred in association with eosinophilia. In nodular rheumatoid arthritis, thickening of the valve cusps has been observed, but true mitral stenosis does not result. However, in systemic lupus erythematosus, treatment of Libman-Sachs endocarditis with steroids may lead to fibrosis of the cusps with commisural fusion. The combination of ostium secundum atrial septal defect and mitral stenosis, Lutembacher's syndrome, is probably fortuitous. 2.2.2. Pathogenic and hemodynamic abnormalities The normal cross-sectional area of the mtirla valve ranges from4 to 6 sq cm. The hemodynamic abnormalities in mitral stenosis (Figure 1) result from obstructed diastolic blood flow into the left ventricle during ventricular diastole, and turbulence of diastolic blood flow occurs when the valvular orifice is reduced below 2 sq cm. Figure 1. Pathological haemodinamics of mitral stenosis. 21 Therefore the pressure difference, or pressure gradient, between the chambers rises; normally the pressure gradient is minimal. The left atrial musculature hypertrophies increase its pumping force. The active contribution of atrial contraction to ventricular filling becomes increasingly important. The primary function of the left atrium ceases to be that of passive reservoir and conduit for blood flowing to the ventricle. Atrial dilation occurs when the left atrial volume rises, because of eh inability of the chamber to empty normaly. Increased demands for cardiac output, such as exercise or fever, may be necessary to produce the diastolic murmur with the mitral vavle orifice is as small as 1,5 to 2 sq cm.. In the second stage of progressive reduction in the mitral orifice size, a diastolic gradient develops between the left atrium and left ventricle under resting conditions when the valvular area is 1 to 1,5 sq cm. In the advanced stage, mitral orifice size is < 1sq cm, and left atrial and pulmonary hypertension becomes significant ( about 20 to 25 mmHg) and this leads to significant pulmonary arterial hypertension, pressure overload on the right ventricle, and compensatory hypertrophy of the right ventricle. 2.2.3. Pathophysiology of mitral stenosis 1 left atrial hypertrophy; 2 left atrial dilation; 3 pulmonary venous congestion; 4 pulmonary congestion; 5 pulmonary hypertension; 6 right ventricle hypertrophy; 7 fixed cardiac outpu 2.2.4. Clinical features The clinical picture may be deferent depending on the underlying hemodynamics; however the earliest symptom is usually dyspnea on exertion.Two hemodinamic changes associated with exertion are poorly tolerated in mitral stenosis: tachycardia and elevated left atrial pressure. As the disease progresses, respiratory symptoms become more pronounced- dry cough or cough with little amount of mucous stupum often with blood-hemoptysis. Paroxysmal nocturnal dyspnea at rest may be noted. On physical examination orthopnea may be. Patients may display typical “mitral facies” with florid congestion of the cheeks. End-stage mitral stenosis is associated with the right heart failure with consequent systemic venous engorgement, distended neck veins, hepatomegaly, peripheral edema, and ascities. Pulsus differens may be determined. Pulse in the left hand is weaker because of compression the subclavicular vein by the dilated right atrium. Inspecting the precordium may reveal activity along the left sternal border, indicating right ventricular enlargement and pulmonary hypertension. On palpation diastolic rumble can sometimes be felt at the apex. With the significant right ventricular dilatation, the left ventricular apical impulse may be displaced later. 22 On percussion with significant right ventricular dilatation right cardiac border may be percussed to the right of the sternum and displaced of the upper cardiac boarder to up due to the dilation and hypertrophy of the left atrium. On auscultation direct findings of mitral stenosis are determined: 1) accentuated first heart sound (AV valve closure) at the apex; 2) third heart sound (opening snap resulting from the loss of leaflet pliability) at the apex; 3) low-frequency diastolic murmur ( rumble) at the apes; 4) accentuated second heat sound at the pulmonary artery. As because the murmur of mitral stenosis may be faint in the early stages, to complete the physical examination, a complete examination should include mild exercise to increase heart reate and intensify the diastolic rumble. 2.2.5. Investigations Electrocardiography The electrocardiographic changes of mitral stenosis (Figure 2) include the left artial enlargement and the right ventricular hypertrophy due to pulmonary hypertension: 1) notching and prolonged P wave in leads II, III avf, V 4-6; 2) the terminal portion of the P wave is usually negative in lead V 1; 3) right-axis deviation; 4) increased amplitude of the R wave in V1. Figure 2. The electrocardiographic changes of mitral stenosis. 23 Chest radiograph shows mitral configuration of the heart: 1) left atrium enlargement; 2) pulmonary venous hypertension; 3) right ventricle prominence. Echocardiograhy Echocardiography has totally changed the diagnosis and management of patients with mitral valve disease. The characteristic feature of rheumatic mitral valve disease on M-mode echocardiography is a reduced middiastolic closure rate of the anterior cusp of the mitral valve to less than 50mm/s for mild mitral stenosis and to 0 to 20mm/s for severe involvement. Cusp fusion causes forward, rather than backward movement of the posterior cusp during diastole. On cross-sectional echocardiography, the mobility of the anterior cusp is reduced, particularly near its tip. Valve area can be estimated semiquantitatively from the parasternal minor axis view, provided that the cusps are not calcified. It is also possible to assess the degree of subvalve involvement. Occasionally atrial thrombus can be detected. The diastolic pressure drop can be estimated by cross continuous wave Doppler, and valve area estimated from the pressure half time. Peak right ventricular pressure is estimated from systolic velocities of tricuspid regurgitation. The aortic and tricuspid valves can also be checked. Cardiac catheterization This investigation is rarely necessary, either to make the diagnosis or to assess its severity. It is performed only to assess the state of the coronary arteries in older patients and as a prelude to balloon valvuloplasty, or in very occasional cases in which diagnostic echocardiograms cannot be obtained. 2.2.6. Diagnosis The diagnosis of mitral stenosis is usually straightforward on the basis of history, physical signs, and chest radiograph, and can be confirmed rapidly by echocardiography. When the ventricular rate is rapid, the diastolic murmur may be inaudible, but becomes apparent when the rate is controlled by digoxin. Cases of silent mitral stenosis may present difficulties, and may mimic primary pulmonary hypertension. In such patients, mitral stenosis can often be excluded or confirmed only by echocardiography or direct measurement of left ventricular and left atrial pressures. Mitral stenosis should also be suspected as a source of systemic emboli and as a cause of unexplained atrial fibrillation, particularly in the elderly. In these circumstances, the valve lesion itself may be very mild, and the usual physical signs not are present, although the characteristic abnormalities can be demonstrated by echocardiography. One should always try to quantify the severity of the mitral stenosis as well as diagnosing its presence. Probably the most reliable way to do this is still from the extent to which exercise tolerance is reduced, as judged from the history. The length of the mid-diastolic murmur gives some indication, as 24 described above, although this is unsatisfactory when the ventricular rate is increased or the murmur inaudible. 2.2.7. Management Medical treatment 1. In patients below the age of 21, penicillin prophylaxis against further attacks of acute rheumatic fever should be given. 2. Atrial fibrillation should be treated with a digitalis preparation to control the ventricular rate. Anticoagulant therapy should be given to reduce the risk of systemic embolism to all patients with atrial fibrillation, unless there are very strong contraindications. It is also advisable to consider giving this treatment to patients in sinus rhythm with mitral stenosis, particularly the middle-aged and elderly, as the incidence of embolism is not negligible in these patients, particularly at the onset of atrial fibrillation. The incidence of embolism is also high when an unanticoagulated patient with atrial fibrillation, is admitted to hospital with a rapid heart rate and pulmonary oedema. Intravenous heparin should thus be given until adequate anticoagulation with an oral agent has been established. 3. Fluid retention associated with mitral stenosis responds well to treatment with diuretics. 4. Chest infections should be treated promptly with appropriate antimicrobials. Patients should be given a supply of antimicrobial to take prophylactically at the start of a head cold. Chest infections often precipitate, or may be precipitated by fluid retention so that a diuretic is also often useful. 5. In all patients with valvular heart disease, prophylactic antimicrobial should be given for all dental manipulations and potentially septic hazards. This should preferably be amoxycillin, unless the patient is sensitive to penicillin, when cephaloridine or erythromycin should be used. Mitral valvuloplasty So, as rheumatic mitral stenosis results from fusion of the commisures between the two mitral cusps. This fusion is susceptible to rupture by inflating a cathetermounted balloon across the valve orifice. Mitral valvuloplasty has the great advantage over surgery of avoiding thoracotomy. A catheter is introduced through the inferior vena cava to the right atrium. The atrial septum is crossed, and the catheter stabilized across the mitral valve, usually by a guidewire, which is passed out through the aortic valve. The balloon itself may be single, often with a waist; less commonly, two balloons are placed simultaneously across the orifice. The balloon is inflated to a predetermined size for 20s. Not all patients are suitable for valvuloplasty. There should be no more than minimal regurgitation. Ideally, the cusps should be pliable, with calcification in the commissures. The subvalve 25 apparatus should not be scarred or contracted, and clot in the left atrial appendix should have been excluded by transoesophageal echocardiography. Using this procedure, a satisfactory fall in transmitral pressure difference is usually achieved, and maintained in the short and medium term. Embolism is unusual provided that clot in the left atrium has been excluded. Mitral regurgitation may be provoked, sometimes severe enough to require valve replacement on an elective or even an emergency basis. The majority of patients are left with a small atrial septal defect at the site of passage of the catheter, but this is not of any haemodynamic significance. Mitral surgery The decision to operate is based on symptoms of pulmonary congestion developing during activity or at rest. In addition to pulmonary congestion, recurrent atrial fibrillation with aggravation of pulmonary congestion, thromboembolic pneumonia, and hemoptysis can be indication for surgery, Mitral comissurotomy remains the procedure of choice with a pliable mitral vavle without calcification of mitral regurgitation and carries an operative mortality of < 1 %. A number of surgical procedures are available. These include mitral valvotomy, open or closed, and mitral valve replacement. The choice of operation depends on the anatomy of the mitral valve determined on the basis of the physical signs, the echocardiogram, the age of the patient, and the surgical resources available. Closed mitral valvotomy is a relatively simple procedure in terms of the resources that are required, although a satisfactory result presupposes considerable experience with the operation, experience that is now becoming rare in the developed world. This closed operation is particularly suitable in a Third World country, where the major radiographic and expensive disposables necessary for balloon valvuloplasty are not available. It is particularly appropriate in a young patient, in sinus rhythm, with evidence of a mobile anterior cusp. Symptom-free follow-up of 30 years or more occurs regularly after this procedure. Open valvotomy requires cardiopulmonary bypass but allows a more complete procedure to be undertaken and, in particular, the subvalvular apparatus can be inspected and adherent chordae divided. If the results of valvotomy are found to be unsatisfactory, then it is possible to proceed to valve replacement at the same operation. Mitral valve replacement will be required if the valve cusps are greatly thickened or calcified. This operation should not be considered in patients in whom the haemodynamic disturbance caused by the valve disease is mild, as the prosthesis causes a resting diastolic pressure drop across it, as well as interfering with systolic and diastolic left ventricular function. This is particularly the case if the left ventricular cavity is small, when a low profile prosthesis is preferred. 26 2.2.8. Prognosis In the absence of surgical treatment, mitral stenosis is usually a progressive disease, although the rate is unpredictable. Unfavourable features include a gradual increase in the severity of the valve disease with disorganization of its structure and superimposed calcification, an increase in pulmonary vascular resistance, and the development of functional tricuspid valve disease, with chronic elevation of the venous pressure leading to cardiac cirrhosis and impaired liver function. Surgical treatment has improved the prognosis considerably, although mitral valvotomy does not prevent progression of the rheumatic process, nor does it reduce the risk of infective endocarditis. It is still premature to assess the long-term prognosis of patients who have been treated by valvuloplasty. That of closed mitral valvotomy, which has been available for over 40 years, can be remarkably satisfactory, and it is not unusual to see a patient several decades after surgery still with effectively normal flow velocities across the mitral valve. It has also become clear that the life of biological mitral valve substitutes, particularly the porcine xenograft, is limited to no more than 10 years in the majority of patients above the age of 21, and considerably less than this in children. The use of valves should thus be confined to the very elderly, and to young women who wish to undertake pregnancy knowing that repeat surgery will be needed. There are minor differences in the haemodynamic effects of the different types of mechanical valve substitutes, but in individual cases, these are of little consequence. 2.3. Mitral regurgitation 2.3.1. Etiology Unlike mitral stenosis, which is almost invariably due to chronic rheumatic heart disease, there are a number of causes of pure mitral regurgitation. The most common of these is the floppy mitral valve. This condition has been described under a number of names, based either on its pathology or on its clinical features. Thus it has been referred to as mucinous or myxomatous degeneration, or as a ballooning or billowing mitral valve. As described below, it is probably responsible for a significant proportion of patients described as having mitral valve prolapse, or the midsystolic click late systolic murmur syndrome. Floppy mitral valve, which is relatively common above the age of 50, is a non-inflammatory process which may affect either cusp, partially or completely. The most striking abnormality is an increase in cusp area, causing folding and upward doming into the left atrium during systole. The chordae may become elongated, tortuous, and thinned, predisposing to chordal rupture. The abnormal chordae can undergo fibrosis, as can the cusps, leading to an erroneous diagnosis of chronic rheumatic involvement. Ulceration of the cusps may also occur, predisposing to thrombosis on their surface, and also to infective endocarditis. The ring circumference may be normal or increased. The papillary muscles are normal. Histologically, the central valve fibrosa is abnormal with large areas in which fibrous tissue is either absent 27 altogether, or where the collagen bundles are fragmented, coiled, or disrupted. These lie in pools of abnormal acid mucopolysaccharide. A dense layer of laminated collagen forms over the atrial surface of the cusp. There is no evidence of vascularization or of inflammatory cells in the absence of secondary infective endocarditis. The cause of sporadic cases of floppy mitral valve is unknown. However, similar appearances may complicate Marfan's syndrome, pseudoxanthoma elasticum, Ehlers-Danos syndrome, and osteogenesis imperfecta. The incidence of the sporadic condition tends to rise with age, and individual case histories suggest that it can be a very benign and chronic process. Infective endocarditis is an important cause of mitral regurgitation, which may affect the valve directly or be secondary to an infection on the aortic valve. Vegetations developing on the cusps vary from small nodules along the line of apposition to large, friable masses up to 10mm or more in diameter, which are especially common when then organism, is a fungus. Lesions on the anterior cusp of the mitral valve may occur in association with aortic endocarditis, usually involving the right coronary cusp. These “jet lesions” may appear as localized aneurysms, or perforate the cusp causing mitral regurgitation severe enough to need valve replacement. Rarely, mitral vegetations may be large enough to cause mitral valve obstruction: much more commonly, the haemodynamic disturbance is of pure regurgitation. Infective endocarditis may also involve the chordae, particularly in patients with floppy mitral valve. It may occur on an otherwise normal valve, particularly in the old or debilitated, but more commonly the valve is abnormal due to minor congenital abnormality, previous rheumatic involvement, floppy mitral valve, hypertrophic cardiomyopathy, or calcification of the mitral valve ring. Mitral prostheses or xenografts are also liable to be infected by endocarditis, which either may be blood borne, or introduced at the time of operation. 2.3.2. Pathogenic and hemodynamic abnormalities Mitral regurgitation permits retrograde blood flow from the left ventricle to the left atrium and a result of incomplete valve closure (Figure 3). During systole the ventricle simultaneously ejects blood forward into the into the aorta and back ward into the left atrium. The volume work of both the left ventricle and the left atrium must increase to preserve cardiac output. The left ventricle must pump a sufficient volume of blood to maintain a normal forward flow into; the aorta and the regurgitant flow though the mitral valve. The additional volume load created by the regurgitant valve prompts ventricular dilation. According to Starling’s law of the heart, venricular dilation increases myocardial contractility. Eventually the ventricular wall hypertrophies to further increase contractile force. 28 Figure 3. Mitral regurgitation. In the early stages of chronic mitral regurgitation, the left ventricle is able to compensate for the increased volume load. Eventually, however, the ventricle begins to fail, reducing cardiac out and increasing residual venricular volumes and regurgiatant flow. Regurgitation creates a volume load not only for the left ventricle, but also for the left atrium. The left atrium dilates to accommodate the increased volume and to increase the force of atrial contraction. Subsequently- the atrium hypertrophies. Atrial dilation occurs as the left atrial volume rises, what results in pulmonary hypertension. Gradually the predictable sequence of pulmonary and right heart involvement ensure: 1- pulmonary venous congestion; 2- interstitial edema, 3pulmonary arterial hypertension and 4- right ventricular hypertrophy. Pathophysiology of mitral regurgitation 1. 2. 3. 4. 5. 6. 7. 8. Left ventricular dilation. Left ventricular hypertrophy. Left atrial dilation. Left atrial hypertrophy. Pulmonary venous congestion. Pulmonary congestion. Pulmonary artery hypertension. Right ventricular hypertrophy. 29 2.3.3. Clinical picture When mitral regurgitation results from primary defects in the mitral apparatus, significant enlargement of the left ventricle develops, but the patient may remain symptom- free with normal exercise tolerance. Complains The earliest symptoms of mitral regurgitation are: 1) weakness and fatique cause by the reduction on forward flow; 2) exertional dyspnea; 3) palpitations. As pulmonary venous hypertension and congestion are not early features of mitral regurgitation, fatigue is a more frequent symptom than dyspnea. Atrial fibrillation commonly develops when the left atrial enlarges and further aggravates heart failure. On physical examination. There are no specific findings on general examination, but sometimes patients may display typical “mitral facies” with florid congestion of the cheeks. Inspecting the precordium may the determined by a “cardiac hump” on the left of the stermun, due to the left ventricle enlargement. On palpation the apex impulse is displaced to the left and down. It is diffuse and intensive (hyperdynamic) due to left ventricle hypertrophy. On percussion with significant the left ventricular dilatation left cardiac border may be percussed to the left of the left midclavicularis line, and displaced of the upper cardiac boarder to up due to dilation and hypertrophy of left atrium. On auscultation direct findings of mitral regurgitation are determined: 1. the first heart sound diminished because of not-comlete closure of mitral valve at the apex; 2. apical holosystolic murmur radiates into the axilla and often to the left sternal border because of not-comlete closure of mitral valve; 3. accentuated second heat sound at the pulmonary artery. 2.3.4. Laboratory examination Electrocardiography The electrocardiographic changes of mitral regurgitation (Figure 4) show the evidence of the left atrium and left ventricle hypertrophy: 1) notching and a prolonged P wave in leads II, III avf,V 4-6; 2) the terminal portion of the P wave is usually negative in lead V 1; 3) left-axis deviation; 4) increased QRS voltage and ST-T changes in the V5-V6. 30 Figure 4. ECG changes of mitral regurgitation. Chest radiograph shows mitral configuration of the heart: 1) left ventricle enlargement; 2) left atrium enlargement; 3) pulmonary venous congestion. Echocardiogram On the M-mode, the mitral valve echo may be abnormal, showing prolapse, with cusp remnants visible in the left atrium during systole. Early in the natural history of the condition, the large left ventricular stroke volume is mediated by a small end-systolic volume. As the left ventricle adopts, so the end-diastolic dimension increases. Finally, when irreversible left ventricular disease supervenes, there is a progressive increase at the end-systolic left ventricular cavity size. The left ventricular filling rate may be very rapid indeed. Cross-sectional echocardiography confirms the presence of the very active left ventricular wall motion and allows a clearer view of the extent of systolic cusp prolapse into the left atrium; the affected cusp is also recognized more reliably. Regurgitation can be confirmed by continuous wave Doppler (Figure 9) 1734 and the jet mapped within the left atrium by colour flow. Apparent jet area, whether or not normalized to the left atria cavity size, has proved a disappointing measure of the severity of the regurgitation. Transoesophageal echo may give more information about valve anatomy, allowing small vegetations to be detected. If the regurgitation is severe, retrograde flow can be detected in the pulmonary veins. Cardiac catheterization 31 This is not usually necessary to make the diagnosis when the clinical features and echocardiography are typical, though many surgeons require views of the coronary arteries in older patients. 2.3.5. Diagnosis The diagnosis of the mitral regurgitation is usually straightforward on the basis of the physical signs. These may be atypical when the regurgitation is severe enough to cause the pressures in the left atrium and left ventricle to equalize by endejection, so that the murmur ends prematurely. In very severe cases, it may be absent altogether. Such patients may present with pulmonary oedema of sudden and unexplained onset with a chest radiograph showing a normal sized heart shadow. Echocardiography demonstrates a very active left ventricular wall movement, showing that the poor peripheral blood flow is due to the valvular regurgitation rather than left ventricular disease, and in addition, may show the abnormal mobility of one or both mitral valve cusps. Doppler may be atypical, showing an abbreviated regurgitant flow signal of low velocity. In patients who present with more typical signs, the main diagnostic problem is to decide the relative contributions of the valvular regurgitation and left ventricular disease to the overall clinical state. This may be difficult and even after full investigation; the final decision may not be clear cut. 2.3.6. Management Medical therapy Medical treatment consists of high doses of diuretics and vasodilators if valvular regurgitation is severe. Decortication of the ventricular cavities may be possible surgically, along with replacement of mitral or tricuspid valve. Mild or moderately severe mitral regurgitation is well tolerated and does not require treatment apart from prophylactic antimicrobial for all dental manipulations and potentially septic hazards. Such patients should be followed up at annual intervals, as mitral regurgitation, particularly when due to degenerative disease, may be progressive. When mitral regurgitation is due to papillary muscle dysfunction, treatment is that of the underlying condition, which usually means administering a diuretic and a vasodilator. Mitral regurgitation due to hypertrophic cardiomyopathy does not require specific treatment. Severe mitral regurgitation, which causes significant symptoms in spite of medical treatment, is best managed by mitral valve surgery. This will involve either mitral valve replacement, or in suitable cases, mitral valve repair. After acute chordal rupture, it is often possible to treat the patients medically with rest, diuretics, and vasodilators for 1 to 2 weeks, while the left ventricle enlarges to compensate for the increased volume load. Clinical improvement may be striking, so that surgery becomes a less hazardous procedure than an emergency operation in the acute stage would have 32 been. Very severe mitral regurgitation may require emergency treatment on account of intractible pulmonary oedema or a low output state. Such pulmonary oedema is best treated by intermittent positive pressure respiration. The most effective means of managing a low cardiac output state associated with mitral regurgitation is not to administer drugs with a positive inotropic effect, such as isoprenaline or dobutamine, but rather to use a vasodilator. This reduces the peripheral resistance, and thus increases the volume of blood entering the aorta at the expense of that going back into the left atrium. Sodium nitroprusside, by continuous intravenous infusion, at a dose of 20 to 200mgr/min as a 0.01 per cent solution is the agent most frequently used. This requires the arterial pressure, and preferably pulmonary wedge pressure and cardiac output are measured with frequent intervals in order to control the infusion rate. Vasodilators, with or without intermittent positive pressure respiration may make it possible for the cardiac state to be stabilized long enough to allow the underlying diagnosis to be confirmed and cardiac surgery stabilized long enough to allow the underlying diagnosis to be confirmed and cardiac surgery arranged. Vasodilators are also very effective in patients with papillary muscle dysfunction associated with severe left ventricular disease where their use may lead to a temporary, but nevertheless, useful symptomatic improvement. 2.4. AORTIC VALVE DISEASE 2.4.1. Etiology The combination of aortic stenosis and regurgitation usually results from chronic rheumatic heart disease, but may also be caused by infective endocarditis on a previously stenotic valve. As with mixed mitral valve disease, the additional regurgitation may not be severe enough to constitute a significant volume load on the left ventricle, but nevertheless, increases the systolic pressure drop across the valve during systole. In addition, the increased cavity size which accommodates the larger stroke volume is not usually associated with extreme values of wall thickness sometimes seen in pure aortic stenosis, which reduces diastolic stiffness. 2.4.2. Clinical picture The clinical features of mixed aortic valve disease do not differ significantly from those of pure aortic stenosis, except ahen breathlessness is usually the most prominent symptom. On examination, uncomplicated cases remain in sinus rhythm until late in the disease, and atrial fibrillation suggests the presence of additional rheumatic mitral valve disease. A subgroup of patients, in whom the mitral valve is normal, however, should be recognized in those where left ventricular enddiastolic pressure is greatly raised, and in theses that resemble those with rheumatic mitral valve disease by developing atrial fibrillation, selective left atrial enlargement, and severe pulmonary hypertension. The character of the carotid 33 pulse is modified in mixed aortic valve disease, being bisferiens, a term that describes the presence of a notch half way up the upstroke. As in pure aortic stenosis, left ventricular hypertrophy is shown by a sustained apical impulse, with or without a palpable left atrial contraction. The diagnosis is confirmed by aortic systolic and diastolic murmurs, maximal down the left sternal edge. If the patient is in atrial fibrillation, evidence of additional rheumatic mitral valve disease should be sought, particularly a palpable first heart sound, an opening snap, and a middiastolic murmur. Chest radiography shows moderate cardiac enlargement and, in older patients, evidence of aortic valve calcification. Selective enlargement of the left atrium suggests the presence of additional mitral valve disease. Electrocardiography confirms left ventricular hypertrophy. Echocardiography can be used to measure left ventricular cavity size and thus to gain some idea of the severity of the regurgitation from the stroke volume. The technique can also be used to confirm or exclude the presence of rheumatic mitral valve disease. The pressure drop across the aortic valve is estimated by continuous wave Doppler, and colour flow mapping can give some idea of the extent of regurgitation. 2.4.3. Diagnosis and management The main differential diagnosis is from pure aortic stenosis, or regurgitation, though it is not very useful to try and assess the relative importance of stenosis and regurgitation. The indications for surgery are similar to those for pure stenosis or regurgitation. 2.4.4. Classification Aortic valve disease 1. Aortic regurgitation. 2. Aortic stenosis. 3. Aortic valvular heart disease (compound) with prevalence of aortic regurgitation. 4. Aortic valvular heart disease (compound) with prevalence of aortic stenosis. 5. Aortic valvular heart disease (compound) without accurate prevalence of aortic regurgitation or aortic stenosis. Aortic valvular heart diseases are aortic regurgitation and aortic stenosis. 2.5. AORTIC STENOSIS Valvar aortic stenosis is an important cause of cardiac disability and, though it is most common in the elderly, it may present at any time of life. Congenital aortic stenosis, due to a valve with only a single commissure is most frequent in infancy 34 or childhood. A much more common abnormality, the congenital bicuspid valve, consisting of fusion of one of the three commissures, may be detected as an incidental finding early in life, but does not usually give rise to significant haemodynamic abnormality unless it becomes calcified or involved by infective endocarditis. Rheumatic aortic stenosis develops as the result of commissural fusion in a tricuspid valve and may subsequently become calcified. Senile or degenerative aortic stenosis results from deposition of calcium in a tricuspid valve in the absence of any inflammatory process. Very rarely, vegetations in infective endocarditis, or lipid deposits occurring in hyperlipidaemia may be bulky enough to cause significant left ventricular outflow tract obstruction. 2.5.1. Etiology Aortic stenosis can result from a congenital abnormality, rheumatic fever, or degeneration with calcification in the aging patient. A bicuspid valve is the most common congenital abnormality, and invariably a raphe in one of the cusps indicates failure of the commisure to develop. Rarely, an unicuspid valve can be present at birth. Although the bicuspid valve may not be initially stenotic, fibrosis and thickening lead eventually to a reduced orifice size with calcification. Rheumatic fever scars the leaflet margins, and eventually the commissures fuse and calcify. In the aging patient with degeneration aortic stenosis, calcium deposits usually develop in sinuses and annulus, whereas the margins of the leaflets remain free. 2.5.2. Pathogenic and hemodynamic abnormalities Aortic stenosis (Figure 5) obstructs blood flow from the left ventricle into the aorta during ventricular systole. As the resistance to ventricular ejection increases, the pressure work of the left ventricle raises. Systolic hypertension in the ventricular chamber is compensated by concentric hypertrophy of the myocardial mass (the left ventricle normally generates a systolic pressure of 120 mmHg, can develop pressure up to approximately 300 mmHg during ventricular contraction). As myocardial failure develops from depression of the contractile state, dilation of the ventricle occurs. Myocardial oxygen consumption remains high owing to elevation of the pressure with in the left ventricle and increase in the left ventricular mass. Thus, significant aortic stenosis (critical aortic stenosis corresponds to a reduction in valvular orifice from 3 to 4 sq cm), creates conditions in which high myocardial oxygen demands are inadequately supported by reduced oxygen supply, which leads to subendocardial ischemia. Eventually, with a decline in inotropic state of the myocardium, the ventricle dilates and the ejection fraction is decreased below the normal range. Further elevation of the left ventricular enddiastolic pressure results in pulmonary venous hypertension. 2.5.3. Clinical features 35 The increased myocardial oxygen demands in aortic stenosis with the underperfused subendocaridial myocardium can produce arrhythmias, chest pain, and even sudden death. Chest pain, syncope, and heart failure and the characteristic symptoms of aortic stenosis, even though a gradient across the valve can exist for years before the patient develops symptoms. Complains 1. Angina is produced by an imbalance in myocardial oxygensupply and demand. 2. Syncope occurs primary with exertion, asn a result of either dysrhythmias or and inability the cardiac output sufficiently to maintain cerebral perfusion. Survival after the onset of syncope has been estimated at 3 to 4 years. 3. Left ventricle failure impairs ventricular emptying. Cardiac output falls, and vermicular volumes rise. Ventricular dilation occasionally associated with functional mitral regurgitation. Right ventricle failure and systemic congestion are indicated of end- stage disease. On physical examination: the peripheral pulse is diminished in amplitude, delayed in upstroke, and prolonged owing to sustained ejection across the aortic valve (pulsus parvus et tardus). Inspecting the precordium Because the contour of the heart does not become enlarged with concentric hypertrophy, abnormalities may not be visible of chest examination. 36 Figure 5. Aortic stenosis. On palpation: palpable systolic vibration over the primary aortic area, with the patient in the sitting position during full expiration, often correlate with a gradient across the aortic valve of >40mm Hg. When the left ventricle dilates owing to myocardial failure, the impulse is displaced laterally and becomes more diffuse. On percussion with significant left ventricular dilatation the left cardiac border may be percussed to the left of the left midclavicularis line. On auscultation direct findings of aortic stenosis are determined: 1) the first heart sound diminished because of functional mitral regurgitation at the apex; 2) aoritic second sound becomes diminished, mechanically or electrically prolonged left ventiricular systole can creat reverse splitting of S2; 3) diamond-shaped crescendo-decrescendo systolic murmur on the aorta. The murmur is most intensive over the aortic area and disturb to neck vessels and the place between the scapulae; 4) ejection click. 2.5.4. Laboratory studies Electrocardiogram Left ventricular hypertrophy is the most common finding on the electrocardiogram: 1. Increase in QRS amplitude and ST-T changes of a pattern. 2. Left-axis deviation as a result conduction disturbances and left bundle branch block. Chest radiograph 1. Poststenotic dilation and prominence of the ascending aorta may be present. 2. Heart failure enlarges the left ventricle ( aortic configuration). 3. Pulmonary congestion. Heart size is normal in uncomplicated aortic stenosis. If it is increased, the underlying cause is likely to be unsuspected aortic regurgitation, left ventricular cavity dilatation, or very severe left ventricular hypertrophy, when the cavity may be normal in size, but the myocardium up to 50mm in thickness. Increased left ventricular filling pressure may cause left atrial hypertension and thus dilatation of the upper lobe vessels as well as selective enlargement of the left atrium in the absence of organic mitral valve disease. The aortic root is nearly always dilated and the aortic valve a calcified in older patients, which is best seen on the lateral chest radiograph or with screening. Echocardiogram 1. Thickening of the aortic leaflets. 2. Calcification of the valves. 3. Dilation of the left ventricle. If the aortic valve is calcified, disruption of the normal anatomy can be demonstrated by M-mode, but in younger patients, it may appear entirely normal, even when severe aortic stenosis is present. Two-dimensional echocardiography is 37 very useful for demonstrating abnormalities in the left ventricular outflow tract. Thickening and reduced mobility of the valve cups can nearly always be demonstrated. In young patients with a bicuspid valve, doming of the cusps during systole can be seen, while in older patients, a calcified aortic valve appears as an immobile mass. The pressure drop across the outflow tract can be reliably measured by continuous wave Doppler. Additional aortic regurgitation can also be detected. Left ventricular anatomy and function, both in terms of the extent of hypertrophy and cavity size and ejection fraction can be studied. The hypertrophy itself may be concentric, or may involve the septum to a much greater extent than the posterior wall, resembling the pattern seen in hypertrophic cardiomyopathy. Late in the disease, the left ventricle becomes enlarged and its ejection fraction falls to values commonly seen in dilated cardiomyopathy. Cardiac catheterization The abnormal haemodynamics of aortic stenosis and associated left ventricular disease can usually be completely demonstrated by echocardiography. The role of cardiac catheterization is thus to confirm the pressure drop across the valve in the minority of cases in whom this is not possible for technical reasons using continuous wave Doppler, and to display coronary artery anatomy. 2.5.5. Diagnosis A complete diagnosis of aortic stenosis depends not only on establishing the anatomical abnormality but also on its severity and the degree of associated left ventricular disease. Mild aortic stenosis is associated with a normal carotid pulse and a short systolic murmur, which stops well before the second sound, as a pressure difference between the left ventricular cavity and the aorta is present only during the first part of systole. In addition, both components of the second heart sound are audible and splitting is normal. An uncalcified bicuspid aortic valve causes mild stenosis, with an ejection click and systolic murmur, often followed by a short early diastolic murmur. Unless the aortic stenosis is very long standing and accompanied by severe left ventricular disease, the upstroke of the carotid pulse is slow. Left ventricular hypertrophy can be diagnosed from an apical impulse which is characteristically sustained, although not necessarily displaced, as the heart size is normal. A double impulse due to a palpable left atrial contraction is further evidence of left ventricular hypertrophy, and its presence correlates with an increase in the contribution of left atrial systole to overall stroke volume. A raised left ventricular end-diastolic pressure can be deduced from accentuation of pulmonary valve closure, which forms the only component of the second sound. The severity of left ventricular disease does not necessarily parallel that of aortic stenosis, and it is not uncommon to see mild aortic stenosis in association with severe left ventricular disease. 2.5.6. Differential diagnosis Hypertrophic cardiomyopathy 38 Patients with hypertrophic cardiomyopathy frequently have a history of dyspnoea, chest pain, and syncope, which may be very similar to valvular aortic stenosis, although it may have lasted for longer. On physical examination the carotid pulse is normal or jerky, rather than slow rising, and the systolic murmur, when present, tends to be louder down the left sternal edge, or even at the apex rather than at the base of the heart and over the carotid arteries. The definitive diagnosis is made by echocardiography or left ventricular angiography, which shows obliteration of the apical part of the left ventricular cavity at end-systole. In some patients with valvular aortic stenosis, the pattern of left ventricular hypertrophy may resemble that seen in hypertrophic cardiomyopathy, and which may persist postoperatively. This should probably be regarded as a manifestation of severe secondary left ventricular hypertrophy rather than as the coexistence of two independent conditions. Fixed subaortic stenosis This condition usually presents in asymptomatic children and in young adults, but in those with a systolic murmur is found on the routine examination. The physical signs differ from those of aortic valve stenosis in the same age group in that case when an ejection click is absent; and a short early diastolic murmur is usually present. As on valvar aortic stenosis, there is clinical and cardiographic evidence of the left ventricular disease which may be very severe. Two-dimensional echocardiography usually demonstrates the site of obstruction to the left ventricular outflow, and is capable to distinguishing a discrete membrane or a more extensive, tunnel stenosis. M-mode echocardiography may show mid-systolic closure of the aortic valve, which is not specific to this condition, but is none the less rather suggestive of its presence. The diagnosis can also be confirmed by left ventricular angiography which demonstrates the presence of a small chamber immediately under the aortic valve. Congestive cardiomyopathy Patients with long-standing aortic stenosis may present with severe breathlessness, a large heart on radiography, a small volume pulse with a normal upstroke, a third heart sound, and pansystolic murmur due to papillary muscle dysfunction. This clinical picture represents one possible outcome of patients with untreated aortic stenosis. The diagnosis is suspected from the presence of calcification in the aortic valve on the lateral chest radiography and can be confirmed by demonstrating a pressure drop across the valve. Values as low as 30 to 40 mmHg are common in these circumstances, even when the stenosis is severe, so that it is thus necessary to allow for the small forward stroke volume, and calculate the valve area; values of less than 0.7 cm; indicate severe stenosis. If the clinical state can be improved by medical management, then the typical physical signs of aortic stenosis reappear. Heart block 39 Patients with aortic stenosis may develop complete heart block and, with the slow heart-rate and corresponding increase in stroke volume, the slow-rising pulse may not be apparent, so that the condition can mimic uncomplicated complete heart block. The combination should be suspected when the systolic blood pressure is low or normal, since it is raised in uncomplicated heart block with a slow ventricular rate. The true diagnosis becomes apparent when pacing is instituted. 2.5.7. Management Medical therapy Medical treatment has little to offer in aortic stenosis because, in mild cases it is unnecessary, and in severe ones ineffective. However, it is essential that all patients with aortic stenosis, of whatever severity, have prophylactic antimicrobials for any potentially septic hazard. Patients with severe left ventricular disease and fluid retention will benefit from a period of bed rest and treatment with a diuretic before operation is contemplated, but it must be remembered that the primary abnormality is a mechanical one, which cannot be significantly modified by altering renal sodium handling by diuretic agents. Prolonged treatment of such patients with large doses of powerful diuretics merely induces potassium depletion with a corresponding increase in the risk of postoperative rhythm disturbances. Surgical therapy The operative mortality for aortic valve replacement is 2 to 3% and is <5% if coronary bypass surgery is also performed. Severe aortic stenosis requires intervention. Unfortunately, aortic balloon valvuloplasty, though satisfactory in infants and children, is almost uniformly effectless in adults in whom the cusps are calcified, and the procedure has been largely abandoned for this age group. Aortic valve replacement, however, is an extremely effective operation. In uncomplicated cases, it can be carried out with low mortality and morbidity, and thus should be considered in all patients in whom the disease causes significant symptoms. It is likely to relieve breathlessness, angina, and syncope, whether due to ischaemic heart disease or to the aortic stenosis itself. Associated coronary artery disease is usually treated with bypass grafting at the same operation. This combined approach implies that all patients should be studied with coronary arteriography preoperatively, and increases the length of the operation itself. It is possible that left ventricular function can be improved, or that mortality reduced still further by this means, but it has still to be proved. Aortic valve replacement is also effective when significant aortic stenosis is complicated by severe left ventricular enlargement. Although the risks of surgery are greater, so are the benefits, and the remarkable improvement in both symptoms and prognosis that may follow surgery for this combination of valve and ventricular disease is amongst the most gratifying in cardiology. On the other extreme is the patient who is asymptomatic, but who is found to have a systolic murmur and evidence of significant aortic stenosis on the routine examination. Although a decision will clearly be influenced 40 by local surgical facilities and the preferences of the patient, it should be remembered that in the absence of symptoms or limitation of exercise tolerance such patients have an excellent prognosis, so that operation can safely be delayed. Clearly, though, they would be kept under review. 2.6. AORTIC REGURGITAION 2.6.1. Definition Aortic regurgitation produces a reflux of blood form the aorta into the left ventricle during ventricular diastole. 2.6.2. Etiology Rheumatic fever produces scarring and fibrosis of the valvular margins. Hypertension, as well as atherosclerosis can be the reason of aortic incompetence Conditions that affect the ascending aorta and produce valvelar incompetence include siphilis, heritable disorders of connective tissue, arthritic disease, and cystic medial necrosis of the aorta. Degeneration of aortic valve occurs in Marfan’s syndrome. 2.6.3. Pathogenic and hemodynamic abnormalities Although the end –diastolic pressure may be normal or slightly elevated in the early phases, progressive regurgitation dilates the chamber by myocardial fibers slippage and sarcomere replication with eventual hypertrophy and elevated left ventricle end-diastolic pressure. This compensatory mechanism s achieved with an ejection fraction above 50%. A progressive decline in aortic diastolic pressure due to regurgitation of blood into the left ventricle can reduce coronary blood flow in sever chronic aortic regurgitation and thus create conditions for subendocardial ischemia. Eventually, the declining ejection fraction and inotropic state, along with limits to eht dilation /hypertrophymechanism/, markedly elevates the left venricular filling pressure with pulmonary venous capillary congestion. Pathophysiology of aortic regurgitation (Figure 6) 1) left ventricle dilation; 2) left ventricle hypertrophy; 3) hyperdynamic peripheral circulation. 41 2.6.4. Clinical features Figure 6. Aortic regurgitation. The increased myocardial oxygen demands in aortic stenosis with the underperfused subendocaridial myocardium can produce arrhythmias, chest pain, and even sudden death. Chest pain, syncope, and heart failure and the characteristic symptoms of aortic stenosis, even though a gradient across the valve can exist for years before the patient develops symptoms. Complains Early symptoms are: 1. Palpitation. 2. Fatique. 3. Dyspnea on exertion. 4. Angina may also be noted with left ventricular hypertrophy and low diastolice pressures, which increase oxygen demand and decrease oxygensupply, respectively. 42 Physical examination In aortic regurgitation, the physical finding reglect the large left ventricular stroke volume ejected into the systemic circulation and the rapid diastolic run-off into the left vertical. The peripheral pulse is characteristically bounding, and additional manifestation of the wide pulse (Corrigan’s pulse) pressure includes: - head bobbing; - pulse retinal arteroles; - bouding carotid pulse (de Musset’s symptom); - capillary pulsations in the nail beds (Quincke’s symptom). Inspecting the precordium Because the contour of the heart does not become enlarged with concentric hypertrophy, abnormalities may not be visible of chest examination. On palpation When the left ventricle dilates owing to myocardial failure, the impulse is displaced laterally and becomes more diffuse On percussion with significant left ventricular dilatation left cardiac border may be percussed to the left of the left midclavicularis line. On auscultation direct findings of mitral stenosis are determined: 1) the high-pitched, blowing, decrescendo diastolic murmur heart best along the left sternal border during full expiration while the patient is in the sitting position; 2) diastolic rumble or Austin Flint murmur due to vibration of the anterior leaflet of the mitral valve in the regurgitation jet may be audible at the apex; 3) pistos short sounds over the femoral arteries doulbe Traube’s sound); 4) to-and-pro murmur elicited from the femoral artery with slight compression of the stethoscope (Dirozierz’s murmur). 2.6.5. Investigation Electrocardiogram Left ventricular hypertrophy is the most common finding on the electrocardiogram: 1. Increase in QRS amplitude and ST-T changes of a starain pattern. 2. Left-axis deviation as a result conduction disturbances and left bundle branch block. Chest radiograph 1. Left ventricular enlargement. 43 2. Dilation of proximal aorta. Significant aortic regurgitation nearly always causes cardiac enlargement on chest radiography. The aortic root is often dilated, but the aortic valve not necessarily calcified. The pulmonary vessels remain normal until severe left ventricular disease develops. Echocardiogram 1) Regurgitation flow can be appreciated by the vibration of the anterior mitral leaflet. 2) Left ventricular chamber dimensions, and wall thickness. 3) Dilation of left ventricle. Left ventricular cavity size can be measured by echocardiography and stroke volume estimated. The anatomy of the aortic valve and root can be determined. Dissection can sometimes be detected, vegetations on the aortic valve are well seen, and sometimes aortic root abscess can be detected. When infective endocarditis is suspected, transoesophgeal echo may give further useful information. It is also the means of choice for demonstrating aortic root aneurysms. In acute aortic regurgitation, mitral valve movement is very abnormal, showing premature closure . This results from severe regurgitation into a relatively noncompliant left ventricle causing the pressure to rise, and thus closing the mitral valve in mid-diastole. The rise in ventricular diastolic pressure causes difference from aortic pressure to fall to 20mmHg or less. This figure is an important one, as it represents the pressure supporting coronary flow, which may therefore be compromised. Cardiac catheterization It is not usually necessary to resort to cardiac catheterization for the diagnosis of aortic regurgitation. Although an aortogram may give useful information, it should be avoided in seriously ill patients because radiographic contrast medium expands the plasma volume and depresses ventricular function, but many surgeons require coronary arteriography to be performed even in the absence of clinical evidence of significant coronary artery disease. 2.6.6. Diagnosis As with aortic stenosis, it is not enough to establish the presence of aortic regurgitation; its severity must be estimated and the state of the left ventricle assessed. In uncomplicated cases, the severity can be judged indirectly from the carotid pulse and from the heart size on chest radiography, but direct measurement of left ventricular cavity size and stroke volume by echocardiography or angiography is a much more satisfactory method. Left ventricular disease can be suspected clinically from accentuated pulmonary valve closure, and from chest radiography by the presence of pulmonary vascular congestion and inappropriate cardiac enlargement. But again, left ventricular function is most satisfactorily assessed by echocardiography or angiocardiography: the characteristic feature 44 being enlargement of end-diastolic and in particular, end-systolic cavity size out of proportion to stroke volume so that ejection fraction falls. Acute aortic regurgitation may present difficulties in diagnosis when the classical physical signs are modified, but it should be suspected in any patient with severe systemic infection, low cardiac output state, and a normal or sightly collapsing pulse, with pulmonary congestion on chest radiography. Echocardiography is particularly useful in making a definite diagnosis non-invasively, demonstrating aortic vegetations, a large stroke volume, and premature mitral valve closure. It is also important to confirm or exclude other types of valve disease. Coexistent aortic stenosis is often diagnosed on the basis of an ejection systolic murmur, but this does not constitute adequate evidence, and in order to confirm its presence clinically, a bisferiens pulse should be present. Additional rheumatic mitral stenosis is best confirmed or excluded by echocardiography, although the presence of atrial fibrillation, a palpable first sound, or an opening snap makes its presence very likely on clinical grounds. Mitral regurgitation leads to an additional pansystolic murmur at the apex, which may sound continuously with the early diastolic murmur across the second sound. It is usually caused by a dilated valve ring in the absence of organic mitral valve disease, and thus indicates considerable left ventricular enlargement. It is usually unnecessary to establish the exact considerable left ventricular enlargement. It is usually unnecessary to establish the exact aetiology of the aortic regurgitation, although it is important to exclude infection and investigate the presence of disease of the aortic root. This should be suspected if there is a history of chest pain that is not clearly anginal in nature, and also from excessive dilatation of the ascending aorta on chest radiography or a long P-R interval on electrocardiography. 2.6.7. Differential diagnosis In the presence of severe pulmonary hypertension, the pulmonary artery may dilate, causing functional pulmonary regurgitation and a soft early diastolic murmur (Graham-Steell murmur). The carotid pulse is normal. Difficulty in diagnosis usually arises when the patient has pulmonary hypertensive mitral valve disease, and an early diastolic murmur. In these circumstances, aortic regurgitation may not necessarily cause an abnormal carotid pulse. In many cases, the differential diagnosis can only be made by Doppler echocardiography. But on clinical grounds, pulmonary incompetence is more likely when there is other evidence of severe pulmonary hypertension, and in particular, when chest radiography shows the main pulmonary artery to be appreciably dilated. Aortic regurgitation should also be distinguished from other causes of aortic run-off, which include persistent ductus arteriosus, ruptured sinus of Valsalva aneurysm or coronary arteriovenous fistula. These all cause an increase in pulse pressure, and a continuous murmur down the left sternal edge, which may be confused with the combination of aortic regurgitation and mitral regurgitation. Additional abnormalities which may give rise to confusion are the combination of aortic regurgitation and a ventricular septal defect, and finally, the rare anomaly of aortic45 left ventricular tunnel, in which the haemodynamic disturbance is identical to that of aortic regurgitation. This differential diagnosis can usually be established by Doppler echocardiography, but contrast angiography may sometimes be needed. 2.6.8. Management Mild or moderately severe aortic regurgitation is well tolerated and requires no treatment other than prophylactic antimicrobial to prevent infective endocarditis. Severe aortic regurgitation should be treated by aortic valve replacement. If the patient is symptomatic, the decision as to timing of the operation is not difficult. In an asymptomatic patient with severe disease, this may depend on local facilities, and the views of the patient must be taken into account. In general, however, evidence of left ventricular disease, aortic root disease, increasing heart size on chest radiography, or cavity size on echocardiography, or a history of infective endocarditis, are all indications for early operation. To depend on the results of any single investigation, convenient as it may seem, is inflexible in practice and not recommended. However, the patient must be kept under regular review, since severe left ventricular disease may become apparent over a period as short as 1 to 2 years in the absence of significant symptoms, increasing the risk of operation and reducing the functional improvement postoperatively. Acute aortic regurgitation is a surgical emergency. As it is nearly always due to infective endocarditis, blood cultures should be taken so that the organism can be isolated retrospectively and antibiotics strated preoperatively. One of the most useful single criteria for emergency aortic valve replacement has proved to be the presence of premature mitral valve closure on the M-mode echocardiogram. When emergency surgical facilities are not available, the haemodynamic state can sometimes be stabilized with vasodilator treatment as with severe mitral regurgitation. However, the patient should be transferred as soon as possible to a centre capable of performing open heart surgery, if progress is not maintained. A prolonged preoperative course of antibiotics is contraindicated in such patients, since the valve is rarely sterilized, and the delay causes further deterioration in the left ventricular function. 2.6.9. REFERENCES 1. Ball C.J.D., Williams A.W., Davies J.N.P. Endomyocardial fibrosis. //Lancet. – 1954. - № 1. – Р.1040-1049. 2. Further evidence relating mitral valve prolapse to cerebral ischemic events. / Barnett H.J.M., Boughner D.R., Taylor D.W., Cooper P.E., Kostuk W.J., Nichol P.M. //New England Journal of Medicine. – 1980. – Vol. 302. – Р.139-144. 3. Mitral annular calcification and the risk of stroke in an elderly cohort. / Benjamin E.J., Plehn J.F., D'Agostino R.B., et al. //New England Journal of Medicine. – 1992. - Vol. 327. – Р.374-379. 4. Blackstone E.H., Kirklin J.W. Recommendations for prophylactic removal of heart valve prostheses. // Journal of Heart Valve Disease. – 1992. - № 1. – Р.3-14. 46 5. Bulkley B.H., Roberts W.C. The heart in systemic lupus erythematosis, and change induced in it by corticosteroid therapy. // American Journal of Medicine. – 1976. - №58. - Р.243-264. 6. Predictors of long-term outcome after percutaneous balloon mitral valvuloplasty. / Cohen, D.J., Kuntz, R.E., Gordon, S.P.F. et al. // New England Journal of Medicine. – 1992. - Vol.327. – Р.1329-1335. 7. Davies M.J., Moore B.P., Braimbridge M.C. The floppy mitral valve. Study of incidence, pathology, and complications in surgical, necropsy and forensic material. //British Heart Journal. – 1978. - № 40. – Р.468-481. 8. Fowler N., van der Bel-Kahn J.M. Indications for surgical replacement of the mitral valve with particular reference to common and uncommon causes of mitral regurgitation. //Americal Journal of Cardiology. – 1979. - № 44. – Р.157. 9. Groves P.H., Hall R.J.C. Late tricuspid regurgitation following mitral valve surgery. //Journal of Heart Valve Disease. – 1992. - № 1. – Р.80-86. 10. Jeresaty R.M. Sudden death in the mitral prolapse click syndrome // Americal Journal of Cardiology. – 1976. - №37. – Р.317-318. 11. Leatham A. Brigden W. Mild mitral regurgitation and the mitral prolapse fiasco. // American Heart Journal. – 1990. - № 99. – Р.659-664. 12. Oakley C.M. Burkhardt D. Optimal timing of surgery for chronic mitral or aortic regurgitation. //Journal of Heart Valve Disease. – 1993. - № 2. – Р.223-229. 13. Rahimtoola S.H. Valvular heart disease; a perspective. //Journal of American College of Cardiology. – 1983. - № 1. – Р.199-215. 14. Cardiac valvular lesions in rheumatoid arthritis. / Roberts W.C., Hehoe J.A., Carpenter D.F., Golden A. // Archives of Internal Medicine. – 1968. - Vol. 122. – Р.141-146. 15. Congential bicuspid aortic valve causing severe, pure aortic regurgitation without superimposed infective endocarditis. / Roberts W.C., Morrow A.G., McIntosh C.L., Jones M., Epstein S.E. // Americal Journal of Cardiology. – 1987. № 47. – Р.206-209. 16. Ruttley M.S.T. The chest radiograph in adult heart valve disease.// Journal of Heart Valve Disease. – 1992. - № 2. – Р.205-217. 17. Selzer A. Changing aspects of the natural history of aortic stenosis. //New England Journal of Medicine. – 1987. - Vol.317. – Р.91-98. 18. Smith N., McAnulty J.H., Rahimtoola S.H. Severe aortic stenosis with impaired left ventricular function and clinical heart failure: results of valve replacement. //Circulation. – 1978. - № 58. Р.255-264. 19. The natural history of rheumatic aortic regurgitation and the indications for surgery./ Smith H.J., Neutze J.M., Roche A.H.G., Agnew T.M., Barratt-Boyes B.G. //British Heart Journal. – 1976. - № 38. – Р.147-154. 20. Vaughton K.C., Walker D.R., Sturridge M.F. Mitral valve replacement for endocarditis caused by Libman Sachs endocarditis. //British Heart Journal. – 1979. - № 41. – Р.730-733. 47 21. Rheumatic aortic stenosis in young patients presenting as combined aortic and mitral stenosis. / Vijayaraghavan G., Cherian G., Krishnaswami S., Sukamar I.P., John S. //British Heart Journal. – 1977. - № 39. – Р.294-298. 22. Rheumatic aortic stenosis in young patients presenting as combined aortic and mitral valve disease. / Vijayaraghavan G., Cherian G., Krishnaswami S., Sukamar John S. //British Heart Journal. – 1977. - № 39. – Р.294-298. 23. Vlodaver Z., Edwards J.E. Rupture of ventricular septum or papillary muscle complicating myocardial infarction. //Circulation. – 1977. - № 55. – Р.815-822. 24. Etiology of clinically isolated, severe, chronic pure mitral regurgitation: analysis of 97 patients over 30 years of age having mitral valve replacement. / Waller B.F., Morrow A.G., Maron B.J., et al. // American Heart Journal. 1982. Vol.104. – Р.276-288. 25. Wood P. An appreciation of mitral stenosis. Part 1. Clinical features. //British Medical Journal. 1954. - № 1. – Р.1051-1063. 26. Wood P. An appreciation of mitral stenosis. Part II. Investigations and results. //British Medical Journal. – 1954. - № 1. – Р.1113-1124. 2.7. TRICUSPID REGURGITATION 2.7.1. Frequency Internationally: Incidence of tricuspid regurgitation appears to be less than 1%. The morbidity and mortality of the disease process are secondary to the underlying cause. In rheumatic disease, mortality rates with treatment are less than 3%. In Ebstein anomaly, mortality depends upon the severity of the deformity and the feasibility of correction. Mortality rates with correction are approximately 10%. Tricuspid regurgitation resulting from myocardial dysfunction or dilatation has a mortality rate of up to 50% at 5 years. No race predilection is apparent. No sex predilection is apparent. Ebstein anomaly can be detected in birth and during early childhood. In patients older than 15 years, the most common form of tricuspid regurgitation is rheumatic valvular disease. In the adult population, other predisposing factors, including carcinoid, bacterial endocarditis, and CHF, takes precedence. 2.7.2. Background The causes of pure tricuspid regurgitation are multiple, and this lesion is the fifth most frequently excised native cardiac valve in patients older than 15 years. Tricuspid regurgitation may result from structural alterations of any one or all of the components of the tricuspid valve apparatus. Components include the leaflets, chordae tendinea, annulus, and papillary muscles or adjacent right ventricular (RV) muscle. The lesion may be classified as primary when it is caused by an intrinsic abnormality of the valve apparatus or as secondary when it is caused by RV pressure or volume overload. 48 2.7.3. Classification of tricuspid regurgitation According to the ISD -10 the Tricuspid Regurgitation related to the Diseases of the circulatory system (I00-I99), Chronic rheumatic heart diseases (I05-I09), Rheumatic tricuspid valve diseases (107) and includes Tricuspid insufficiency I07.1, Tricuspid (valve) insufficiency (rheumatic). 2.7.4. Etiology Pure tricuspid regurgitation can be caused by at least 9 conditions. Rheumatic heart disease o o o Tricuspid regurgitation secondary to rheumatic involvement is usually associated with mitral valve pathology. The valve develops diffuse fibrous thickening without commisural fusion, fused chordae, or calcific deposits. Occasionally, the chordae may be mildly thickened by fibrous tissue. Rheumatic disease is the most common cause of pure tricuspid regurgitation due to deformation of the leaflets. Endocarditis o o This is an important cause of pure tricuspid regurgitation. Precipitating factors that can contribute to infection of the valve include alcoholism, opiate addiction, neoplasms, infected indwelling catheters, extensive burns, and hereditary immune deficiency disease. The clinical presentation is that of pneumonia from septic emboli rather than CHF. Heart murmurs are frequently absent. Annular abscesses are not uncommon. Ebstein anomaly o o This entity is a congenital malformation of the tricuspid valve characterized by annular insertion of the septal and posterior leaflets displaced apically and atrialization of a portion of the ventricular myocardium. Prognosis for these patients depends upon the degree of apical displacement of the tricuspid annulus and the severity of the regurgitation. Prolapse (floppy, redundant) o o The incidence of floppy tricuspid valve varies from 0.3-3.2%. The lesion appears to be associated with prolapse of the mitral valve and does not appear to occur in an isolated fashion. 49 o Histological examination of the floppy tricuspid valve shows alterations on the valve spongiosa. Carcinoid o o o Pure tricuspid regurgitation can occur as part of the carcinoid heart syndrome. Fibrous white plaques form on the ventricular aspect of the tricuspid valve and endocardium, causing the valve to adhere to the RV wall. Proper systolic coaptation does not occur, resulting in tricuspid regurgitation. Papillary muscle dysfunction o o Papillary muscle dysfunction may result from necrosis (secondary to myocardial infarction), fibrosis, or infiltrative processes. Although dysfunction secondary to myocardial infarction is less common than the same pathology observed with the mitral valve, the underlying cause must be determined in order to plan treatment. Trauma o o Occasionally, trauma to the right ventricle may damage the structures of the tricuspid valve, resulting in insufficiency of the structure. More commonly associated with stab wounds, projectile destruction of the valve can also occur. Connective-tissue diseases o o o Patients with Marfan syndrome or other connective-tissue diseases (eg. osteogenesis imperfecta, Ehlers-Danlos syndrome) may have pure tricuspid regurgitation. Other valvular dysfunction is also observed in the same patient. The etiology of the regurgitation can be attributed to a floppy tricuspid valve and a mildly dilated tricuspid valve annulus. Anatomically normal tricuspid valves o o o 50 A common etiology of tricuspid regurgitation is dilatation of the RV cavity. The valve structures are normal; however, because of enlargement of the cavity and dilatation of the annulus, proper coaptation of the leaflets is not possible. Causes of the dilatation include mitral stenosis, pulmonic stenosis, pulmonary hypertension, dilated cardiomyopathy, and RV failure. 2.7.5. Pathophysiology The pathophysiology of tricuspid regurgitation focuses on the structural incompetence of the valve. The incompetent nature of the valve can result from primary structural abnormalities of the leaflets and chordae or from secondary myocardial dysfunction and dilatation. Tricuspid valve insufficiency is generally found in combination with tricuspid stenosis. The most common cause of this problem appears to be rheumatic in origin. Ebstein anomaly accounts for the most common congenital form of this abnormality. In tricuspid regurgitation, chronic right atrial overload results in right-sided congestive heart failure (CHF) manifested by hepatic congestion, peripheral edema, and ascites. Because of the impedance of flow to the pulmonary vasculature, hypoxemia, cyanosis, and polycythemia may result. 2.7.6. Clinical features Anamnesis The patient with tricuspid regurgitation presents with the signs and symptoms of right-sided heart failure. The spectrum of presenting symptoms is dependent upon whether the condition is secondary to left ventricular (LV) dysfunction. If it is, dyspnea on exertion and paroxysmal nocturnal dyspnea accompany ascites and peripheral edema as common presenting complaints. Exercise intolerance and hypoxemia may also be observed. The patient rarely reports angina, which may be present in the absence of coronary artery disease secondary to RV overload and strain. These patients must be questioned regarding the drug use, the history of rheumatic fever and febrile episodes because bacterial endocarditis is a common cause of tricuspid valvular disease. Physical S3 gallop is present, and the following physical findings may be found: Jugular venous distention with a prominent V wave: When present, a pansystolic murmur is heard along the lower left sternal border with inspiratory accentuation. Diminished peripheral pulse volume secondary to impaired forward blood flow: Patients with this sign may have relative hypotension secondary to therapeutic interventions used to decrease volume overload. 51 Pulmonary rales associated with LV dysfunction or mitral stenosis. RV heave and gallop that increases with inspiration. Ascites. Peripheral edema. Cachexia, cyanosis, and jaundice. Atrial fibrillation. A high-pitched pansystolic murmur (loudest in the fourth intercostal space in the parasternal region): The murmur is usually augmented during inspiration and is reduced by intensity and duration in the standing position and during a Valsalva maneuver. A short, early diastolic flow rumble may be present. 2.7.7. Laboratory changes Chemistry findings may show abnormal hyperbilirubinemia secondary to liver congestion. liver function and 2.7.8. Instrumental examination Chest radiography o Marked cardiomegaly is evident. o Evidence of elevated right atrial pressure may include distention of the azygous vein and pleural effusions. o Ascites with diaphragmatic elevation may be present. o Pulmonary arterial and venous hypertension is common. Echocardiography 52 o The right ventricle is dilated. o Paradoxical motion of the ventricular septum is observed and is similar to that found in an atrial septal defect. o Delayed closure of the tricuspid valve is observed. o Prolapse of the tricuspid valve may be evident, as well as vegetations if endocarditis is present. 2.7.9. Other Tests Electrocardiography o Findings are usually nonspecific. o Incomplete right bundle-branch block, Q waves in lead V1, and atrial fibrillation are found. 2.7.10. Procedures Cardiac catheterization o Right atrial pressure and RV end-diastolic pressure are elevated. A rise or no change of the right atrial pressure on deep inspiration is characteristic of tricuspid regurgitation. o The use of angiography in this setting is controversial. 2.7.11. Classification of tricuspid regurgitation There are classification of tricuspid regurgitation by the ECG (table 2) and Echo CG (table 3). Classification by the ECG By the tricuspid insufficiency is observed the electric axis of heart deviation to the right side and marked signs of right atrium and right ventricle hypertrophy. For the right atrium hypertrophy is typical high amplitude of P in II, III, aVF, V1 and sometimes in V2 levels. ECG characteristics of the right ventricle hypertrophy: displasment of the ST lower and appearing of negative T in the III, aVF, V1, sometimes in V2; increasing of R amplitude in the V1 and V2, Increasing of the S amplitude in V5 and V6, R in V1>7 mm, in this case R V1+SV5>10,05 mm; sometimes ventricular complexes has form rSR; turn of the heart in longitudinal axis by the clock stocking, that is saying about the displacement of intermediate zone to the V5,V6; Increasing of time of internal deviation over the right ventricle in V1 more than 0,03 sec. 53 Table 2 - Classification of tricuspid regurgitation by EchoCG (Gogin E.E. 2003) Tricuspid insufficiency degree I degree II degree The extension of Profound of regurgitation mosaic regurgitation flow flow 3 mm Till to the low third part of atrium 3-5 mm Till to the middle part of atrium III degree 5-8 mm Till to the base of atrium IV degree More than 8 mm Till to the vena cava inferior 2.7.12. Differential diagnostics Table 3 - Differential diagnostic signs of the systolic murmur between tricuspid and mitral insufficiencies Signs Systolic murmur by the tricuspid insufficiency The place of Along with the left side of the best listening sternum, sometime in the epigastria zone and xiphoid processus The apex zone Irradiation Up the III, II rib, sometime The left axillaries zone to the clavicle, even the right axillaries zone Influence of the Valsalve’s test In the phase of strain intensity and extension of murmur decreased, after the stop of strain the murmur intensity and extension restore immediately Not changed The influence of the vasopressors 54 Systolic murmur by the mitral insufficiency In the phase of strain the murmur volume is decreased, after the stop of strain the murmur intensity restore during the 3-4 sec Increased Influence of amyl- nitrites Increased (due to the venous outflow increasing) Influence of the liver palpation Increased Decreased (due to the decreasing of the peripheral blood circulation) Not changed Differentials with the following diseases: Ascites. Atrial Fibrillation. Biliary Disease. Carcinoid Tumor, Intestinal. Cardiac Cirrhosis. Cardiogenic Shock. Cardiomyopathy, Dilated. Cirrhosis. Cor Pulmonale. Ebstein Anomaly. Eisenmenger Syndrome. Heart Failure. Mitral Regurgitation. Other problems to be considered: Marfan syndrome. Inborn errors of collagen formation. 2.7.13. Management Medical Care For patients in whom tricuspid regurgitation is secondary to the left-sided heart failure, treatment centers on adequate control of fluid overload and failure symptoms. Diuretic therapy with interventions to address the primary pathology is of paramount importance. Surgical Care Surgical intervention is indicated when structural deformity of the valve (eg, Ebstein anomaly) exists, when the valve is destroyed by bacterial endocarditis, or when ventricular dilatation is severe and uncontrolled with medical therapy. 55 Tricuspid regurgitation associated with mitral valve disease and pulmonary hypertension: o Assess the severity of the regurgitation by palpation of the valve at the time of mitral valve intervention. Patients with mild tricuspid regurgitation do not require intervention. o As pulmonary vascular pressures fall with successful mitral valve therapy, the tricuspid regurgitation tends to disappear. o Severe regurgitation has been successfully treated with tricuspid annuloplasty. Organic disease of the tricuspid valve: o o Tricuspid valve endocarditis: o Total excision of the tricuspid valve without immediate replacement is recommended and is well tolerated. o Diseased valvular tissue is excised to eradicate the endocarditis, and antibiotic treatment is continued. Most patients tolerate loss of the tricuspid valve well. If medical management does not control the tricuspid regurgitation well and the infections have been controlled, an artificial valve can be inserted. o Corrective measures for organic disease of the tricuspid valve usually involve valve replacement. Because of the increased incidence of prosthetic valve thrombosis in this low-flow position, a porcine heterograft is the valve of choice. Tricuspid valve replacement has been used in carcinoid heart disease and cardiogenic shock with RV infarction, and after cardiac transplantation. Ebstein anomaly: If this anomaly produces uncontrollable tricuspid regurgitation, then tricuspid valve replacement is necessary. Management The medical therapy used in the treatment of tricuspid regurgitation is directed towards the control of CHF that is causing or contributing to the problem: Diuretics - Are used to control the fluid overload associated with the process. 56 Furosemide (Lasix) - Increases excretion of water by interfering with chloridebinding cotransport system, which in turn inhibits sodium and chloride reabsorption in ascending loop of Henle and distal renal tubule. Dose must be individualized to the patient. Depending on response, it is administered with increments of 20-40 mg, no sooner than 6-8 h after the previous dose, until desired diuresis occurs. When treating infants, titrate with 1 mg/kg/dose increments until a satisfactory effect is achieved. Adult Dose. 20-80 mg/d PO/IV/IM; titrate up to 600 mg/d for severe edematous states Contraindications. Documented hypersensitivity; hepatic coma, anuria, and state of severe electrolyte depletion. Interactions. Metformin decreases concentrations; interferes with hypoglycemic effect of antidiabetic agents and antagonizes muscle-relaxing effect of tubocurarine; auditory toxicity appears to be increased with coadministration of aminoglycosides; hearing loss of varying degrees may occur; anticoagulant activity of warfarin may be enhanced when taken concurrently with this medication; increased plasma lithium levels and toxicity are possible when taken concurrently with this medication. Interactions. B - Usually safe but benefits must outweigh the risks. Precautions. Avoid dehydration and depletion of potassium and other electrolytes. Cardiac glycosides - These drugs (primarily digoxin) are used to control atrial fibrillation and to increase myocardial contractility. Digoxin (Lanoxin) - Cardiac glycoside with direct inotropic effects in addition to indirect effects on the cardiovascular system; acts directly on cardiac muscle, increasing myocardial systolic contractions. Its indirect actions result in increased carotid sinus nerve activity and enhanced sympathetic withdrawal for any given increase in mean arterial pressure. Adult Dose. 0.125-0.375 mg PO qd. Contraindications. Documented hypersensitivity; beriberi heart disease, idiopathic hypertrophic subaortic stenosis, constrictive pericarditis, and carotid sinus syndrome Interactions. IV calcium may produce arrhythmias in digitalized patients; medications that may increase digoxin levels include alprazolam, benzodiazepines, bepridil, captopril, cyclosporine, propafenone, propantheline, quinidine, diltiazem, aminoglycosides, oral amiodarone, anticholinergics, diphenoxylate, erythromycin, felodipine, flecainide, hydroxychloroquine, itraconazole, nifedipine, omeprazole, quinine, ibuprofen, indomethacin, esmolol, tetracycline, tolbutamide, and verapamil; medications that may decrease serum digoxin levels include aminoglutethimide, antihistamines, cholestyramine, neomycin, penicillamine, aminoglycosides, oral colestipol, hydantoins, hypoglycemic agents, antineoplastic treatment combinations (including carmustine, bleomycin, methotrexate, cytarabine, doxorubicin, cyclophosphamide, vincristine, procarbazine), aluminum or magnesium antacids, rifampin, sucralfate, sulfasalazine, barbiturates, kaolin/pectin, and aminosalicylic acid. 57 Pregnancy. C - Safety for the use during pregnancy has not been established. Precautions. Hypokalemia may reduce positive inotropic effect of digitalis; hypercalcemia predisposes patient to digitalis toxicity, and hypocalcemia can make digoxin ineffective until serum calcium levels are normal; magnesium replacement therapy must be instituted in patients with hypomagnesemia to prevent digitalis toxicity; patients diagnosed with incomplete AV block may progress to complete block when treated with digoxin; exercise caution in hypothyroidism, hypoxia, and acute myocarditis; adjust dose in renal impairment; highly toxic (overdoses can be fatal). Angiotensin-converting enzyme (ACE) inhibitors - Are used to provide afterload reduction, thereby decreasing the volume load on the right ventricle. Captopril (Capoten) -- Prevents conversion of angiotensin I to angiotensin II, a potent vasoconstrictor, resulting in increased levels of plasma renin and a reduction in aldosterone secretion. Adult Dose. 6.25-12.5 mg PO tid; not to exceed 150 mg tid. Contraindications. Documented hypersensitivity; renal impairment Interactions. NSAIDs may reduce hypotensive effects; ACE inhibitors may increase digoxin, lithium, and allopurinol levels; rifampin decreases levels; probenecid may increase levels; the hypotensive effects of ACE inhibitors may be enhanced when administered concurrently with diuretics. Pregnancy. C - Safety for use during pregnancy has not been established. Precautions. Category D in second and third trimester of pregnancy; caution in renal impairment, valvular stenosis, or severe CHF. Anticoagulants - are used to prevent thrombosis and embolization from the prosthetic valve used in the treatment of tricuspid regurgitation. Warfarin (Coumadin) -- Interferes with hepatic synthesis of vitamin K–dependent coagulation factors. Used for prophylaxis and treatment of venous thrombosis, pulmonary embolism, and thromboembolic disorders. Tailor dose to maintain an international normalized ratio (INR) in the range of 2-3. Adult Dose. 5-15 mg/d PO qd for 2-5 d; adjust dose according to desired INR. Pediatric Dose. Administered weight-based dosis of 0.05-0.34 mg/kg/d PO; adjust dose according to desired INR. Contraindications. Documented hypersensitivity; severe liver or kidney disease; open wounds or GI ulcers. Interactions. Drugs that may decrease anticoagulant effects include griseofulvin, carbamazepine, glutethimide, estrogens, nafcillin, phenytoin, rifampin, barbiturates, cholestyramine, colestipol, vitamin K, spironolactone, oral contraceptives, and sucralfate; medications that may increase anticoagulant effects include oral antibiotics, capecitabine, phenylbutazone, salicylates, sulfonamides, chloral hydrate, clofibrate, diazoxide, anabolic steroids, ketoconazole, ethacrynic acid, miconazole, nalidixic acid, sulfonylureas, allopurinol, chloramphenicol, cimetidine, disulfiram, metronidazole, phenylbutazone, phenytoin, propoxyphene, sulfonamides, gemfibrozil, acetaminophen, and sulindac. Pregnancy. X - Contraindicated in pregnancy 58 Precautions. Do not switch brands after achieving therapeutic response; caution in active tuberculosis or diabetes; patients with protein C or S deficiency are at the risk of developing skin necrosis 2.7.14. Further Inpatient Care Inpatient care requires control of any heart failure and treatment of any infectious process that may have affected the valve. Postoperative care encompasses these principles. If the valve has been replaced, the control of arrhythmias is paramount because patients succumb to this problem even if heart failure has been adequately controlled. Anticoagulation is generally in order if valve replacement has been undertaken because of the low flow state of the right side of the heart. Maintenance of the INR between 2,5-3 should prove sufficient to prevent thrombosis and embolization. 2.7.15. Further Outpatient Care Patients should be carefully monitored for the control of any heart failure. Repeat echocardiography is indicated at 6-month intervals for patients in whom the valve has been removed. Annual echocardiography should be considered in patients whose valve has been replaced. 2.7.16. In/Out Patient Meds Digitalis, diuretics (including potassium-sparing agents), ACE inhibitors, and anticoagulants are all indicated in the care of these patients. Antiarrhythmics are added as needed to control atrial fibrillation. Deterrence/Prevention Prevention of tricuspid regurgitation from bacterial endocarditis can be undertaken by securing good dental care and avoiding the use of illicit drugs, particularly by the intravenous route. 2.7.17. Complications Complications of tricuspid regurgitation include cardiac cirrhosis, ascites, thrombus formation, and embolization. Complications of operative intervention can include heart block, arrhythmias, thrombosis of the prosthetic valve, and infection. 59 2.7.18. Prognosis Prognosis in these patients is fair. If the cause of the regurgitation is an infection, removal of the valve generally cures the problem, provided that the inciting cause is removed (eg, poor dentition, illicit drug use). For patients with accompanying pulmonary hypertension or cardiac dilatation, the prognosis is directly associated with the prognosis for these problems. 2.7.19. Patient Education Patients should be instructed to reduce the use of salt and water. Elevation of the head of the bed may improve symptoms of shortness of breath. Careful instruction in the use of anticoagulants must be given. 2.7.21. Medical/Legal Pitfalls Failure to accurately diagnose and treatment. Failure to adequately monitor patients for the control of heart failure. Failure to adequately educate patients about their condition and its management. 2.7.22. REFERENCES 1. Prolapse of the antero-superior leaflet of the tricuspid valve secondary to congenital anomalies of the valvar and sub-valvar apparatus: a rare cause of severe tricuspid regurgitation. / Abrams DJ, Kilner P, Till JA, et al.// Cardiol Young. – 2005, Aug. – Vol. 15. №4. – Р.417-421. 2. Prolapse of the antero-superior leaflet of the tricuspid valve secondary to congenital anomalies of the valvar and sub-valvar apparatus: a rare cause of severe tricuspid regurgitation. / Abrams DJ, Kilner P, Till JA, et al. //Cardiol Young. – 2005, Aug. - Vol. 15. №4. – Р.417-421. 3. Optimal management of severe tricuspid regurgitation in cardiac transplant recipients. / Anderson C, Filsoufi F, Farivar RS, Adams DH. // J Heart Lung Transplant. – 2001, Feb. - Vol. 20 №2. – Р.247. 4. Burgess MI, Aziz T, Yonan N. Clinical relevance of subclinical tricuspid regurgitation after orthotopic cardiac transplantation. // J Am Soc Echocardiogr. – 1999, Feb. - Vol. 12 №2. – Р.164. 60 5. Daniels SJ, Mintz GS, Kotler MN. Rheumatic tricuspid valve disease: twodimensional echocardiographic, hemodynamic, and angiographic correlations. //Am J Cardiol. – 1983, Feb. - Vol.51 №3. – Р.492-496. 6. Successful palliation of Ebstein's malformation on the first day of life following fetal diagnosis. / DeLeon MA, Gidding SS, Gotteiner N, et al. // Cardiol Young. – 2000, Oct. - Vol.10 №4. – Р.384-387. 7. Long-term outcomes of tricuspid valve replacement in the current era. / Filsoufi F, Anyanwu AC, Salzberg SP, et al. //Ann Thorac Surg. – 2005, Sep. Vol.80 №3. – Р.845-850. 8. Long-term outcomes of tricuspid valve replacement in the current era. / Filsoufi F, Anyanwu AC, Salzberg SP, et al. //Ann Thorac Surg. – 2005, Sep. Vol.80, №3. – Р.845-850. 9. Frater R: Tricuspid insufficiency.// J. Thorac Cardiovasc Surg. – 2001, Sep. Vol.122, №3. – Р.427-429. 10. Tricuspid valve repair with the Cosgrove-Edwards annuloplasty system: early clinical and echocardiographic results. / Gatti G, Maffei G, Lusa AM, Pugliese P. // Ann Thorac Surg. – 2001, Sep. - Vol. 72, №3. – Р.764-767. 11. Tricuspid stenosis and regurgitation: Doppler and color flow echocardiography and cardiac catheterization findings. / Ha JW, Chung N, Jang Y, Rim SJ. // Clin Cardiol. – 2000, Jan. - Vol.23, №1. – Р.51-52. 12. Native valve salvage for post-traumatic tricuspid regurgitation. / Hachiro Y, Sugimoto S, Takagi N, et al.// J Heart Valve Dis. – 2001, Mar. - Vol.10, №2. – Р.276-278. 13. Right ventricular dP/dt/P(max), not dP/dt(max), noninvasively derived from tricuspid regurgitation velocity is a useful index of right ventricular contractility. / Kanzaki H, Nakatani S, Kawada T, et al. // J Am Soc Echocardiogr. – 2002, Feb. - Vol. 15, №2. – Р.136-142. 14. Khan I.A. Ebstein's anomaly of the tricuspid valve with associated mitral valve prolapse.// Tex Heart Inst J. – 2001. - Vol.28, №1. – Р.72. 15. Permanent cardiac pacing and its influence on tricuspid valve function. / Krupa W, Kozlowski D, Derejko P, Swiatecka G. // Folia Morphol (Warsz). – 2001, Nov. - Vol. 60, №4. – Р.249-257. 16. Tricuspid valve surgery for functional tricuspid valve regurgitation associated with left-sided valvular disease. / Kuwaki K, Morishita K, and 61 Tsukamoto M, Abe T. // Eur J Cardiothorac Surg. – 2001, Sep. - Vol.20, №3. – Р.577-582. 17. Traumatic tricuspid valve insufficiency: case report. / Leszek P, Zielinski T, Rozanski J, et al. // J Heart Valve Dis. – 2001, Jul. - Vol. 10, №4. – Р.545-547. 18. Correction of traumatic tricuspid insufficiency using the double orifice technique. / Luo GH, Ma WG, Sun HS, et al. // Asian Cardiovasc Thorac Ann. – 2005, Sep. - Vol. 13, №3. – Р.238-240. 19. Correction of traumatic tricuspid insufficiency using the double orifice technique. / Luo GH, Ma WG, Sun HS, et al. // Asian Cardiovasc Thorac Ann. – 2005, Sep. - Vol. 13, №3. – Р.238-240. 20. Asymptomatic traumatic tricuspid regurgitation: a case report with an early diagnosis./ Mansencal N, Mourvillier B, Schwob J, et al. //Ann Emerg Med. – 2002, Feb. - Vol.39, №2. – Р.205. 21. Matsunaga A, Duran C.M. Progression of tricuspid regurgitation after repaired functional ischemic mitral regurgitation. //Circulation. – 2005, Aug 30. - Vol. 112, №9 Suppl. – Р.I453-1457. 22. Matsunaga A, Duran C.M. Progression of tricuspid regurgitation after repaired functional ischemic mitral regurgitation. //Circulation. – 2005, Aug 30. - Vol. 112, №9 Suppl. – Р.I453-1457. 23. Mild tricuspid regurgitation: a benign fetal finding at various stages of pregnancy. / Messing B, Porat S, Imbar T, et al. // Ultrasound Obstet Gynecol. – 2005, Nov. - Vol.26, №6. – Р.606-609; discussion 610. 24. Mild tricuspid regurgitation: a benign fetal finding at various stages of pregnancy. / Messing B, Porat S, Imbar T, et al. // Ultrasound Obstet Gynecol. – 2005, Nov. - Vol.26, №6. – Р.606-609; discussion 610. 25. Cleft on tricuspid anterior leaflet./ Motoyoshi N, Tofukuji M, Sakurai M, et al. // Ann Thorac Surg. – 2001, Apr. - Vol.71, №4. – Р.1350-1351. 26. Replacement of the morphologically tricuspid valve in children with discordant atrioventricular connections. / Numata S, Uemura H, Yagihara T, et al. // J Heart Valve Dis. – 1999, Nov. - Vol. 8, №6. – Р.649-654. 27. Predisposing conditions for atrial fibrillation in atrial septal defect with and without operative closure./ Oliver JM, Gallego P, Gonzalez A, et al. // Am J Cardiol. – 2002, Jan 1. - Vol.89, №1. – Р.39-43. 62 28. Surgical pathology of carcinoid heart disease: a study of 139 valves from 75 patients spanning 20 years./ Simula DV, Edwards WD, Tazelaar HD, et al. // Mayo Clin Proc. – 2002, Feb. - Vol. 77, №2. – Р.139-147. 29. Atypical ductus venosus blood flow pattern in fetuses with severe tricuspid valve regurgitation. / Smrcek JM, Krapp M, Axt-Fliedner R, et al. // Ultrasound Obstet Gynecol. – 2005, Aug. - Vol.26, №2. – Р.180-182. 30. Atypical ductus venosus blood flow pattern in fetuses with severe tricuspid valve regurgitation. / Smrcek JM, Krapp M, Axt-Fliedner R, et al. // Ultrasound Obstet Gynecol. – 2005, Aug. - Vol.26, №2. – Р.180-182. 31. Long-term evaluation of treatment for functional tricuspid regurgitation with regurgitant volume: characteristic differences based on primary cardiac lesion. / Sugimoto T, Okada M, Ozaki N, et al. // J Thorac Cardiovasc Surg. – 1999, Mar. - Vol.117, №3. – Р.463-471. 33. Quantification of tricuspid regurgitation by measuring the width of the vena contracta with Doppler color flow imaging: a clinical study. / Tribouilloy CM, Enriquez-Sarano M, Bailey KR, et al. // J Am Coll Cardiol. – 2000, Aug. Vol.36, №2. – Р.472-478. 34. Adults with Ebstein's anomaly-Cardiopulmonary exercise testing and BNP levels Exercise capacity and BNP in adults with Ebstein's anomaly./ Trojnarska O, Szyszka A, Gwizdala A, et al. // Int J Cardiol. – 2005, Oct 18. 35. Adults with Ebstein's anomaly-Cardiopulmonary exercise testing and BNP levels Exercise capacity and BNP in adults with Ebstein's anomaly./ Trojnarska O, Szyszka A, Gwizdala A, et al. // Int J Cardiol. – 2005, Oct 18. 36. Exact quantitative selective annuloplasty of the tricuspid valve. / Yiwu L, Yingchun C, Jianqun Z, et al. // J Thorac Cardiovasc Surg. - 2001, Sep. - Vol. 122, №3. – Р.611-614. 63 3. MYOCARDITIS 3.1. Background Myocarditis is collection of diseases of infectious, toxic, and autoimmune etiologies characterized by inflammation of the heart. It is manifested by the including in pathologic processes of cardiomyocites, microvessels interstitial tissue, sometimes of the pericardium. It is current with developing of myocardium inflammatory infiltration and with damaging of cardiomyocites and their function. Subsequent myocardial destruction can lead to dilated cardiomyopathy. 3.2. Frequency of Myocarditis Myocarditis is a rare disease. The World Health Organization reports that incidence of cardiovascular involvement after enteroviral infection is 1- 4%, depending on the causative organism. Incidence varies greatly among countries and is related to hygiene and socioeconomic conditions. Availability of medical services and immunizations also affect incidence. Occasional epidemics of viral infections have been reported with an associated higher incidence of myocarditis. Studies give a wide spectrum of mortality and morbidity statistics. With suspected Coxsackie virus B, the mortality rate is higher in newborns (75%) than in older infants and children (10-25%). Complete recovery of ventricular function has been reported in as many as 50 percent of patients. Some patients develop chronic myocarditis (ongoing or resolving) and/or may eventually require cardiac transplantation. 3.3. Mortality/Morbidity Because of its difficulty in diagnosis and its possible role in sudden dysrhythmic death, morbidity is not known exactly. Rarely, acute myocarditis is fulminant and leads to death. The appropriate delicate balance of the immune response to viral invasion of myocytes indicates that a certain number of individuals, perhaps with genetic predispositions, will advance to dilated cardiomyopathy and heart failure, the most common long-term sequelae in those patients who do not recover completely. Appreciating that an unknown group of subclinical cases exists, of patients with apparent disease that is identified by a physician, it is often quoted that one third of patients recover without incident, in one third some mild myocardial scarring is developed, and in one third develop dilated cardiomyopathy is developed. Sex: The male-to-female ratio is 1.5:1. Age: The average age of patients with myocarditis is 42 years. 64 3.4. Etiology of myocarditis Causes The causes of myocarditis are numerous and can be roughly divided into infectious, toxic, and immunologic etiologies, with viral etiologies. Amongst the infectious causes, viral acute myocarditis is by far the most common. o Identification of the Coxsackie’s-adenovirus receptor protein explains the prevalence of these viruses as the causative agents in more than one half of cases. The receptor is the common target of the coxsackievirus B of the enterovirus family and serotypes 2 and 5 of the adenovirus family. o Other viruses implicated in myocarditis include influenza virus, echovirus, herpes simplex virus, varicella-zoster virus, hepatitis, Epstein-Barr virus, and cytomegalovirus. Hepatitis C, in particular, is becoming a major focus of research. o Human immunodeficiency virus (HIV) deserves special mention because it seems to function differently than other viruses. Although some evidence indicates that HIV directly invades myocytes, HIV genomes can be amplified from patients without histologic signs of inflammation. In addition, in patients who are infected with HIV, Tcell–mediated immune suppression increases the risk of contracting myocarditis due to other infectious causes, and this has been shown in myocardial biopsy samples. Nonviral infectious causes are numerous and various. Worldwide the most common bacterial cause is diphtheria, and, in South America, the protozoal Chagas disease is a common entity. Streptococcal and staphylococcal species and Bartonella, Brucella, Leptospira, and Salmonella species can spread to the myocardium as a consequence of severe cases of endocarditis. Borrelia burgdorferi, the spirochete agent in Lyme disease, is also a known cause of myocarditis. Parasitic myocarditis from trypanosomiasis, trichinosis, and, in the immunocompromised host, toxoplasmosis have been identified. Toxic myocarditis has a number of etiologies including both medical agents and environmental agents. o Among the most common drugs that cause hypersensitivity reactions are penicillin, ampicillin, and hydrochlorothiazide, methyldopa, and sulfonamide drugs. This syndrome is associated with peripheral 65 eosinophilia, fever, and rash in patients who have biopsy findings of an eosinophilic infiltrate of the myocardium. o Numerous medications (eg, lithium, doxorubicin, cocaine, numerous catecholamines, and acetaminophen) may exert a direct cytotoxic effect on the heart. Zidovudine (AZT) has been associated with myocarditis. o Environmental toxins include lead, arsenic, and carbon monoxide. o Wasp, scorpion, and spider stings. o Radiation therapy may cause myocarditis with the development of a dilated cardiomyopathy. Immunologic etiologies of myocarditis encompass a number of clinical syndromes and include the following: o Connective tissue disorders such as systemic lupus erythematosus (SLE), rheumatoid arthritis, and dermatomyositis that can often result in a dismal prognosis. o Idiopathic inflammatory and infiltrative disorders such as Kawasaki disease, sarcoidosis, and giant cell arteritis. Rejection of the posttransplant heart may present as inflammatory myocarditis. 3.5. Pathophysiology Myocarditis is defined as inflammatory changes in the heart muscle and is characterized by myocyte necrosis. Animal models of viral myocarditis have lead to a much greater understanding of the pathophysiology of acute, severe myocarditis and correlate with the findings in susceptible patients who apparently uptake viral RNA and develop a cytotoxic necrosis and rapid (1-2 d) cell death without the appearance of the interstitial infiltrate usually associated with myocarditis. Over 4-14 days, those cells that survive in initial insult, in response to macrophage activation and cytokine expression, develop the classic, histologically apparent infiltration of mononuclear cells. In this subacute viral-clearing phase, natural killer cells target myocardium expressing viral RNA and continue myocyte necrosis. Tumor necrosis factor is also involved in rapidly clearing virus, but its involvement results in the further recruitment of inflammatory cells, activates endothelial cells, and has negative inotropic effects. In the latter stages of the subacute process, 66 cytotoxic T lymphocytes infiltrate the myocardium and direct lysis of cardiocytes, which present virus fragments via the histocompatibility complex on the surface of myocyte membrane. Neutralizing antiviral antibodies also are developed to assist in the clearing of virus. In the chronic phases, the deleterious effects of either inadequate or inappropriately abundant immune response can lead to the unfortunate long-term sequelae of dilated cardiomyopathy and heart failure. In animal models of insufficient immune response, viral replication can continue and cause chronic destruction of myocytes. Biopsy results of patients with acute myocarditis who develop dilated cardiomyopathy demonstrate changes consistent with those seen in polymerase chain reaction (PCR) amplifying RNA from enteroviruses. On the opposite spectrum of immune activity, overabundant T cells may continue activity in the chronic phase and also may cause tissue destruction and heart failure. 3.6. Classification of myocarditis Classification of myocarditis, (Palaev N.R. 1981) By the etiologic characteristics: Infectional; drug induced; allergic; toxic; radiological; developing by the diffuse diseases of connecting tissue and by the system vasculities; burning; because of transplantant; idiopathic. By the pathogenetic variants: infectional- toxic; allergic; toxically- allergic. By the pathogenetic phases: infectional- toxic; immune-allergic; dystrophic; myocardiodistrophic. 67 By the morphological characteristics: alterative; exudative-prolipherative; a) dystrophic; b) inflammatory-infiltrative; c) mixed; d) vascular. By extension: diffuse; local. By the currency: acute; abortive; recidivic; latent; chronic. By the clinical variants of myocarditis: with small symptoms; pseudocoronar variant; pseudovalvular variant; arrhythmic variant; thromboembolic variant; decompensate variant; mixed variant. By the heaviness: easy; mean; heavy. According to the ICD-10 classification the myocarditis related by the Diseases of the circulatory system (I00-I99) and the other forms of heart disease (I30-I52.) The international classification of diseases (ICD-10) 68 140 I40.0 Acute myocarditis Infective myocarditis Septic myocarditis I40.1 I40.8 I40.9 Isolated myocarditis Other acute myocarditis Acute myocarditis, unspecified 141* I41.0* Myocarditis in diseases classified elsewhere Myocarditis in bacterial diseases classified elsewhere Myocarditis: · diphtheritic ( A36.8+ ) · gonococcal ( A54.8+ ) · meningococcal ( A39.5+ ) · syphilitic ( A52.0+ ) · tuberculous ( A18.8+ ) I41.1* Myocarditis in viral diseases classified elsewhere Influenzal myocarditis (acute): · avian influenza virus identified ( J09+ ) · other virus identified ( J10.8+ ) · virus not identified ( J11.8+ ) Mumps myocarditis ( B26.8+ ) I41.2* Myocarditis in other infectious and parasitic diseases classified elsewhere Myocarditis in: · Chagas' disease (chronic) ( B57.2+ ) · acute ( B57.0+ ) · toxoplasmosis ( B58.8+ ) I41.8* Myocarditis in other diseases classified elsewhere Rheumatoid myocarditis ( M05.3+ ) Sarcoid myocarditis ( D86.8+ ) I51.4 Myocarditis imprecisely defined 3.7. Clinical manifestation of myocarditis Complains 69 70 Many patients with the presence of a nonspecific illness are characterized by fatigue, mild dyspnea, and myalgias. A few patients are acutely ill with fulminant congestive heart failure (CHF) secondary to widespread myocardial involvement. Small and focal areas of inflammation in electrically sensitive areas may be by the etiology in patients whose initial presentation is a sudden death. Most cases of myocarditis are subclinical; therefore, the patient rarely seeks medical attention during acute illness. These subclinical cases may have transient ECG abnormalities. An antecedent viral syndrome is present in more than in one half of patients with myocarditis. The appearance of cardiac-specific symptoms occurs primarily in the subacute virus-clearing phase; therefore, patients commonly have it 2 weeks after the acute viremia. Fever is present in 20% of patients. Other symptoms include fatigue, myalgias and arthralgias, and malaise. Chest pain. o Chest discomfort is reported in 35% of patients. o The pain is most commonly described as a pleuritic, sharp, stabbing precordial pain. o It may be substernal and squeezing and, therefore, difficult to distinguish from that typical of ischemic pain. Dyspnea on exertion is common. Orthopnea and shortness of breath at rest may be noted if CHF is present. Palpitations are common. Syncope in a patient with myocarditis present consistently with should be carefully observed because it may be a signal of high-grade AV block or the risk for sudden death. Pediatric patients, particularly infants, present with nonspecific symptoms include the following: o Fever. o Respiratory distress. o Poor feeding or, in cases with CHF, sweating while feeding. o Cyanosis in severe cases. Physical examination Physical findings can range from nearly normal examination findings to signs of fulminant CHF. Patients with mild cases of myocarditis have a nontoxic appearance and simply may appear to have a viral syndrome. Tachypnea and tachycardia are common. Tachycardia is often out of proportion to fever. More acutely ill patients have signs of circulatory impairment due to the left ventricular failure. A widely inflamed heart shows the classic signs of ventricular dysfunction including the following: o Jugular venous distention. o Bibasilar crackles. o Ascites. o Peripheral edema. S3 or a summation gallop may be noted with significant biventricular involvement. Intensity of S1 may be diminished. Cyanosis may occur. Hypotension caused by the left ventricular dysfunction is uncommon in the acute setting and indicates a poor prognosis when present. Murmurs of mitral or tricuspid regurgitation may be present due to ventricular dilation. In cases where a dilated cardiomyopathy has developed, signs of peripheral or pulmonary thromboembolism may be found. Diffuse inflammation may develop leading to pericardial effusion, without tamponade, and pericardial and pleural friction rub as the inflammatory process involves surrounding structures. 71 3.8. Laboratory – instrumental investigation Laboratory tests Cardiac enzyme levels: o These levels are only elevated in a minority of patients. o Normally, a characteristic pattern of slow elevation and fall over a period of days occurs; however, a more abrupt rise is observed in patients with acute myocardial infarction. Cardiac troponin I may be more sensitive because it is present for longer periods after myocardial damage from any cause. Erythrocyte sedimentation rate (ESR) is elevated in 60% of patients with acute myocarditis. Leukocytosis is present in 25% of cases. Instrumental examination 72 Chest radiography: o A chest radiograph often reveals a normal cardiac silhouette, but pericarditis or overt clinical CHF is associated with cardiomegaly. o Vascular redistribution. o Interstitial and alveolar edema. o Pleural effusion. Echocardiography: o Impairment of the left ventricular systolic and diastolic function. o Segmental wall motion abnormalities. o Impaired ejection fraction. o A pericardial effusion may be present, although findings of tamponade are rare. o Ventricular thrombus has been identified in 15% of patients studied with echocardiography. MRI is capable to showing abnormal signal intensity in the affected myocardium: o Cardiac MRI is an emerging field in general and contrast-enhanced T1- weighted MRI has been shown to have sensitivities and specificities approaching 100% for diagnosis. o MRI is also becoming one of the modalities used in the evaluation of young patients with apparently idiopathic dysrhythmias, and this imaging study can differentiate focal and diffuse inflammation from the rare electrically significant myocardial tumor. Other Tests Electrocardiogram: o Sinus tachycardia is the most frequent finding. o ST- segment elevation without reciprocal depression, particularly when diffuse, is helpful in differentiating myocarditis from acute myocardial infarction. o Decreased QRS amplitude and transitory Q-wave development is very suggestive of myocarditis. o As many as 20% of patients will have a conduction delay, including Mobitz I, Mobitz II, or complete heart block. o Left or right bundle-branch block is observed in approximately 20% of abnormal ECG findings and may persist for months. Viral isolation from other body sites may be supportive of the diagnosis. PCR identification of a viral infection from myocardial tissue, pericardial fluid, or other body fluid sites can be helpful. If a systemic disorder (eg, SLE) is suspected, antinuclear antibody (ANA) and other collagen vascular disorder laboratory investigations may be useful. 3.9. Procedures Cardiac catheterization usually reveals normal coronary vessels and regional wall motion abnormalities with diminished ejection fraction. It has no benefit over noninvasive echocardiography. 73 Endomyocardial biopsy is the classically quoted diagnostic procedure of choice, but it has many limitations. o Extensive lymphocytic infiltrates with myocyte necrosis are the classic findings defined by the Dallas criteria. o Because of the patchy nature of myocarditis, significant sampling errors may occur. In addition, inter-rater reliability of the histologic criteria is very low. o The combination of expert pathologists' disagreement on findings and the difficulty of adequately sampling the myocardium results in only 30% of patients who are thought to have myocarditis actually having positive biopsy findings. 3.10. Criterions of the histological diagnostics Dallas Classification (1987) Initial Biopsy 1. Myocarditis: Myocardial necrosis, degeneration, or both, in the absence of the significant coronary artery disease with the adjacent inflammatory infiltrate with or without fibrosis. 2. Borderline myocarditis: The inflammatory infiltrate too sparse or myocyte damage not apparent. 3. No myocarditis Subsequent Biopsies 1. Ongoing (persistent) myocarditis with or without fibrosis. 2. Resolving (healing) myocarditis with or without fibrosis. 3. Resolved (healed) myocarditis with or without fibrosis. WHO Marburg Criteria (1996) First Biopsy 1. Acute (active) myocarditis: A clear-cut infiltrate (diffuse, focal or confluent) of >14 leukocytes/mm² (preferably activated T-cells). The amount of the infiltrate should be quantitated by immunohistochemistry. Necrosis or degeneration are compulsory, fibrosis may be absent or present and should be graded. 2. Chronic myocarditis: An infiltrate of >14 leukocytes/mm² (diffuse, focal or confluent, preferably activated T-cells). Quantification should be made by 74 immunohistochemistry. Necrosis or degeneration are usually not evident, fibrosis may be absent or present and should be graded. 3. No myocarditis: No infiltrating cells or <14 leukocytes/mm². Subsequent Biopsies 1. Ongoing (persistent) myocarditis: Criteria as in 1 or 2 (features of an acute or chronic myocarditis). 2. Resolving (healing) myocarditis: Criteria as in 1 or 2 but the immunological process is sparser than in the first biopsy. 3. Resolved (healed) myocarditis: Corresponds to the Dallas classification. 3.11. Management Emergency Department Care Because many cases of myocarditis are not clinically obvious, a high degree of suspicion is required to identify acute myocarditis. Fortunately, most patients have mild symptoms consistent with viral syndromes, and they will recover with simple supportive care on an outpatient basis. Standard treatment of clinically significant disease includes the detection of dysrhythmia with the cardiac monitoring, supplemental oxygen, and managing the fluid status. Recovering patients should have activity limitations for approximately 6 months, as animal models have shown permanent injury from a rapid return to activity. Left ventricular dysfunction developing from myocarditis should be approached in much the same manner as other causes of CHF with some exceptions). In general, sympathomimetic and beta-blocker drugs should be avoided because they increase the extent of myocardial necrosis and mortality. Patients presenting with Mobitz II or complete heart block require pacemaker placement. Consultations Patients who require emergency room treatment for new-onset of CHF, dysrhythmia, or cardiogenic shock should be admitted to the hospital with the continuous cardiac monitoring and cardiology consultation. 75 Medical therapy for myocarditis is an area of the avid research interest but with a little success in human trials. Treatment primarily involves managing the complications of myocarditis, chiefly thromboembolism, dysrhythmia, and CHF and is addressed in detail in the corresponding eMedicine Journal articles; little is specific to myocarditis except for a few specific aspects of the treatment of myocarditis-related CHF. Many studies have attempted to regulate down the immune response in an attempt to arrest the development of immune-mediated dilated myopathy. Animal and human trials have never shown benefit. Conventional immunosuppressives, such as cyclosporine, prednisone, and azathioprine, are likely to be discarded in the discussion of an immune response to the disease in favor of more tailored approaches. Immunomodulating agents -- The most promising research addressing the immune response in myocarditis involves immune modulators that target particular steps in the immune cascade without eliminating the ability of the body's defenses to shed virus. The role of TNF appears to be central in these approaches. Immune globulin intravenous (Gamimune, Gammagard, Gammar-P, Sandoglobulin) -- Neutralizes circulating myelin antibodies through anti-idiotypic antibodies; down regulates proinflammatory cytokines, including INF-gamma; blocks Fc receptors on macrophages; suppresses inducer T and B cells and augments suppressor T cells; blocks complement cascade; promotes remyelination; may increase CSF IgG (10%). Adult Dose: 2 g/kg IV over 2-5 d. Contraindications: Documented hypersensitivity; IgA deficiency. Interactions: Globulin preparation may interfere with immune response to live virus vaccine (MMR) and reduce efficacy (do not administer within 3 mo of vaccine). Pregnancy: C - Safety for the use during pregnancy has not been established. Precautions: Check serum IgA before IVIG (use an IgA-depleted product, eg, Gammagard S/D); infusions may increase serum viscosity and thromboembolic events; infusions may increase risk of migraine attacks, aseptic meningitis (10%), urticaria, pruritus, or petechiae (2-30 d postinfusion); increases the risk of renal tubular necrosis in elderly patients and in patients with diabetes, volume depletion and preexisting kidney disease; laboratory result changes associated with infusions including elevated antiviral or antibacterial antibody titers for 1 mo, 6-fold increase in ESR for 2-3 wk, and apparent hyponatremia Angiotensin converting enzyme inhibitors -- These agents are beneficial in the management of blood pressure and LV function in the heart failure. Captopril, in particular, has been shown to be beneficial in the treatment of the significant left ventricular dysfunction. Other ACE inhibitors have not shown the same effect in 76 animal trials, indicating captopril's oxygen radically scavenging properties in the morbidity effect. Captopril (Capoten) -- prevents conversion of angiotensin I to angiotensin II, a potent vasoconstrictor, resulting in increased levels of plasma renin and a reduction in aldosterone secretion. Adult Dose: 6.25-12.5 mg PO tid; not to exceed 150 mg tid Contraindications: Documented hypersensitivity; renal impairment. Interactions: NSAIDs may reduce hypotensive effects of captopril; ACE inhibitors may increase digoxin, lithium, and allopurinol levels; rifampin decreases captopril levels; probenecid may increase captopril levels; the hypotensive effects of ACE inhibitors may be enhanced when given concurrently with diuretics Pregnancy: C - Safety for use during pregnancy has not been established. Precautions: Category D in second and third trimester of pregnancy; caution in renal impairment, valvular stenosis, or severe CHF Calcium channel blockers -- While they have limited the use in ischemic causes of CHF, calcium channel blockers may prove to be useful in myocarditis-related myopathies. Amlodipine, in particular, perhaps due to its effect on nitric oxide, showed benefit in animal models and in a placebo controlled trial. Amlodipine (Norvasc) -- Relaxes coronary smooth muscle and produces coronary vasodilation, which, in turn, improves myocardial oxygen delivery. Benefits nonpregnant patients with systolic dysfunction, hypertension, or arrhythmias. Adult Dose: 2.5-5 mg PO qd 10 mg PO qd maximum Contraindications: Documented hypersensitivity Interactions: Fentanyl and alcohol may increase hypotensive effects; calcium channel blocker may increase cyclosporine levels; H2 blockers (cimetidine), erythromycin, nafcillin, and azole antifungals may increase toxicity (to avoid combination or monitor closely); carbamazepine may reduce bioavailability (avoid this combination); rifampin may decrease levels (monitor and adjust dose of calcium channel blocker) Pregnancy: C - Safety for the use during pregnancy has not been established. Precautions: The adjust dose in renal/hepatic impairment; may cause lower extremity edema; allergic hepatitis has occurred but is rare Loop diuretics -- These agents are used for the management of fluid overload. Furosemide (Lasix) -- Increases excretion of water by interfering with chloridebinding cotransport system which, in turn, inhibits sodium and chloride reabsorption in ascending loop of Henle and distal renal tubule. Adult Dose: 20-80 mg/d PO/IV/IM; titrate up to 600 mg/d for severe edematous states 77 Contraindications: Documented hypersensitivity; hepatic coma; anuria; state of severe electrolyte depletion Interactions: Metformin decreases furosemide concentrations; furosemide interferes with hypoglycemic effect of antidiabetic agents and antagonizes musclerelaxing effect of tubocurarine; auditory toxicity appears to be increased with coadministration of aminoglycosides and furosemide; the hearing loss of varying degrees may occur; the anticoagulant activity of warfarin may be enhanced when taken concurrently with this medication; the increased plasma of the lithium levels and toxicity are possible when taken concurrently with this medication. Pregnancy: C - Safety for the use during pregnancy has not been established. Precautions: Perform frequent serum electrolyte, CO2, glucose, creatinine, uric acid, calcium, and BUN determinations during the first few months of therapy and periodically thereafter Cardiac glycosides -- These agents decrease, the AV nodal conduction primarily by increasing of vagal tone. They may aid in the dysrhythmia and in CHF aspects of myocarditis. Digoxin (Digitek, Lanoxicaps, Lanoxin) -- Cardiac glycoside with direct inotropic effects in addition to indirect effects on the cardiovascular system. Acts directly on cardiac muscle, increasing myocardial systolic contractions. Its indirect actions result in the increased carotid sinus nerve activity and the enhanced sympathetic withdrawal for any given increase in mean arterial pressure. Adult Dose: 0.125-0.375 mg PO qd Contraindications: Documented hypersensitivity; beriberi heart disease; idiopathic hypertrophic subaortic stenosis; constrictive pericarditis; carotid sinus syndrome Interactions: Many medications can alter levels of digoxin, which have a fairly narrow therapeutic window. Pregnancy: C - Safety for the use during pregnancy has not been determined. Precautions: Patients with myocarditis seem to be particularly sensitive to digoxin toxicity Angiotensin converting enzyme inhibitors - These agents are beneficial in the management of blood pressure and LV function in heart failure. Captopril, in particular, has been shown to be beneficial in the treatment of the significant left ventricular dysfunction. Other ACE inhibitors have not shown the same effect in animal trials, indicating captopril's oxygen radical scavenging properties in the morbidity effect. Captopril (Capoten) -- Prevents conversion of angiotensin I to angiotensin II, a potent vasoconstrictor, resulting in increased levels of plasma renin and a reduction in aldosterone secretion. Adult Dose: 6.25-12.5 mg PO tid; not to exceed 150 mg tid Contraindications: Documented hypersensitivity; renal impairment Interactions: NSAIDs may reduce hypotensive effects of captopril; ACE inhibitors may increase digoxin, lithium, and allopurinol levels; rifampin decreases captopril 78 levels; probenecid may increase captopril levels; the hypotensive effects of ACE inhibitors may be enhanced when given concurrently with diuretics Pregnancy: C - Safety for the use during pregnancy has not been determined. Precautions: Category D in second and third trimester of pregnancy; caution in renal impairment, valvular stenosis, or severe CHF Calcium channel blockers -- While they have the limited use in ischemic causes of CHF, calcium channel blockers may prove to be useful in myocarditis-related myopathies. Amlodipine, in particular, perhaps due to its effect on nitric oxide, showed benefit in animal models and in a placebo controlled trial. Amlodipine (Norvasc) – Relaxes coronary smooth muscle and produces coronary vasodilation, which, in turn, improves myocardial oxygen delivery. Benefits nonpregnant patients with systolic dysfunction, hypertension, or arrhythmias. Adult Dose: 2.5-5 mg PO qd, 10 mg PO qd maximum Contraindications: Documented hypersensitivity Interactions: Fentanyl and alcohol may increase hypotensive effects; calcium channel blocker may increase cyclosporine levels; H2 blockers (cimetidine), erythromycin, nafcillin, and azole antifungals may increase toxicity (avoid combination or monitor closely); carbamazepine may reduce bioavailability (avoid this combination); rifampin may decrease levels (monitor and adjust dose of calcium channel blocker) Pregnancy: C - Safety for the use during pregnancy has not been determined 3.12. Further Inpatient Care Patients admitted to the hospital are treated for the complications of myocarditis. Patients thought to have the disease often undergo an endomyocardial biopsy, although some believe this invasive procedure should be restricted to posttransplant and HIV-positive patients who have an increased likelihood of identifying a specific and treatable cause. Although temporary pacemaker placement for advanced degrees of the heart block is indicated, in the setting of myocarditis, these conduction disturbances are usually transitory. Therefore, permanent pacemaker placement usually is not necessary. Bedrest with restriction of activity and sodium intake is beneficial. Patients with fulminant heart failure may require transplantation, which can be life saving. Unfortunately, these patients have a higher rate of rejection than patients whose underlying cause of heart failure is not myocarditis. 3.13. Further Outpatient Care 79 The clinician may consider the placement of a Holter monitor to recognize dysrhythmias on an outpatient basis. o This may be done after the initial ED evaluation of a patient who shows no sign of acute dysrhythmia, CHF, or other complication. o Placement may also be done after the initial inpatient treatment. Upon discharge from the hospital, all patients with myocarditis should have follow-up visits to a cardiologist. 3.1.4. In/Out Patient Meds Treatment of pain with a narcotic analgesic (eg, acetaminophen with codeine) is appropriate. Avoid nonsteroidal anti-inflammatory drugs (NSAIDs), which are relatively contraindicated in this condition. 3.15. Complications Congestive heart failure: o Pulmonary edema; o Cardiogenic shock; o Cardiac failure. Dilated cardiomyopathy. Dysrhythmias. Recurrent myositis. 3.16. Prognosis 80 Most cases are believed to be clinically silent and resolve spontaneously without sequelae; therefore, making accurate statements concerning the prognosis of myocarditis is difficult. Patients presenting with CHF experience morbidity and mortality based on the degree of left ventricular dysfunction. Of patients presenting with the cardiogenic shock, elderly patients and patients with giant cell arteritis have a poor prognosis. One half of patients presenting with the new-onset CHF experience the considerable improvement of cardiac function with treatment. One fourth of patients presenting with CHF stabilize with compromised cardiac function. The conditions of the remaining one fourth of patients continue to deteriorate. Patients requiring transplantation have an increased risk of recurrent myocarditis and graft rejection. 3.17. Patient Education Patients are advised to restrict activity since studies have shown that increased activity promotes progression of inflammation. 3.18. Medical/Legal Pitfalls Myocarditis may be present subtly, but it should be considered in the patient who is presented with dyspnea and chest discomfort, particularly if the history includes a recent viral illness. o Careful physical examination looking for signs of CHF and pericarditis is helpful. Electrocardiography, ESR, and cardiac enzyme levels are useful screening tools. o Patients with the evidence of dysrhythmia, CHF, or thromboembolism must be admitted. 3.19. REFERENCES 1. Billingham ME. The diagnostic criteria of myocarditis by endomyocardial biopsy. // Sekiguchi M, Olsen EGJ, Goodwin JF, eds. Myocarditis and Related Disorders: Proceedings of the International Symposium on Cardiomyopathy and Myocarditis. New York: Springer-Verlag; 1985. – Р.133-137. 2. Myocarditis: emergency department recognition and management./ Brady W.J., Ferguson J.D., Ullman E.A., Perron A.D. // Emerg Med Clin North Am. – 2004, Nov. – Vol. 22, №4. – Р.865-885. 3. Braunwald E, ed: Myocarditis. //Heart Disease. 6th ed. – 2001. – Р.17831793. Feldman AM, McNamara D. Myocarditis. //N Engl J Med. – 2000, Nov 9. Vol.343, №19. – Р.1388-1398. 81 4. MRI of acute myocarditis: a comprehensive approach based on various imaging sequences. / Laissy J.P., Messin B., Varenne O., et al. //Chest. – 2002, Nov. - Vol.122, №5. – Р.1638-1648. 5. Liu P.P., Mason J.W. Advances in the understanding of myocarditis. //Circulation. – 2001, Aug 28. - Vol.104, №9. – Р.1076-1082. 6. The coxsackie-adenovirus receptor (CAR) is used by reference strains and clinical isolates representing all six serotypes of coxsackievirus group B and by swine vesicular disease virus./ Martino T.A., Petric M., Weingartl H., et al. // Virology. - 2000 May 25. - Vol.271, №1. – Р.99-108. 7. A clinical trial of immunosuppressive therapy for myocarditis. The Myocarditis Treatment Trial Investigators. / Mason J.W., O'Connell J.B., Herskowitz A., et al. // N Engl J Med. – 1995. - Vol. 333. – Р.269-275. 8. Intravenous immune globulin in the therapy of myocarditis and acute cardiomyopathy./ McNamara D.M., Rosenblum W.D., Janosko K.M., et al. // Circulation. – 1997. - Jun 3. - Vol.95, №11. – Р.2476-2478. 9. Effect of amlodipine on morbidity and mortality in severe chronic heart failure. Prospective Randomized Amlodipine Survival Evaluation Study Group. / Packer M., O'Connor C.M., Ghali J.K., et al. // N Engl J Med. - 1996 Oct 10. Vol.335, №15. – Р.1107-1114. 10. Pisani B., Taylor D.O., Mason J.W. Inflammatory myocardial diseases and cardiomyopathies.// Am J Med. – 1997. - Vol.102. – Р.459-469. 11. Elevations of cardiac troponin I associated with myocarditis. Experimental and clinical correlates. / Smith S.C., Ladenson J.H., Mason J.W., Jaffe A.S. // Circulation. - 1997 Jan 7. - Vol.95, №1. – Р.163-168. 12. Stevens L.W. Diseases of the myocardium. // Bennett JC, Plum F, eds. Cecil Textbook of Medicine. 20th ed. Philadelphia, Pa: WB Saunders Co, 1996. – Р.328-331. 13. Vallejo J., Mann D.L. Antiinflammatory therapy in myocarditis.// Curr Opin Cardiol. - 2003 May. - Vol.18, №3. – Р.189-193. 14. Randomized, placebo-controlled study for immunosuppressive treatment of inflammatory dilated cardiomyopathy: two-year follow-up results. / Wojnicz R., Nowalany-Kozielska E., Wojciechowska C., et al. // Circulation. - 2001 Jul 3. Vol.104, №1. – Р.39-45. 82 3.20. Test control 1. Rheumatic fever is systemic inflammatory disease of connective tissue with specific location of process in: А). smooth muscle and skin В). cardiovascular system С). pulmonary vessels D). musculoskeletal system E). synovial bursa 2. The specific diagnostic sign of rheumatic fever: A). Aschoff- Talalaev’s nodules B). Anitschkow myocytes C). granulocytus basophillus textus (labrocyte) D). lymphoid and plasmatic cells, E). All listed 3. The most high title of antistreptococcal antibody is determined at: A). acute rheumatic fever and III (maximal) level of activity B). II level of activity C). I level of activity D). persistent (permanent) recurrent rheumatic fever E). latent rheumatic fever 4. For rheumatic polyarthritis which is a main sign of primary rheumatic fever occasionally recurrent rheumatic fever is typical: A). polyarthralgia B). migrating synoviitis C). deformation of small joins D). affection of ankle and wrist joins E). intractable pain in muscles and myositis 5. Systolic pressure in the left ventricle is: A). 11-25 mmHg B). 26-35 mmHg C). 36-65 mmHg D). 66-100 mmHg E). 110-120 mmHg 6. Diastolic pressure in the left ventricle is: A). 0-8 mmHg B). 8-12 mmHg C). 14-20 mmHg D). 22-35 mmHg E). 36-65 mmHg 83 7. Mass of the heart is: A). 200-300 g B). 400-600 g C). 500-700 g D). 100-200 g E). 50-100 g 8. Amount of liquid in pericardium is: A). 10-15 ml B). 20-40 ml C). 40-100 ml D). 100-150 ml E). more than 150 ml 9. At recurrent rheumatic fever of II and III level of activity with carditis it is necessary to prescribe: A). corticosteroids B). NSAID (non-steroid anti-inflammatory drugs) C). antibiotics D). vitamin B E). cardiac glycosides 10.The only effective medication method of rheumatic replace prevention is: A). penicillin-injection B). whole-year bicillin prophylactics C). treatment streptococcal infections D). immunosuppresants E). all listed 11.Morphologic changes that characterize pure rheumatic valvular stenosis include all the following except: A). leaflet fusion along the commissures B). shrunken, retracted cusps C). thickening of the chordae tendinae D). thickening of the valvular cusps E). rupture of the papillary muscles 12.The most often agent of immunodeficiency pneumonia is all exept: A). pneumocyst B). Escherichia coli C). pathogenic fungi D). Cytomegalovirus E). Proteus 84 13.Dilation and hypertrophy of the left ventricle occur in: A). mitral stenosis B). mitral regurgitation C). tricuspid regurgitation D). pulmonary stenosis E). tricuspid regurigtation 14.Which of the following disorders in least likely to result I left ventricular strain: A). systemic hypertension B). aortic regurgitation C). mitral stenosis D). vernacular aneurysm E). coronary atherosclerotic disease 15.Atrial fibrillation is least likely to be associated with: A). mitral regurigtation B). mitral stenosis C). aortic stenosis D). coronary atherosclerotic disease E). tricuspid regurigtation 16.Potential symptoms and sings in moderate aortic stenosis include all the following except: A). angina pectoralis B). effort syncope C). peripheral edema D). paroxysmal noctural dyspnea E). cough In aortic stenosis, life expectancy after the onset o fsinificant symptoms averages: A). less than 5 years B). 5 to 8 years C). 9 to11 years D). 12 to15 years E). 15 to30 years 17.Characteristic sings of severe aortic regurgitation include (more than one answer may be correct) : A). pistol- shot sound over the femoral artery B). Corrigan’s pulse C). Austin Flint murmur D). systolic head bobbing E). Quincke capillary pulsation 18.--Pure tricuspid regurgitation is usually: 85 A). associated with rheumatic heart disease B). a functional disorder associated with right ventricular failure C). associated with acute myocardial infarction D). a functional disorder associated with left ventricular failure E). none of the above 20. Morphologic changes that characterize pure rheumatic valvular regurgitation include all the following, except: A). shrunken, retracted cusps B). Shortened, fused chordae tendinae C). rupture of the papillary muscles D). enlargement of the valvular orifice E). leaflet fusion along the commissures 21. Depending on the reason for that causing myocarditis, there are following pathogenetic versions of myocardites: A).Infectious-toxic B). Infectious C). Allergic D). Toxic-allergic E). Entire enumerated 22. Rheumatic myocardites most frequently are caused by: A). Enterococci B). Enterovirus C). b- hemolytic streptococcus is type A D). Parvovirus B12 E). Mycobacteria 23. Reason of myocardites in the majority of the cases is: A). Virus of measles B). Brucella C). Virus of herpes G). Mycobacteria E). The coxacievirus 24. Degree of occurrence in the autopsies is indicative the inflammatory defeat of the myocardium: A). 10-12% B). 1-5% C). 50-70% D). 4-9% E). 20-40% 25. Depending on the development degree of myocardites is separated following pathogenetic phases, besides: A). Infectious-toxic B). Immunological C). Idiopathic D). Dystrophic 86 E). Myocardiosclerotic 26. On the basis of the prevalence is separated the following types of myocardites: A). Abortive B). Focus and diffuse C). Sharp and subacute D). Recidivating E). Chronic latent form 27. Abramov's Fiedler myocarditis is: A). Fungus myocarditis B). Tubercular myocarditis C). Virus myocarditis D). Idiopathic myocarditis E). Parasitic myocarditis 28. By myocardites on ECG are observed the following signs, besides: A). Change of segment ST in the standard removals B). Inversion of tooth T C). Extrasystole D). Blockades E). Pathologic tooth QS 29. Place of the best listening of the mitral valve: A). II intercostals space to the left B). Botkin –Erb’s point C). Lower third of breast bone D). At the level of the top of heart E). In second intercostals space to the right side 30. Unfavorable factors, which aggravate myocardites: A). An increase of the pressure in the left atrium B). Significant reduction in the contracting ability of the myocardium of left ventricle C). Disturbance of conductivity D). Arrhythmia of high gradations E). Entire enumerated is correct Etalons of answers: 1-E, 2- C, 3-E, 4- D, 5- C, 6- B, 7- D, 8- E, 9- D, 10-E. 11-E, 12- A, 13- D, 14- A, 15- C, 16-A, 17- E, 18- E, 19-E, 20- E, 21-A, 22- E, 23- E, 24- D, 25- B, 26-D, 27-A, 28-E, 29- B, 30- C. Contents List of abbreviation-----------------------------------------------------------------3 1. ACUTE RHEUMATIC FEVER-----------------------------------------------4 1.1. Definition--------------------------------------------------------------------4 1.2. Epidemiology---------------------------------------------------------------4 1.3. Etiology----------------------------------------------------------------------5 87 1.4. Pathogenesis---------------------------------------------------------------- 6 1.5. Pathomorphology---------------------------------------------------------- 6 1.6. Pathology--------------------------------------------------------------------7 1.7. Classification----------------------------------------------------------------7 1.8. Acute rheumatic fever-----------------------------------------------------.8 1.9. Repeated rheumatic fever--------------------------------------------------8 1.10. Clinical feature------------------------------------------------------------8 1.11. Other associated features-----------------------------------------------10 1.12. Diagnosis-----------------------------------------------------------------11 1.13. Guidelines for the diagnosis o f initial attack of theumatic fever, (Jones – Kisel, updated 1992)----------------------------------------- ----------11 1.14. Laboratory tests----------------------------------------------------------13 1.15. Acute phase reactants---------------------------------------------------13 1.16. Course---------------------------------------------------------------------13 1.17. Management--------------------------------------------------------------13 1.18. Recurrence of rheumatic fever and prophylaxis---------------------15 1.19. Prognosis------------------------------------------------------------------16 1.20. References----------------------------------------------------------------17 2. VALVULAR HEART DISEASES------------------------------------------17 2.1. Mitral valve diseases-----------------------------------------------------17 2.1.1. Introduction-------------------------------------------------------17 2.1.2. Definition----------------------------------------------------------18 2.1.3. Normal mitral valve ---------------------------------------------18 2.1.4. Mitral valve disease----------------------- -------------------- 20 2.1.5. Epidemiology ------------------------------- --------------------20 2.1.6. Classification of valve heart disease, (MUHARLAMOV 1977) ------------------------------ 20 2.2. Mitral stenosis------------------------------------------------------------ 20 2.2.1. Etiology----------------------------------------------------------- 20 2.2.2. Pathogenic and hemodynamic abnormalities---------------- 21 2.2.3. Pathophysiology of mitral stenosis---------------------------- 22 2.2.4. Clinical features------------------------------------------------- 22 2.2.5. Investigations---------------------------------------------------- 23 2.2.6. Diagnosis----------------------------------------------------------24 2.2.7. Management------------------------------------------------------25 2.2.8. Prognosis--------------------------------------------------------- 27 2.3. Mitral regurgitation------------------------------------------------------ 27 2.3.1. Etiology------------------------------------------------------------27 2.3.2. Pathogenic and hemodynamic abnormalities-----------------28 2.3.3. Clinical picture--------------------------------------------------- 30 2.3.4. Laboratory examination------------------------------------------30 2.3.5. Diagnosis-----------------------------------------------------------31 2.3.6. Management-------------------------------------------------------32 2.4. Aortic valve disease------------------------------------------------------33 88 2.4.1. Etiology-----------------------------------------------------------33 2.4.2. Clinical picture--------------------------------------------------- 33 2.4.3. Diagnosis and management--------------------------------------34 2.4.4. Classification----------------------------------------------------- 34 2.5. Aortic stenosis------------------------------------------------------------ 34 2.5.1. Etiology------------------------------------------------------------ 35 2.5.2. Pathogenic and hemodynamic abnormalities----------------- 35 2.5.3. Clinical features---------------------------------------------------35 2.5.4. Laboratory studies------------------------------------------------ 37 2.5.5. Diagnosis-----------------------------------------------------------38 2.5.6. Differential diagnosis-------------------------------------------- 38 2.5.7. Management-------------------------------------------------------40 2.6. Aortic regurgitation----------------------------------------------------- 41 2.6.1. Definition---------------------------------------------------------- 41 2.6.2. Etiology------------------------------------------------------------ 41 2.6.3. Pathogenic and hemodynamic abnormalities-----------------41 2.6.4. Clinical features-------------------------------------------------- 42 2.6.5. Investigation------------------------------------------------------ 43 2.6.6. Diagnosis-----------------------------------------------------------44 2.6.7. Differential diagnosis---------------------------------------------45 2.6.8. Management-------------------------------------------------- ---- 46 2.6.9. References--------------------------------------------------------- 46 2.7. Tricuspid regurgitation------------------------------------------------ 48 2.7.1. Frequency--------------------------------------------------------- 48 2.7.2. Background------------------------------------------------------- 49 2.7.3. Classification of tricuspid regurgitation-----------------------49 2.7.4. Etiology------------------------------------------------------------49 2.7.5. Pathophysiology------------------------------------------- ----- 51 2.7.6. Clinical features----------------------------------------- -- ------51 2.7.7. Laboratory changes------------------------------------- -- ------52 2.7.8. Instrumental examination------------------------------ -- ------52 2.7.9. Other Tests------------------------------------------------ ------- 53 2.7.10. Procedures--------------------------------------------- ---------53 2.7.11. Classification of tricuspid regurgitation--------- -----------53 2.7.12. Differential diagnostics--------------------------- ------------54 2.7.13. Management------------------------------------ ---------------55 2.7.14. Further Inpatient Care----------------------------- -----------59 2.7.15. Further Outpatient Care---------------------------------------59 2.7.16. In/Out Patient Meds------------------------ -------------------59 2.7.17.Complications---------------------------------------------------59 2.7.18. Prognosis--------------------------------------------------------60 2.7.19. Patient Education----------------------------------------------60 2.7.20. Medical/Legal Pitfalls-----------------------------------------60 2.7.21. References----------------------------------------------- -------60 89 3. MYOCARDITIS-------------------------------------------------------- -------64 3.1. Background------------------------------------------------------- --------64 3.2. Frequency of Myocarditis-----------------------------------------------64 3.3. Mortality/Morbidity------------------------------------------------------64 3.4. Etiology of myocarditis------------------------------------------------- 65 3.5. Pathophysiology----------------------------------------------------------66 3.6. Classification of myocarditis, (Palaev N.R. 1981)-------------------67 3.7. Clinical manifestation of myocarditis----------------------------- ----70 3.8. Laboratory – instrumental investigation-------------------------------72 3.9. Procedures-----------------------------------------------------------------74 3.10. Criterions of the histological diagnostics----------------------------74 3.11. Management------------------------------------------------------------ 75 3.12. Further Inpatient Care------------------------------------------------- 79 3.13. Further Outpatient Care---------------------------------- --------- ----80 3.14. In/Out Patient Meds------------------------------------ ----------------80 3.15. Complications----------------------------------------- --------------- --80 3.16. Prognosis----------------------------------------------- --------- --------81 3.17. Patient Education-------------------------------------- -----------------81 3.18. Medical/Legal Pitfalls---------------------------------------- ----------81 3.19. References---------------------------------------------------- -----------81 3.20. Test control--------------------------------------------------- ---------83 90 Отпечатано в КГМА г. Караганда, Ул. Гоголя, 40 91 92 93